Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista Peruana de Biología
versión On-line ISSN 1727-9933
Rev. peru biol. v.17 n.2 Lima ago. 2010
A fortuitous ant-fern association in the Amazon lowlands of Peru
Una asociación fortuita planta-hormiga en la Amazonía baja del Perú
Blanca León1,2,3 and Kenneth R. Young1
1 Department of Geography & the Environment, University of Texas at Austin.
Email Blanca León: blanca.leon@mail.utexas.edu
Email Kenneth R. Young: kryoung@austin.utexas.edu
2 Plant Resources Center, University of Texas at Austin.
3 Museo de Historia Natural, UNMSM, Av. Arenales 1256, Lima-14, Peru.
Abstract
Myrmecophyly is best documented among tropical epiphytes, and myrmecotrophy occurs especially among domatia-forming plants. We report the case of Polytaenium cajenense with "external" domatia and its use by the same symbiont ant as its host Cordia nodosa. This case is interpreted as the result of stochastic events and their relations to pre-existing adaptations of all species involved.
Keywords: myrmecophytes, ferns, monilophytes, Polytaenium, Azteca, Amazonia, Peru.
Resumen
Mirmecofilia ha sido documentada para epifitas, y mirmecotrofia ocurre especialmente en aquellas con desarrollo de domacios. Se reporta el caso de Polytaenium cajenense con domacio "externo" y su uso por la misma hormiga simbionte que su hospedero Cordia nodosa. Este caso se interpreta como el resultado de eventos e interacciones aleatorias entre todas las especies involucradas con adaptaciones pre-existentes.
Palabras claves: mirmecófitos, helechos, Polytaenium, Azteca, amazonía, Perú.
The diverse kinds of associations of vascular plants with ants are themes of both evolutionary and ecological interest in the tropics (e.g. Davidson 1988, Davidson et al. 1988, Huxley & Cuttler 1991, Beattie & Hughes 2002, Heil & McKey 2003, Rico-Gray & Oliveira 2007). About 200 Neotropical plant taxa are known to present this kind of interaction (Beattie 1989). Myrmecophyly (Benson 1985) is considered to be beneficial to one or both of the participants through defense against predation, and the provision of nesting space and/or food rewards.
Morphological adaptations to myrmecophyly in ferns include the development of hollow structures or domatia (e.g. Gómez, 1974, Benzing 1991) and nectaries (e.g. Koptur et al. 1982, Rashbrook et al. 1991). These are similar in function to those observed in flowering plant species, but are independently evolved. Only two fern genera have been reported to include myrmecophytes, meaning that they develop myrmecodomatia. These are genera in Polypodiaceae, Microgramma subgenus Solanopteris for the Neotropics and Lecanopteris for the Old World.
Myrmecotrophy is best documented among tropical epiphytes (Rico Gray & Oliveira 2003), and it has been mostly found among domatia-forming plants (Heil & McKey 2003). As mentioned by Benzing (1991), there are several factors involved that may be influential in affecting ant usage of plants and the nature of the reciprocal relationships of the plants towards the ants.
Here we report on an epiphytic species of Polytaenium (Pteridaceae) from Peru found in association with an ant species of Azteca, a genus that includes several different lineages adapted to colonize tree species (Benson 1985), including obligate symbionts of Cordia nodosa (Yu et al. 2001). There were other additional plant species involved and we use our observations to tease apart the ecological and evolutionary processes likely at work.
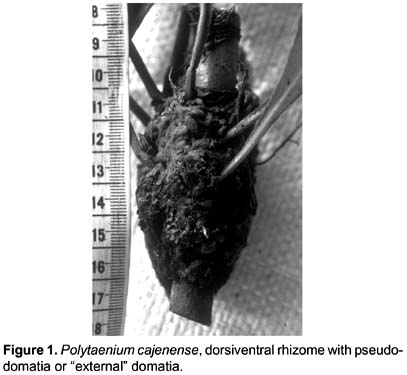
While recording fern species diversity in 1987, in a forest on sandy-loam soils near the settlement of Quebrada Castillo in the Amuesha community of Siete de Junio, in the Palcazu valley of central Peru at 350 m elevation (Young & León 1989), we found two adult sporophyte individuals of Polytaenium cajenense (Desv.) Benedict (León & Young 1085 USM-148929) on a 2m tall tree of Cordia nodosa Lamarck (León & Young 1067 USM-110731). Both the largest P. cajenense individual and its host were inhabited by the same ants of the genus Azteca (collected specimen identified to genus by E. O. Wilson, pers. comm., 6 Jan. 1988) Several gametophytes and young sporophytes were also found growing on the external surface of the mat of roots of the largest sporophyte individual (Fig. 1). The Cordia nodosa tree was surrounded in a 2m radius by a tree and two lianas, each with a distinct epiphyte community (Table 1). The ants apparently kept all surfaces directly in contact with the Cordia free of other plant species except for the fern and a Philodendron epiphyte (P. cf. heterophyllum Engl.) (Araceae)
The dorsiventral rhizome and the dense mat of roots of P. cajenense (Fig. 1) provided the bases for the development of a pseudo-domatia or "external" domatia (sensu Haufler et al. 2003) for the Azteca ants. Internally the mat of roots included debris and some roots of the Philodendron plant, and it appears that this plant might have grown simultaneously or perhaps before the establishment of the fern (Fig. 2).

and none have leaf nectaries. Wall spores found in Polytaenium have micro-ornamentations varying from scattered granules to clustered rod-like structures, none of which appear to resemble the spore walls of the polypods identified by Tryon (1985) as associated with myrmecochory.
There are a number of implications arising from these observations. Ecologically, it is striking how important spatial and temporal contingencies were to the development of the patterns reported here. As we interpret the scene, the Polytaenium spores landed on a Cordia tree probably before it was occupied by Azteca ants (see Yu et al. 2001 for colonization in Cordia nodosa). The Polytaenium might have grown to a size such that its rhizomes permitted an additional colonization space, through the development of a domatia-like space for the ants. Polytaenium is found only as an epiphyte and its rhizomes and roots grow in such a fashion that it can adhere to irregular shapes and on rough surfaces. The outside of the rhizome mass of the Polytaenium supported gametophytes and young sporophytes of additional plants of Polytaenium, showing that the site was favorable for its growth and establishment. The other epiphytic species in the assemblage observed, a Philodendron species, was also not removed/pruned by the Azteca ants. In fact, the aroid was partially rooted into the domatia of the Polytaenium. Thus, the series of events involved likely included colonization of a myrmecophyte tree species, starting with the two unrelated epiphytic plant species, followed by the Azteca ants, which later pruned other plant species that neighbored on or dispersed to the Cordia tree, and accompanied by the ant use of the domatia-like shelters of the Polytaenium. These events would have collectively created this palimpsest of a multi-species assemblage.
Evolutionarily, these observations point to a prevalence of diffuse selection acting upon the outcomes of stochastic dispersal events and as shaped by pre-existing species mutualisms (Azteca-Cordia, in this case) and by pre-existing morphological adaptations and growth responses of the two epiphytic plant species permitted by the ants to remain on the Cordia.
Bronstein et al. (2006, p. 419) pointed out that often ". . . outcomes of ant-plant associations are strongly context-dependent." For Azteca-Cecropia relations, Longino (1989) stressed the prevalence of spatial variation, effects of the order of colonization of host trees, and other stochastic events that lead to non-equilibrium conditions. Our observations show that several unlinked events can set up a fairly complicated multi-species assemblage. Those same authors, and as earlier also noted by Fonseca & Ganade (1996), note that frequently ant-plant associations are asymmetrical, with many costs and benefits (sensu Bronstein 1998) assumed by the ants. In this case, the ants provide pruning that presumably benefited and even encouraged successful colonization by the two epiphytic species. Benefits to the ants are not obvious, although the rhizome mass of the Polytaenium and the intertwined roots of the Philodendron at least appeared to provide a habitat space to the ants. They were not using that space for the raising of their young, an activity taking place in the axillary bud (domatia) of the Cordia tree.
There is also likely to be an important additional dimension associated with nutrients and nutrient limitations. Walkins et al. (2008) recently showed through the use of stable isotopes that ants benefit epiphytic nonspecialized (ie. non-myrmecophilous) ferns by bringing in substances (or producing wastes) that provided extra nitrogen to the fern roots. This was demonstrated in Antrophyum lanceolatum (=Polytaenium lanceolatum (L.) Benedict) in Costa Rica, with Phediole flavens ants, making their study directly relevant to our observations in Peru. The unspecialized domatia of the Costa Rican fern, nevertheless provided considerable benefits in terms of nitrogen when fortuitously the fern had ant residents (62% of 93 sampled ferns had ants). Epiphytism in general has been proposed as an evolutionary strategy for plants found in nutrient-limited tropical environments (Benzing 1991, Dubuisson et al. 2009). The rhizome mats of the Polytaeium of our study can form microenvironments attractive to ants. Presumably this fern species evolved its morphology to facilitate epiphytism, but secondarily may also benefit from increased nitrogen availability when ants utilize the "accidental" domatia. Speculatively, it also seems possible that the Philodendron would fortuitously have benefited from more nitrogen availability in what is a very low-nutrient environment (Young & León 1989).
In conclusion, we use observations of a particular example of a complex ant-plant species assemblage where the respective spatio-temporal arrangements appear to provide some insights into the complexity of ecological relationships that potentially give rise to mutualistic relationships, some of which are facultative, some of which are dependencies, and some of which are only expressed fortuitously.
Acknowledgments
We thank the community of Siete de Junio for their hospitality, E. O. Wilson for kindly identifying the ants, and CONCYTEC for financial assistance.
Literature cited
Beattie, A. J. 1989. Myrmecotrophy: plants fed by ants. Trends Ecol. Evol. 4: 172-176.
Beattie, A. J. & L. Hughes. 2002. Chapter 8. Ant-plant interactions. Pp. 211-235. In: C.M. Herrera and O. Pellmyr, eds. Plant-animal interaction: an evolutionary approach. Blackwell Sciences Ltd. Great Britain.
Benson, W. W. 1985. Amazon ant-plants. Pp. 239-266. In: G.T. Prance and T. Lovejoy, eds. Amazonia. Oxford. Pergamon Press.
Benzing, D. H. 1990. Vascular epiphytes. Cambridge University Press, Cambridge, United Kingdom.
Benzing, D. H. 1991. Myrmecotrophy: origins, operation, and importance. Pp. 353-418. In: C.R. Huxley and D.F. Cutler, eds. Ant-Plant Interactions. Oxford University Press, Oxford, United Kingdom.
Bronstein, J.L. 1998. The contribution of ant-plant protection studies to our understanding of mutualism. Biotropica 30: 150-161.
Bronstein, J.L, R. Alarcón & M. Geber. 2006. The evolution of plant-insect mutualisms. New Phytol. 172: 412-428.
Davidson, D. W. 1988. Ecological studies of Neotropical ant-gardens. Ecology 69: 1138-1152.
Davidson, D. W., J. T. Longino & R. R. Snelling. 1988. Pruning of host plant neighbors by ants: an experimental approach. Ecology 69:801-808.
Dubuisson, J.-Y., H. Schneider & S. Hennequin. 2009. Epiphytism in ferns: Diversity and history. Comptes Rendus Biologies 332: 120-128.
Fonseca, C.R. & G. Ganade. 1996. Asymmetries, compartments and null interactions in an Amazonian ant-plant community. J. Animal Ecol. 65: 339-347.
Gay, H. 1993. Ant-fed plants: an investigation into the uptake of ant-derived nutrients by the far-eastern epiphytic fern Lecanopteris Reinw. (Polypodiaceae). Biol. J. Linn. Soc. 50: 221-233.
Gómez, L. D. 1974. Biology of the potato-fern, Solanopteris brunei. Brenesia 4: 37-61.
Haufler, C. H., W. A. Grammer, E. Hennipman, T.A. Ranker, A.R. Smith & H. Schneider. 2003. Systematics of the ant-fern genus Lecanopteris (Polypodiaceae): testing phylogenetic hypotheses with DNA sequences. Syst. Bot. 28: 217-227.
Heil, M. & D. McKey. 2003. Protective ant-plant interactions as model systems in ecological an evolutionary research. Ann. Rev. Ecol. Syst. 34:425-453.
Huxley, C. R. & D.F. Cutler, eds. Ant-Plant Interactions. Oxford University Press, Oxford, United Kingdom.
Koptur, S., A.R. Smith & I. Baker 1982. Nectaries in some Neotropical species of Polypodium (Polypodiaceae): preliminary observations and analyses. Biotropica 14: 108-113.
Koptur, S. 1992. Extrafloral nectary-mediated interactions between insects and plants. Chapter 4: 81-130. In: E. A. Bernays, ed. Insect-Plant Interactions. Vol. IV. CRC Press, Boca Raton, FL.
Koptur, S., V. Rico-Gray & M. Palacios-Rios. 1998. Ant protection of the nectaried fern Polypodium plebeium in central Mexico. Amer. J. Bot. 85: 736-739.
Longino, J.T. 1989. Geographic variation and community structure in an ant-plant mutualism: Azteca and Cecropia in Costa Rica. Biotropica 21: 126-132.
Rashbrook, V. K., S. G. Compton & J. H. Lawton. 1991. Bracken and ants: Why is there no mutualism? In: C.R. Huxley and D.F. Cutler, eds. Ant-Plant Interactions. Oxford University Press, Oxford, United Kingdom.
Tryon, A. F. 1985. Spores of myrmecophytic ferns. Proc. R. Soc. Edinb. 86B: 105-110.
Watkins, J. E., C. L. Cardelus & M. C. Mack. 2008. Ants mediate nitrogen relations of an epiphytic fern. New Phytol. 180:5-8.
Young, K. R. & B. León, 1989. Pteridophyte species diversity in the central Peruvian Amazon: importance of edaphic specialization. Brittonia 41: 388-395.
Yu, D.W., H.B. Wilson & N.E. Pierce. 2001. An empirical model of species coexistence in a spatially structured environment. Ecology 86: 1761-1771.
Presentado: 09/11/2009
Aceptado: 05/04/2010
Publicado online: 14/12/2010













