INTRODUCTION
Mastitis is defined as the inflammation of the mammary gland and is considered the most important disease within dairy goat herds worldwide. Its prevalence varies between regions and countries; however, it ranges between 5 and 36% depending on several factors, some specific to the animal such as age and number of births, and others such as management measures and the nature of the infectious agents involved in the disease (Wakwoya et al., 2006; Contreras et al., 2007; Megersa et al., 2010; Alemu &Abraha, 2017). In Colombia, the reported prevalence of subclinical mastitis in goats is 26%, and the main associated risk factors are age (more than 3 years), more than five births, days in milk production (>60 days), absence of disinfection processes before milking, poor body condition and presence of teat lesions (Tarazona-Manrique et al., 2022).
Its denomination varies according to its clinical course and infectious etiology. According to its clinical manifestation, it is classified into clinical and subclinical mastitis. In the first form, there are observable changes in the mammary gland such as inflammation, redness, and pain, as well as alterations in the milk produced due to enzymatic processes typical of inflammation, added to the bacterial or fungal growth. If the treatment is conducted properly, the possibility of losing the affected mammary half decreases; however, if the process is very serious, the animal may lose its productive capacity and, consequently, be discarded (Andrade-Becerra et al., 2012, 2018). In the second form, there are no clinical symptoms in the mammary gland, as well as no apparent alterations in the milk; however, it has been shown that the quality and quantity of milk decrease by up to 45%. This form of mastitis presents difficulties for its treatment due to the failure in detection causes the process to take a chronic course and gradually affects the goat’s health status and its productive capacity (Contreras et al., 2007; Megersa et al., 2010; Tarazona-Manrique et al., 2019; Novac & Andrei, 2020).
On the other hand, according to its infectious etiology, it can be classified as contagious and environmental mastitis. Contagious mastitis is transmitted between animals, a situation that occurs mainly during milking processes due to teat disinfection failures. The main etiological agents involved in this process are coagulase-positive staphylococci (CPS) such as Staphylococcus aureus and some Streptococci such as Streptococcus agalactiae (Contreras et al., 2011; Andrade-Becerra et al., 2012, 2018). Environmental mastitis is mainly caused by gram-negative bacteria, species of coagulase-negative staphylococci (CNS), some streptococci, and fungi, because these agents are found mainly in the environments where the animals stay, such as pens contaminated with faecal matter and paddocks with little infiltration capacity (Contreras & Rodríguez, 2011; Andrade-Becerra et al. 2012; Andrade-Becerra et al. 2018). Regardless of their infectious etiology, microorganisms can cross the teat cistern and gradually colonize the interior of the mammary gland, generating inflammatory processes that vary in severity according to the infectious agent involved and the immune response capacity of the goat (Machado, 2018; Omar & Mat-Kamir, 2018).
In Colombia, studies related to this topic are scarce, however, some have been developed in which the risk factors associated with its manifestation have been determined (Tarazona-Manrique et al., 2022), but the main infectious agents involved in its manifestation have not yet been determined and, therefore, it has not been possible to generate adequate control mechanisms according to its infectious etiology. Therefore, the objective of this study was to determine the bacterial and fungal infection etiology involved in the prevalence of subclinical mastitis in dairy goats in Boyacá.
MATERIALS AND METHODS
Study and Animals
The animals and farms present in this article are part of a previous study published by Tarazona-Manrique et al. (2022), in which 25 goat farms were included. Of these, 256 lactating animals with an average age of 27 months were included. In all production systems, milking was done manually once a day in the morning. Animals with clinical manifestations of mastitis were not included (CMT >4), likewise, only animals between 3 and 120 days in milk (DIM) production were sampled.
Sampling and Transport
The sampling was performed as mentioned in Tarazona-Manrique et al. (2022), where a disinfection process was conducted on each half before the collection, taking approximately 20 mL of milk from each udder half (each half was considered as an individual sample) (NMC, 2009). Samples were labelled and transported at 4-6 °C without additives to the Laboratory of Milk Quality Analysis and Mastitis Control at the Universidad Pedagógica y Tecnológica de Colombia in Tunja-Boyacá.
California Mastitis Test
The CMT procedure was performed in the laboratory within five hours after taking the sample. For this, 3 mL of milk was taken and mixed in a CMT plastic palette with 3 mL of the reagent (alkyl aryl sulfonate), followed by a circular motion for 20 s (NMC, 2009; Persson & Olofsson, 2011). The score was given between 0 and 5, where the gelation results were analysed as follows: CMT 0 and CMT 1: negative sample, CMT 2-3: positive sample for subclinical mastitis, CMT 4-5: positive sample for clinical mastitis (Persson & Olofsson, 2011).
Microbiological Tests and Microbial Identification
Positive samples for subclinical mastitis (CMT 2 and 3) were subjected to microbiological culture and biochemical tests to identify the microorganisms present. For this, the microbial culture and biochemical identification methodologies proposed by the National Mastitis Council of the United States were followed, due to these being used as a routine in the laboratory and other investigations of the group (Andrade-Becerra et al., 2014; Tarazona-Manrique et al., 2019).
Milk samples were cultivated in blood agar for 24-48 hours and in MacConkey agar. After this, Gram coloration was performed. The Gram-positive cocci were tested using the coagulase test and differentiated as positive and negative coagulase. Then the colony was cultivated in ID32 STAPH, to identify and confirm gender and species, thus Staphylococcus aureus differed from other CPS. For Streptococcus spp, CAMP, hydrolysis of esculin, Hippurate, and inulin, and growth in NaCl were used. For Gramnegative bacilli: oxidase, indole, triple sugar, Methyl Red-Voges Proskauer (RM-VP), Lys Agar decarboxylase (LIA), urea, and citrate tests were used. All samples were cultivated in Saboureaud agar for yeast and molds and on Hayflick agar for 7 days for the detection of Mycoplasma spp (Andrade-Becerra et al., 2014; NMC, 2017, Tarazona-Manrique et al., 2019).
RESULTS AND DISCUSSION
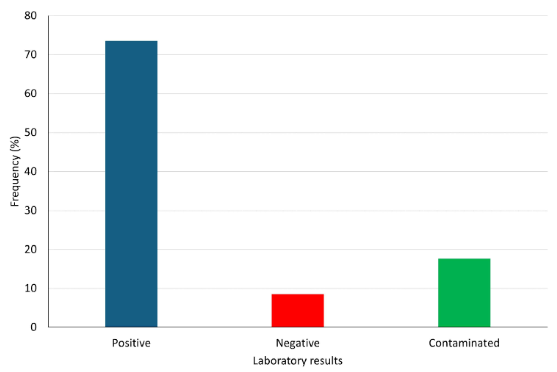
The general prevalence of subclinical mastitis was 26.56% (136/512 samples). However, not all 136 samples resulted positive in the laboratory (Figure 1).
The microorganisms found in positive samples are shown in Table 1. The main microorganisms associated with the prevalence of subclinical mastitis in dairy goats in Boyacá correspond to CPS and CNS.
Table 1. A list of microorganisms in milk samples of dairy goats with subclinical mastitis
| Microorganism | Samples | |
|---|---|---|
| n | % | |
| Coagulase-negative Staphylococci | 33 | 24.26 |
| Staphylococcus aureus | 13 | 9.55 |
| Coagulase-positive Staphylococci | 26 | 19.11 |
| Streptococcus agalactiae | 6 | 4.41 |
| Other streptococci | 6 | 4.41 |
| Mycoplasma spp | 3 | 2.20 |
| Escherichia coli | 8 | 5.88 |
| Fungi | 3 | 2.20 |
| Contaminated | 26 | 19.11 |
| Not growth | 12 | 8.82 |
| Total | 136 | 100 |
The results presented here correspond, to the best of the authors’ knowledge, to the first report of microorganisms associated with the prevalence of subclinical mastitis in dairy goats in Boyacá and Colombia. The results shown in Figure 1 show that more than 70% of the samples with subclinical mastitis presented microbial growth, these results agree with those reported by Gelasakis et al. (2016) and Hristov et al. (2016) who found that around 80% of the samples with subclinical mastitis are positive for microbial growth of various kinds, mainly bacterial.
It is interesting to point out that in 8.82% of the samples were negative to microorganisms, and thus was probably due to aseptic processes that can trigger an increase in the somatic cell count, such as the beginning of lactation, or some other risk factors (Andrade-Becerra et al., 2018; Tarazona-Manrique et al., 2022). The goats in this study live in steep landscapes with the presence of thorny plants and an environment that can be considered hostile, facilitating the occurrence of subclinical mastitis without microbial presence.
The main bacterial agents corresponded to two large groups, CPS and CNS. These results agree with the reported by Gelasakis et al. (2016) and Mishra et al. (2018) in small goat’s dairy farms. Its presence in dairy herds has been associated with failures in disinfection management in milking, this is a result that can be corroborated with the risk factors determined in Tarazona-Manrique et al. (2022) where failures in the disinfection process a factor that was statistically related to the presence of subclinical mastitis in the same animals of this study.
Hristov et al. (2016) determined that the most common pathogens in subclinical mastitis were Staphylococci, especially those of the negative coagulase genus (52.75%), a value much greater than that determined in this study. Another important aspect of the study by Hristov is the presence of co-infections between Staphylococci species and gram-negative microorganisms in subclinical mastitis, an aspect that was not found in this study where none of the samples showed coinfection.
Marogna et al. (2012) and Suwito et al. (2019) showed that, in order of importance, the pathogens associated with subclinical mastitis in goats were Staphylococci, Streptococci, Enterococci, and Mycoplasma species, results similar to those found in this study, showing that despite the geographical differences from the studies it is possible to consider that these pathogens may be the main agents involved in all cases of subclinical mastitis in herds worldwide, therefore, the management measures that have been reported worldwide can be applied to Colombian herds hoping to find similar results.
Studies in specialized national dairy cattle farms have shown the transmission of Streptococci species from the milkers to cows, as well as from the environment, demonstrating the importance of health controls of workers and control of the milking routine to reduce their presence as causative agents of subclinical mastitis (Cobo-Angel et al. 2018, 2019). This is a situation that could have occurred in this study, however, the investigation of the sources of contamination or transmission of each of the pathogens was not performed.
An important aspect to take into account is the presence of rare pathogens such as fungi and gram-negative bacteria such as Escherichia coli as agents that trigger subclinical mastitis because these pathogens have been commonly associated with clinical manifestations of the disease due to biological factors, such as virulence factors in E. coli and the biological structure of fungi, which prevent efficient elimination by leukocytes in the mammary gland (McDougall et al., 2010). Other studies have reported the presence of microbial agents such as Listeria spp, Neisseria spp, and Bacillus spp as agents of subclinical mastitis and especially important due to their impact on public health, principally for Listeria spp (Okoko et al., 2020). Although in this study they were not determined, it is important to carry out epidemiological surveillance of these important agents for public health.
It has also been proposed that the presence of agents such as small ruminant lentiviruses predisposes bacterial colonization of the udder, however, there are still unknowns about their true role (Urbañska et al., 2021). Another important aspect to consider mastitis is the microbiome present in the milk because it varies according to the type of mastitis present, therefore, this could directly influence the ability it has the host to eliminate the infection because a multiplicity of potential pathogenic bacterial agents can aggravate the clinical outcome, as well as reduce the efficacy of antibiotic treatments (Polveiro et al., 2022).
In conclusion, for the first time in Boyacá, CPS and CNS have been found as the main infectious agents involved in subclinical mastitis in goat herds. However, it is also necessary to highlight the presence of other agents such as fungi and gram-negative bacteria such as E. coli.












 uBio
uBio