1. Introduction
Wetlands associated with tropical river habitats sustain diverse specialized flora and fauna (Chakraborty et al., 2023), acting as essential longitudinal and transversal corridors. These ecosystems facilitate biodiversity dispersion and provide critical ecological functions, including water retention, flood regulation, purification processes, and resources for fishing and forestry (Ghosh et al., 2024). Coastal wetlands encompass a variety of environments, both natural and artificial, that are characterized by being permanently or temporarily inundated by fresh, estuarine (brackish) or saline water, and include marine regions that do not exceed 6 meters in depth with respect to sea level. middle of the sea, as established in the 1971 RAMSAR Convention.
In Ecuador, the "Abras de Mantequilla" wetland has been designated as a Ramsar site since March 14, 2000, which implies that the biodiversity present is protected under the regulations and guidelines of the RAMSAR Convention, which seeks to promote the development of the territory, based on its ecological, botanical, zoological, limnological and hydrological conditions (Painii-Montero et al., 2020). Despite this designation, the Abras de Mantequilla wetland faces a series of pressures of human origin, including the extraction of water for irrigation, and consumption, deforestation, agricultural intensification and fishing exploitation (Quevedo, 2008; Alvarez-Mieles et al., 2013). These human activities are putting significant pressure on the biodiversity and overall health of the Abras de Mantequilla wetland. The ecosystem comprises permanent flooding lagoons, temporary flooding areas, and remnants of tropical dry forests. This area is lo cated within the Southwestern Tropical zoogeographic zone (Irvine et al., 2022), and the predominant type of ecosystem is the flooded riparian grassland, with small remnants of coastal lowland deciduous forest (MAE, 2022). Furthermore, agricultural land use predominates in the area, where a forested area called “Noé Morán Forest” composed of a patch or remnant of forest with little human intervention and the presence of secondary succession forest species, is considered as a sustaina ble agroecosystem of the site; this area is approxi mately 10.6 hectares.
Abras de Mantequilla wetland faces a series of multifaceted challenges, including nutrient scarcity, water stress, erosion, phosphorus fixation, soil acidity, and soil biological diversity, which make soils especially susceptible to degradation and, there-fore, negatively affect the growth and development of plants (Cardoso & Kuyper, 2006; Qiu et al., 2019). Also, this Ecuadorian wetland system offers a range of ecosystem services such as flood regulation, nutri ent retention, erosion control, and habitat provision (Portalanza et al., 2024). Gradual environmental dy namics significantly influence on the distribu tion of trees and associated organisms, such as my corrhizal fungi. Arbuscular mycorrhizal fungi (AMF), whose hyphal network improves water absorption and essential minerals by colonized roots (Usman et al., 2021; Vieira et al., 2020). These fungi establish symbiotic relationships with approximately 80% of terrestrial vascular plants (Solís-Rodríguez et al., 2020), playing a crucial role in the establishment and maintenance of plant communities in forest ecosystems (Wang et al., 2019). Its effects on host plant physiology, including nutrient uptake from the root system and enhancement of soil carbon stor age through the extraradical mycelium, provide an im portant pathway for the translocation of photosynthesis-derived carbon to microsites, which contributes to maintaining the soil structure (Rillig, 2004; Islam et al., 2022).
AMF also plays a key role in mobilizing nitrogen and phosphorus from organic polymers, releasing nutrients from mineral particles through weathering, and mediating plant responses to stress factors such as drought, soil acidification, toxic metals and plant pathogens. Furthermore, they establish a variety of interactions with other soil microorganisms, which contributes to improving soil fertility in terms of its physical, chemical and biological properties (Hartmann & Six, 2023). A series of physical, chemical, and biological indicators may be used to evaluate soil fertility. Physical indica tors include soil texture, aggregation, moisture, po rosity and bulk density, while chemical indicators in clude soil pH, total carbon and nitrogen, mineral nutrients, organic matter and soil cation exchange capacity. Biological indicators, such as microbial biomass, are also important to understand and monitor soil fertility.
The formation of symbiosis between fungi and plants in lightly disturbed environments, such as the “Noé Morán” secondary forest, is crucial due to the benefits that AMF provides to plants. These benefits include more significant absorption of essential nutrients, such as phosphorus, nitrogen, potassium and calcium, through external hyphal networks, facilitated by transporters such as H+-ATPase (Guzmán & Farías, 2005), as well as an increase in resistance against pathogenic agents, thanks to the ability of mycorrhizae to inhibit the growth of other competing microorganisms that could negatively affect plants (Ullah et al., 2024).
Currently, inadequate forest management and continuous loss of vegetation have led to adverse effects such as increased soil degradation and alteration of beneficial microflora and fauna in conventional agriculture. Faced with this problem, it is crucial to implement control measures, such as organic or ecological nutrition, to establish self-sustaining systems that take advantage of the ecosystem's natural responses, such as using green manures, humus, compost and beneficial microorganisms. AMF are particularly relevant in this context, as they contribute to mobilizing and recycling nutrients to improve soil fertility. In this study, we sought to identify which AMF species are establishing beneficial symbioses with the most representative forest species of the "Noé Morán" secondary forest of the Abras de Mantequilla Wetland. This analysis allowed us better to understand the soil-plant-microorganism interactions in this sustainable agroecosystem.
2. Methodology
Study area and sampling design
This study was carried in the “Noé Morán” forest in the Abras de Mantequilla Wetland, Vinces Canton, Los Ríos Province, Ecuador. This wetland covers 56,000 ha and is in the province's central region (1° 28' 0" S; 79° 34' 59" W). In 2000, it was designated as a Ramsar site due to its importance a nesting area for various migratory bird species and its rich native ichthyological fauna. This wetland comprises natural depressions in the terrain that are fed mainly by water from rainfall, and the water supply from rivers and streams, with altitudes ranging between 5 and 10 meters (Quevedo, 2008).
The present investigation was carried out in the rainy season of 2023. Four the most numerous and representative forest tree species of the “Noé Morán” forest were selected, triplicate soil samples were collected in the vicinity of the Guazuma ulmifolia (Guasmo), Albizia guachapele (Guachapelí), Eugenia pustulescens (Mountain guayabo) and Cecropia peltata (Guarumo) at randomly established points within the forest, giving a total of 12 samples for the entire experiment. Each sample consisted of three subsamples taken within a one-meter radius of the main collection point, totaling approximately 2 kg of soil per sample. Collection was carried out using a Dutch auger (Efco) at a depth of 0-20 cm and the tool was meticulously cleaned with 98% alcohol to avoid contamination between each sampling. Soil samples were placed in previously identified and hermetically sealed plastic bags, then stored in cold boxes (20.7 L Elec trolux) with ice. Subsequently, they were transferred to the Chemistry and Biochemistry laboratory of Universidad Técnica Estatal de Quevedo, Quevedo, Ecuador, where they were refrigerated at a temper ature of 6 to 10 °C. After this, the processes of iso lation, counting and identification of AMF spores were carried out, as well as the physicochemical analysis of the soil samples of the "Noé Morán" secondary forest.
Granulometric and chemical analysis of the soil
A portion of 300 cm3 of each soil sample was sent to the soil and water laboratory of the INIAP (National Agricultural Research Institute) Tropical Pichilingue Experimental Station, located at km 5 Vía Quevedo-El Empalme, Cantón Mocache, Province of Los Ríos, Ecuador to carry out the soil analyses. pipette method determined the soil texture (Teixeira et al., 2017). Soil pH was measured with distilled water (1:1), and P, K, Cu, Fe, Zn, and Mn were extracted in the Mehlich-1 solution. The elements Ca, Mg, and Al were extracted with a solution a (CH3 COO)2 CaH2O solution. Sulfur was extracted in a solution of SO4. The boron was extracted in hot water. Organic matter was evaluated by oxidation of soil samples with a sulfochromic solution and was determined using the Black Method (Teixeira et al., 2017).
Extraction and identification of AMF spores
AMF spores were extracted from 100 g of each soil sample by the wet sieving method (Gerdemann & Nicoloson, 1963) followed by centrifugation in a sucrose gradient (20% and 60%). Nested sieves (Fisher Scientific) with 200, 100, and 50 µm openings were used for wet sieving the 200 µm sieve was transferred to a Petri dish and inspected with a dissecting microscope to detect and collect spores and large sporocarps. The materials collected on the other sieves were transferred to Falcon tubes containing the sucrose gradient and centrifuged at 3000 rpm for 3 minutes. The supernatant from the centrifugation was decanted onto a 50-µm sieve, washed with sterile distilled water to remove excess sucrose, and transferred to Petri dishes for closer inspection of AMF spores under a dissecting microscope (Leica DM 2500). Phenotypic characteristics of the spores, such as size, color, shape and hyphal attachment, were used to distinguish the various morphotypes. Spores of each morphotype were permanently mounted on slides with polyvinyl-lactoglycerol (PVLG) and PVLG mixed with Melzer's reagent (1:1, v/v) and examined with a compound and camera mounted micros-cope (Leica DM 2500).
Spores were identified at the genus and species level by analyzing the spore wall structure, Melzer reaction, and other taxonomically informa tive features. For the AMF spore identification at the genus and species levels, comparisons were made with the original catalog of the species description, with those described in Blaszkowski (2012) and with online photos of reference crops from INVAM (http://invam.wvu.edu - West Virginia University, USA) to identify AMF spores at the genus and species level. Glomoid spores that could not be attributed to species were assigned to Glomus. The taxonomic classification followed as a ref erence is the one proposed by Redecker et al. (2013). The total abundance of AMF spores (per 100 g of soil) was obtained by counting the spores in each soil sample. The number of samples in which spores of a specific species were detected was used to calculate the frequency of occurrence (FO), ex pressed as a percentage. AMF species richness (S) was determined by counting fungal species per soil sample. The number of spores of each species was used as a measure of species abundance to calcu late the Shannon-Wiener diversity index (H), and the relationship between the observed diversity and the expected maximum diversity among forest types was determined based on the species composition of AMF using the Pielou evenness index (J).
Statistical analysis
Data on total spore abundance were transformed using the log (x + 1) function before analysis, to meet the requirements of homogeneity of the variance (Levene's test) and the normality of the residuals (Kolmogorov-Smirnov test) for the datasets. Differences in total spore abundance and species richness between different locations were examined using a one-way analysis of variance (ANOVA), followed by a Tukey test (p < 0.05). All of these statistical analyzes were carried out using the VEGAN package within the R software environment (R Core Team, 2015).
Two matrices were obtained to correlate the compositions of the AMF communities with the physical and chemical properties of the soil. The first matrix was obtained with spore abundance for AMF species in each tree species type, and these data were subjected to the Hellinger transformation before analysis. Rare species (those comprising <10% of the total frequency) were removed from the analysis as the ordination power of the data decreased (Hill & Gauch, 1980). The second soil property matrix was obtained, and collinear variables were removed based on a variance inflation factor (VIF) ≥ 10. Both matrices were subjected to discriminant redundancy analysis (RDA) followed by a forward step of selection (Blanchet et al., 2008) to deter mine the proportion of AMF community variance explained by soil properties, followed by an ANOVA (p < 0.05). All multivariate analysis were per formed using the “for” packages (Dray et al., 2016) and "vegan" (Oksanen et al., 2015) on the R software platform (R Core Team, 2015).
3. Results and discussion
Soil physicochemical properties
The set of physicochemical variables revealed relative differentiations according to the forest tree species in cultivation. The soil of the Albizia guachapele exhibited a predominance of clay, with a value of 167%, significantly surpassing the other cultivars. In this context, no alterations were observed in soil pH levels, which remained in a range between 5.46 and 6.07. Generally, certain forest species have the capacity to release organic acids through the decomposition of organic matter in the soil (Kupka & Gruba, 2022); these acids can acidify the soil and decrease its pH.
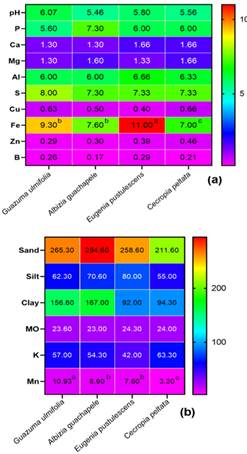
Figure 1 Physical and chemical analysis (a, b) of rhizosphere soils of four forest species from the “Butter wetlands”.
In addition, differences were identified in the iron content, with a value of 11 mg/kg in the soil with a predominance of Eugenia pustulescens, compared to the Cecropia peltata soil, which registered an average of 7 mg/kg. Several studies have shown that forest species have different nutrient requirements, including iron (Singh et al., 2021; Epihov et al., 2021). Some species can extract large amounts of iron from the soil to meet their growth needs, which can reduce the available iron content in the soil (Lambers et al., 2021).
Another important parameter that revealed outstanding values was the concentration of manganese (Mn), where the soil sample of Guazuma ulmifolia presented the highest values (Figures 1a and 1b), with an average of 10.93, in contrast to the lowest values obtained in the soil sample of Cecropia peltata tree, with an average of 3.26.
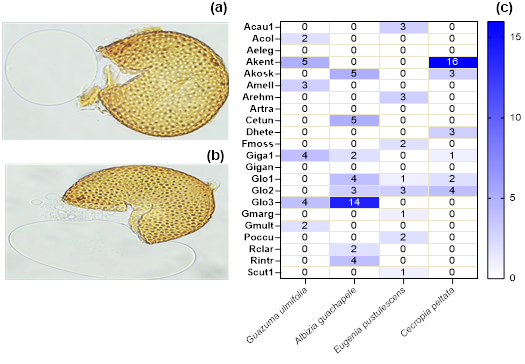
Figure 2 Predominance of mycorrhizal species and families associated with forest crops - Spores (a, b) and abundance of mycorrhizal species (c). *AMF species abbreviation: Acaulospora sp1. (Acau1); Acaulospora colombiana (Acol); Acaulospora elegants (Aeleg); Acaulospora kentinensis (Akent); Acaulospora koskei (Akosk); Acaulospora mellea (Amell); Acaulospora rehmii (Arehm); Archaespora trapei (Artra); Claroideoglomus etunicatum (Cetun); Dentiscutata heterogama (Dhete); Funneliformis mosseae (Fmoss); Gigaspora sp1. (Giga1); Gigaspora gigantea (Gigan); Glomus sp1. (Glo1); Glomus sp2. (Glo2); Glomus sp3. (Glo3); Gigaspora margarita (Gmarg); Glomus multicaule (Gmult); Paraglomus occultum (Poccu); Rhizophagus clarus (Rclar); Rhizophagus intraradices (Rintr); Scutellospora sp1. (Scut1).
AMF species and family’s abundance
It was observed that the species Albizia guachapele showed a remarkably high presence of AMF, with a total of 39 spores per gram of soil (Figure 2a-c). The Glo3 strain was the most frequent within this species, with an average of 14 spores. On the other hand, the species Eugenia pustulescens presented a lower presence of AMF compared to Albizia guachapele, recording a total of 16 spores per gram of soil. This finding reveals considerable variability in the mycorrhizal associations among the tree species studied, suggesting that certain species may form stronger or more frequent symbiotic relationships with arbuscular mycorrhizal fungi than others.
Furthermore, both the Guazuma ulmifolia and Cecropia peltata species exhibited a majority presence of AMF in the analyzed soil. Guazuma ulmifolia showed a total of 20 spores per gram of soil, while Cecropia peltata recorded 29 spores per gram. The Akent strain was the most predominant in both species, with an average of 5 spores in Guazuma ulmifolia and 16 spores in Cecropia peltata. These results underscore that different forest species can exhibit distinct mycorrhizal associations, influencing the diversity and distribution of mycorrhizal fungi within the forest ecosystem. This association is consistent with previous studies showing how host plant characteristics shape mycorrhizal communities. Luo et al. (2024) further emphasized these differences, revealing how the diversity of mycorrhiza-associated fungi and trees plays a crucial role in shaping the functioning of subtropical mountain forest ecosystems. In fact, tree mycorrhizal association types regulate the biodiversity productivity relationship in subtropical forests, highlighting the pivotal role of mycorrhizal diversity in driving ecosystem dynamics (Deng et al., 2023).
Mean species richness (S), Shannon index (H), and Pielou equitability index (J)
Simpson's index was used to evaluate the diversity of fungal species present in the soil of each sampling site (Figure 3). The results showed a Simpson index of greater than 0.79 in all studied areas, suggesting a high diversity of mycorrhizal fungal species. Studies carried out by Jerbi et al. (2020) refer to the fact that the availability of water in the soil, influenced by precipitation and environmental humidity, can significantly impact on mycorrhi zae. However, seasonal variations in climatic condi tions can affect nutrient availability (Faghihinia et al., 2020) and host plant activity, which in turn influences mycorrhizal diversity (Bennett et al., 2020).
These results demonstrated that, regarding mycorrhizal diversity, the forest species analyzed have a similar impact on the ecosystem, at least in the specific context and conditions of the study. Carteron et at. (2022) suggested that although tree species may differ, they manage to associate with similar mycorrhizal fungi. These fungi may provide similar benefits to plants, such as increased absorption of nutrients from the soil (Rożek et al., 2020; Zhang et al., 2021), which may lead to similar ecological outcomes in terms of health and growth (Singavarapu et al., 2022). Under this context, the lack of statistical significance does not necessarily imply that there are no differences in mycorrhizal diversity between species, but rather that the data collected do not provide sufficient evidence to demonstrate a significant difference.
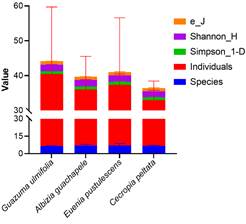
Figure 3 Simpson, Shannon diversity indices and equity index for the mycorrhizal species present in the “Butter wetland”.
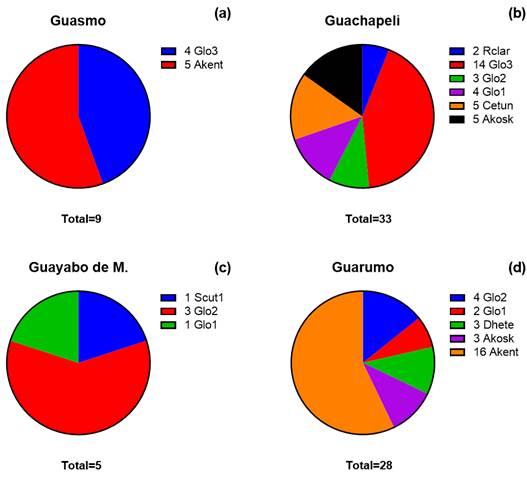
Figure 4 Species/family composition of arbuscular mycorrhizae associated with four forest species: a Guasmo, b Guachapeli, c Guayabo de monte, and d Guarumo. *AMF species abbreviation: Acaulospora kentinensis (Akent); Acaulospora koskei (Akosk); Claroideoglomus etunicatum (Cetun); Dentiscutata heterogama (Dhete); Glomus sp1. (Glo1); Glomus sp2. (Glo2); Glomus sp3. (Glo3); Rhizophagus clarus (Rclar); Scutellospora sp1. (Scut1).
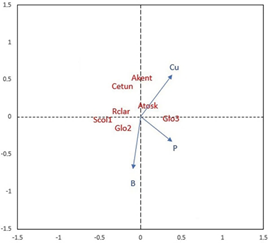
Figure 5 Redundancy Analysis (RDA) of the mycorrhizal species communities with soil parameters in the “Butter wetland”. *AMF species abbreviation: Acaulospora kentinensis (Akent); Acaulospora koskei (Akosk); Claroideoglomus etunicatum (Cetun); Glomus sp2. (Glo2); Glomus sp3. (Glo3); Rhizophagus clarus (Rclar); Scutellospora sp1. (Scut1).
Composition of species/families in coverage
The composition analysis highlighted that the Albizia guachapele ecosystem exhibits a notable diversity of mycorrhizae associated with the root system, accounting for six identified species (Figure 4a-d). This reflects a significant level of symbiotic association in this tree species. A study carried out by Waring et al. (2016) highlighted that each plant species can have a unique relationship with certain species of mycorrhizal fungi. The ability of A. guachapele to interact and associate with a variety of mycorrhizae may be due to specific characteristics of its root system, root exudates or metabolites that favor this diversity (Blair et al., 2004; Balieiro et al., 2007).
Cecropia peltata mycorrhizal species, suggesting a healthy interaction with its underground environment. Certain species may provide suitable habitat for a variety of mycorrhizae (Tedersoo et al., 2020; Huey et al., 2020). Less association of mycorrhizae in other plant species could be attributed to soil and environmental conditions that may not be optimal to sustain diversity (Janowski & Leski, 2022). Other factors such as nutrient availability, soil moisture and pH can influence the diversity and composition of the mycorrhizal community associated with C. peltata (Ma et al., 2021; Liu et al., 2021). On the other hand, the Eugenia pustulescens and Guazuma ulmifolia trees showed a moderate count of 3 and 2 mycorrhizal species, respectively, indicating lower diversity compared to A. guachapele and C. peltata.
Redundancy analysis
The redundancy analysis (RDA) performed reveals the relative contributions of various environmental variables to the variability in ecosystem composition (Figure 5). This approach allows dis cerning the specific influence of each environmental factor on plant community structure, providing a deeper understanding of the underlying ecological processes.
The redundancy analysis (RDA) is illustrated in Figure 5, highlighting the influence of specific soil parameters on the composition of arbuscular mycorrhizal fungi (AMF) communities within the ecosystem. The RDA results indicate that phosphorus (P), boron (B), and copper (Cu) have significant impacts on the variance in AMF composition among the sampled sites, as denoted by their p-values below 0.05. These nutrients play essential roles in plant-mycorrhizal interactions, with phosphorus and copper crucial for fungal metabolic processes and symbiotic efficiency (Shi et al., 2023; Bhupenchandra et al., 2024). In contrast, iron (Fe) and sulfur (S) did not reach statistical significance, suggesting, according to Zheng et al. (2024), that their concentrations may not substantially influence AMF community structure. This pattern underscores the selective role of certain soil nutrients in shaping AMF diversity and distribution, as forest species likely interact differently with AMF depending on soil nutrient availability.
4. Conclusions
This study highlights the diversity and function of arbuscular mycorrhizae (AMF) in the ecosystem of the "Abras de Mantequilla" wetland in Ecuador. Significant differences were observed in the physical-chemical properties of the soil between the tree species studied, as well as variations in the abundance and diversity of AMF species associated with each tree species. The findings highlighted the capacity of certain forest species, such as Albizia guachapele and Cecropia peltata, to favor the presence of AMF in the soil, which underlines the importance of conserving and adequately managing these natural areas to guarantee their ecological function and the sustainability of the system. This study underscores the role of certain forest species in enhancing AMF diversity in Ecuadorian wetlands. Future research should explore the application of AMF in reforestation and soil restoration, particularly in degraded areas, to support ecosystem resilience and soil health.















