Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista Peruana de Biología
versión On-line ISSN 1727-9933
Rev. peru biol. v.15 n.2 Lima 2008
TRABAJOS ORIGINALES
Long-term effects of the consumption of Stevia rebaudiana (Magnoliopsida, Asteraceae) on fertility mices
Efecto a largo plazo del consumo de Stevia rebaudiana (Magnoliopsida, Asteraceae) en la fertilidad de ratones
Juan C. Gil, Paulo Lingan, Carlota Flores* y Pedro J Chimoy
Facultad de Biología, Universidad Nacional Pedro Ruiz Gallo. Lambayeque- Perú.
* Laboratorio de Anatomía Patológica, Hospital Almanzor Aguinaga Asenjo. Chiclayo-Perú
Email Pedro Chimoy pchimoy@yahoo.com
Abstract
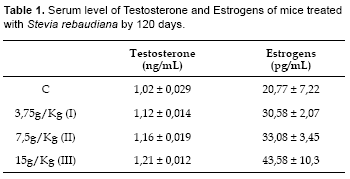
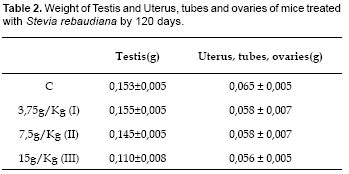
The objective of this work was to determine the long-term effects of consuming aqueous extracts of Stevia rebaudiana (Bertoni) leaves on the number of offspring, microscopic morphology of genitalia, and levels of testosterone and estrogens in albino mice. We studied four groups of mice, each consisting of 6 males and 6 females. One group was the control (C) and received food and plain water. Three groups received treatment of food and water with extract of S. rebaudiana at concentrations of (g/Kg) 3,75g (I), 7,5 (II) and 15 (III) for a period of 120 days. The offspring were counted and the histology of genitalia among the groups was compared. Serum levels of total testosterone and estrogens were measured by immunoassay and sexual behaviour was observed. Levels of total testosterone (ng/mL) were significantly different between groups (C= 1,02±0,03; I=1,12±0,01; II= 1,16±0,02; III= 1,21±0,01). Likewise were also significantly different the levels of total estrogens (pg/mL) (C= 20,77±7,22; I= 30,58±2,07; II= 33,08±3,45; III= 43,58±10,3). But no significant differences were observed between the weights (g) of the uterus, tubes and ovaries in group (C= 0,065±0,005; I= 0,058±0,007, II= 0,058±0,007; III= 0,056±0,005). The weight (g) of the testicles showed significant differences only with the treatment III (C= 0,153±0,005; I= 0,155±0,005, II= 0,145±0,005; III= 0,110±0,008). Histological differences in the genital organs between control group and the treatment groups were not detected. Libido in male mice treated with SrB was increased. The weight of the offspring (g) was higher and statistically different in groups II and III (C= 7,53±0,26; I= 7,86±0,47; II= 9,37±0,66; III= 9,35±0,45). The number of offspring was lower and significantly different for treatments II and III (C= 10,83±0,75; I= 11,83±1,17; II= 8,66±0,52; III= 9,17±0,98). We conclude that consumption of SrB does not affect fertility in mice. However, SrB consumption is associated with markedly diminished weight of the testicles accompanied by increased testosterone and estrogens levels.
Keywords: Stevia, testis, fertility, testosterone, estrogen
Resumen
Este trabajo fue realizado con el objetivo de conocer los efectos a largo plazo del consumo de extractos acuosos de hojas de Stevia rebaudiana (Bertoni) sobre el número de crías, la micromorfología de los órganos genitales, niveles de testosterona y estrógeno totales, de ratones albinos. Se Trabajó con cuatro grupos de ratones, cada grupo estuvo conformado por seis macho y seis hembras. Un grupo control (C) y tres tratamientos, el primero consumió alimento y agua mientras que los grupos tratamientos consumieron el mismo alimento pero en vez del agua un extracto de S. rebaudiana a concentraciones de (g/Kg) 3,75 (I), 7,5 (II) y 15 (III), por 120 días. Las crías fueron contadas y pesadas después de cada nacimiento, la histología de los órganos genitales entre los grupos fue comparada, se midió los niveles séricos de testosterona y estrógeno total por inmunoquimio-luminiscencia, así mismo se observó el comportamiento sexual. Los niveles de testosterona total (ng/mL) fueron significativamente diferentes entre los grupos (C= 1,02±0,03; I= 1,12±0,01; II= 1,16±0,02; III= 1,21±0,01). Así mismo los niveles de estrógeno total (pg/mL) también mostraron diferencias significativas (C= 20,77±7,22; I= 30,58±2,07; II= 33,08±3,45; III= 43,58±10,3). Sin embargo no se observaron diferencias significativas entre los pesos (g) de los úteros, trompas y ovarios (C= 0,065±0,005; I= 0,058±0,007; II= 0,058±0,007; III= 0,056±0,005). El peso (g) de testículos mostró diferencias significativas solamente con el tratamiento III (C= 0,153±0,005; I= 0,155±0,005; II= 0,145±0,005; III= 0,110±0,008). No se detectaron diferencias histológicas en los órganos genitales del grupo control y de los grupos tratamientos. La libido en los ratones machos tratados con SrB estuvo aumentada. Finalmente, el peso de las crías (g) fue mayor y estadísticamente diferente en los grupos II y III (C= 7,53±0,26; I= 7,86±0,47; II= 9,37±0,66; III= 9,35±0,45), mientras que el número de crías fue menor y significativamente diferente para los tratamientos II y III (C= 10,83±0,75; I= 11,83±1,17; II= 8,66±0,52; III= 9,17±0,98). Se concluye que el consumo a largo plazo de SrB no afectó la fertilidad de los ratones pero sí disminuyó notablemente el peso y el tamaño de los testículos. Sin embargo los niveles de testosterona y estrógenos aumentaron notablemente.
Palabras claves: Stevia, testículo, fertilidad, testosterona, estrógeno.
Introduction
Stevia rebaudiana (Bertoni) Hemsl. is known as wild plant in Paraguay and Brazil, is cultivated in Mexico, the United States, Thailand, Russia, England, Japan and Canada.(Lee et al. 1979; Donalisio et al. 1982; Goenadi, 1983; Saxena & Ming 1988; Brandle & Rosa 1992; Fors 1995). Stevia rebaudiana recently has been introduced in Peru and cultivated in the Amazon basin. The leaves of the plant have been used as an oral contraceptive and sweetener (Mazzei-Planas & Kuc, 1968), a hypoglycemic agent (Curi et al, 1986; Gregersen et al, 2004), a diuretic, natriuretic and kaliuretic (Melis 1992, 1995; Kinghorn & Soejarto, 1985), and a hypotensive agent (Soejarto et al, 1982; Kinghorn & Soejarto, 1985; Melis, 1992; Haebisch, 1992; Hsu et al, 2002).
Mazzei-Planas & Kuc (1968), Portella & Álvares (1982), and Melis (1999) found that the effects of aqueous Stevia rebaudiana extracts in rats are the fertility decrease, an effect not reversed following withdrawal. Results from other groups, however, did not confirm the antifertility effect (Schvartzman et al, 1977; Yodyingyuad & Bunyawong, 1991; Geuns, 2003). In light of these controversial findings, the use of S. rebaudiana in the treatment of human diseases requires further elucidation of its side effects. In addition, we must take into account the fact that S. rebaudiana already is commercialized in many countries, for example as a sweetener for diabetics (Stevita-GNC).
We undertook this study to determine the long term effects of consuming extracts of different concentrations of S. rebaudiana on fertility of mice and the effect on the genitalia of mice. Other authors have studied the effect of S. rebaudiana on male rats, designing the research with male rats isolated (Melis, 1999, Oliveira Filho et al, 1989; we work with pairs of mice because rodents live with a partner from weaning, and the isolation of male can affect his fertility.
Material and methods
Plant Material: Leaves of S. rebaudiana were collected in Rodriguez de Mendoza (Departamento de Amazonas, Perú), identified by Dr. Horacio de la Cruz, and deposited in the Herbarium of the Pedro Ruiz Gallo National University (Lambayeque Perú; specimen 12635). Two kilograms of leaves were dried at ≈50 ºC under low pressure to yield 0,5 kg, which were crushed in a clean, sterile mortar. The resulting powder then was used as test samples, prepared according to Mazzei-Planas & Kuc (1968) and administered orally to mice.
Animals: Animals consisted of 24 male and 24 female mice (Mus musculus var. albinus) that were 45 days old and had free access to food (Purina rat chow) and water. Mice were housed in a temperature-controlled room (25±1 ºC) and kept under a 12:12 h automatic light-dark cycle throughout the experiment (lights on from 19:00 to 07:00 h).
Experiment design: The animals were separated into four groups, each consisting of six pairs of male-female. Each pair was placed in a separate cage (26x41x18 cm). The control group (C) was given food and physiological saline solution, whereas the treatment groups were given food and varying concentrations of S. rebaudiana extract corresponding to 3,75 g/kg (I), 7,5 g/kg (II), and 15 g/kg (III) for 120 days. The number and weight of the offspring were recorded for 21 days after birth. The behaviour of the animals was observed in all experiments.
On the last day of the experiment, tissue and serum collection was performed on the anesthetized animals. Pentobarbital (30 mg/kg, Sigma Chemical) was administered intraperitoneally for anaesthesia. After decapitation blood samples were collected and then centrifuged (4000 rpm, 4ºC, 10 min) to obtain serum for hormonal analysis. The reproductive organs of the females (uterus, tubes, ovaries) and males (testes) were immediately extracted, weighed in an analytical balance, and placed in a solution of buffered formalin (10%).
All experiments were performed in accordance with the guidelines of UNPRG on animal care and the Ethics Commission.
Histology: Microtomy of the reproductive organs was performed using the method of Prophet et al. (1995). Tissue sections were stained with eosin-hematoxylin, and images were photographed and analyzed using a microscope with an attached camera.
Testosterone and Estrogens assay: Serum levels of testosterone and estrogens were determined using a double-antibody immunoassay method (Belanger et al, 1980; Immunolite kit, Immunolite Co).
Statistical Analysis: Data were analyzed using Statistics 5.0 software and Microsoft Excel 2007. The variance of the data and confidence intervals were determined using the ANOVA and Dunnet tests, respectively. Results are presented as the means±SEM, with significance defined as p<0,01.
Results
Levels of both total testosterone and total estrogens increased with increasing concentration of S. rebaudiana were statistically significant (Testosterone, ANOVA: Fc= 100,28; Ft= 2,5; Dunnet D I= 0,098; II= 0,142; III= 0,192; Dc= 0,029; p<0,01. Estrogens, ANOVA: Fc= 12,08; Ft= 2,5; Dunnet: D I= 9,815; II= 12,322; III= 22,818; Dc= 9,69; p<0,01) (Table 1).
The testis of mice treated with S. rebaudiana was a decreased in weight of the testes in male mice control (Table 2). Penis size was approximately the same in all groups (data not shown). The weights of the testes (g) in were statistically significant for the treatment III (ANOVA: Fc= 63,6; Ft= 2,5; Dunnet: D I= 0,167; II= 0,833; III= 4,333; Dc= 0,947; p<0,01). The weights of the testes in Stevia-treated males were significantly less than that of control animals at a S. rebaudiana concentration of 15 g/kg.
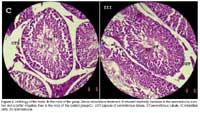
Histological analysis of the testes in the control and treatment groups revealed the following differences: testis, in the control group the Leydig cells differentiate to form spermatozoa within the seminiferous tubules. In the treatment group III (Stevia) the Leydig cells also differentiate to form spermatozoa, but a more abundant blood supply is noted (Fig. 1).
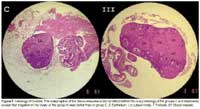
The weight of the uterus, tubes and ovaries (g) in the treatment groups did not vary significantly from that in the control group (Table 2) (ANOVA: Fc= 1,928; Ft= 2,5; Dunnet: D I= 0,007; II= 0,007; III= 0,008; Dc= 0,01; p<0,01). Histological analysis of the ovaries, in the control group there is simple cuboidal epithelium with typical features throughout its extent. In addition, the cortex is well-formed with follicles in different stages of development, and 34 luteal bodies. The medulla is well-formed and contains blood vessels and an abundant amount of fibroconnective tissue. The medulla is well-formed and shows the normal histologic structure of the mouse ovary. A precise border between the cortex and the medulla cannot be distinguished, characteristic of the normal histologic structure of the mouse ovary. In the treatment group there is a well-formed cortex with 13-15 follicles in different stages of development, and 5-7 luteal bodies (Fig. 2).
Male mice treated with S. rebaudiana demonstrated an increase of sexual desire, attempting to copulate with the female despite resistance. This behaviour was not observed in the group control.
The weights of offspring (g) were statistically significant for the treatments II and III (ANOVA: Fc= 24,397 Ft= 2,5; Dunnet: D I= 0,328; II= 1,843; III= 1,82; Dc= 0,707; p<0,01). The number of offspring were statistically significant for the treatments II y III (ANOVA: Fc= 16,333; Ft= 2,5; Dunnet: D I= 1,00; II= 2,167; III= 1,667; Dc= 1,305; p<0,01). There was a relative decrease in offspring with increasing doses of S. rebaudiana (Table 3).

Discussion and conclusions
The results shown here prove that mice that consumed S. rebaudiana reproduced without significant differences from mice in the control group, without altering fertility. The experimental design used in this study allows evaluation of the biological effect of S. rebaudiana on male-female interactions in reproduction. Isolating the male or female can affect fertility. Another important aspect to consider is the dose of S. rebaudiana that is administered, which is discussed below.
Unlike the shown for Melis (1999) y Mazzei-Planas & Kuc (1968), in rats, the testosterone levels in mice treated with S. rebaudiana grew larger, the same fact was observed in estrogenic-total levels, however the levels of estrogens were not uniform in all the repetitions as if it occurred with the testosterone. Regarding the histology of testis the abundant blood supply noted in the testis may promote greater gamete production than in the control group, which may translate to greater fecundity. However, semen should be evaluated to determine whether this increase in gamete production results in viable spermatozoa (Fig. 1)
In the ovaries also was observed a better irrigation, although the Stevia-treated tissue is not cut in the same plane as that of the control tissue, it can be seen that there is greater vascularization than in the control group, which would explain the greater number of follicles (Fig. 2). The decrease in weight of the testes was accompanied by an elevation in testosterone and estrogens levels. These results differ from those reported by Melis (1999), in which diminution of testosterone levels suggested an accompanying diminution in fertility. Taken together, the data suggest that the toxic dose for mice is three times greater than for rats, and therefore extrapolation to doses in humans is limited.
The number of offspring in the litters varied significantly between the control group and treatment groups II and III. Also, the weight of the offspring of mice in the control group differed significantly from the weight of the offspring of mice treated with S. rebaudiana (Table 3). These results are similar to those obtained by Yodyngtyuad & Bunyawong (1991) in hamsters, but partially contradict the results reported by Mazzei-Planas & Kuc (1968) and Melis (1999), who reported a decrease of testosterones levels and decrease of the fertility in rats, to the isolating the male rat before the copulation.
The histology of the reproductive organs of both male and female mice that consumed S. rebaudiana did not reveal substantial abnormalities (e.g. atrophy) in the spermatocytes, spermatogones, ovarian follicles or tubes; On the contrary a better irrIgation was noted in testes and ovaries of mice that consumed S. rebaudiana (Figs. 1, 2). This finding is in concordance with data that consumption of S. rebaudiana does not produce toxic effects in rats (Mori et al., 1981; Yamada et al., 1985; Mitsuhashi, 1976; Chan et al., 2000).
The increased sexual libido in the male mice that consumed S. rebaudiana was similar to that experienced by type 2 diabetics who took S. rebaudiana orally (3 g/L, dairy, for a month) and stated that their sexual performance recovered (data not shown). In male mice this behaviour may be related to the increase in testosterone levels. The hormonal phase of the female may also be relevant. It is known that females emit pheromones that attract the male during ovulation, and exhibit receptive behaviour. In our experiments we observed that the females were aggressive and resistant to copulation, suggesting that synthesis of pheromones may be altered; this fact suggests that consumption of S. rebaudiana affected positively the libido of males but negatively in the females.
In addition, the data shown that when the level of S. rebaudiana in the diet is 15 g/kg, the estrogens and testosterone levels are the first to be modified, next the weight of litters , the number of litters and the weight testes. Stevia rebaudiana added to the diet could be generate positive changes in the male and female gonads, and this going to produce a low number of litters but each one with better weight to the birth, and better survival.
The dose of S. rebaudiana administered (g/kg) is an important factor to discuss. Mazzei-Planas & Kuc (1968) reported that in female rats consuming 0,25 g/kg of S. rebaudiana for 12 days the fertility was diminished by 57 to 79%. Melis (1999) administered S. rebaudiana to male rats at a concentration of 10,64 g/kg for 60 days and reported a marked diminution of the cauda epididymis, seminal vesicle, and testis, as well as a decrease in testosterone levels.
But, other authors did not find any abnormality in rats treated with different doses of S. rebaudiana (0,525 g/kg, Mori et al., 1981; 4 g/kg, Silva et al., 1986). We used the same dose that is used commercially in diabetic patients (15 g/kg) and found that the main side effect was a decreased in size of the testes in male mice treated with S. rebaudiana (Table 2, Fig.1). This same effect was reported by Melis (1999) in rats treated with a concentration of 5,32 g/kg. It must be remembered that the LD50 reported for rats is 15g/kg (Akashi & Yokoyama, 1975), a concentration that did not produce death in mice (group III).
It has been reported that extracts of S. rebaudiana or its active component, stevioside, produce hypoglycaemia (Jeppesen et al., 2000, 2002, 2003; Abudula et al. 2004; Chen et al. 2005) and hypotension (Chan et al., 2000; Hsu et al. 2002; Jeppesen et al. 2003) in both rats and humans. These effects have been attributed to a putative affinity of S. rebaudiana compounds (stevioside, Steviol, Rabdovioside A) for certain hormone receptor sites (Melis, 1999). This mechanism may play a role in testosterone homeostasis, wherein stevioside competes with testosterone, causing an increase in endogenous hormone production to surpass the competitor; i.e., the organism detects that receptor signalling with the baseline level of testosterone is not adequate.
Stevia rebaudiana is used for treatment of type 2 diabetes due to its antihyperglycemic effect beyond that induced by insulin. Diabetes is a progressive disease that affects both insulin secretion and insulin action (Defronzo, 1988). Stevioside does not seem to act via the entero-insular axis, but via a direct action on beta cells (Jeppesen et al, 2000, 2002). It is not well understood how stevioside apparently uses the same mechanism of action to compete with receptors of hormones with completely different chemical structures.
An extensive study on the security of consumption of S. rebaudiana as sweetener, including the fertility and the effect of the dose is reported for Jan (2004). Our results confirm the innocuousness of consumption of S. rebaudiana to the dose of 7,5 g/Kg.
Our findings on the decrease of the weight of the testis of mice treated with S. rebaudiana to the dose of 7,5 g/Kg and the alteration of the libido in the female mice and those on carcinogenesis in mouse skin (Yasukawa et al. 2002) and inhibition of growth and reproduction of bacteria and other microorganisms suggest that S. rebaudiana extract contains substances that inhibit or regulate the cell cycle (Yabú et al., 1977; Berry & Henrry, 1981; Das et al., 1992; Tomita et al. 1997). Finally, the effect to decrease the size and weight of testis would be used for the treatment of hyperplasia of prostate.
Acknowledgments
We express our gratefulness to Dr Andrew L. Feldman (Mayo Clinic, Rochester, Minnesota) for revision of the manuscript, and Dr. César Díaz C (UNPRG) for commentaries on the histology.
Literature cited
Abudula R., P.B. Jeppesen, S.E. Rolfsen, et al. Rebaudioside A potently stimulates insulin secretion from isolated mouse islets: studies on the dose-, glucose-, and calcium-dependency. Metabolism. 2004 Oct; 53(10):1378-81.
Akashi H. & Y. Yokoyama. 1975. Dried-Leaf Extracts of Stevia. Toxicological Tests. Shokuin Kogyo. 18(20):34-43.
Belanger A., S. Caron & V. Picard. 1980. Simultaneous radioimmunoassay of progestins, androgens and estrogens in rat testis. J Steroid Biochem. Feb; 13(2):185-90.
Berry C.W. & C.A. Henry. Effect of stevioside on the growth and acid production of Streptococcus mutans. J.Dent. Res., 1981, vol 60, p 430.
Brandle J.E. & N. Rosa. 1992. Heritability for yield, leaf:stem ratio and stevioside content estimated from a landrace cultivar of Stevia rebaudiana. Can. J. Plant Sci. 72: 1263-1266.
Chan P., B. Tomlinson & Y-J. Chen, et al. 2000. A double-blind placebo-controlled study pf the effectiveness and tolerability of oral stevioside in human hypertension. Br. J. Clin. Pharmacol. 50, 215–220.
Chen T.H., S.C. Chen, P. Chan, et al. 2005. Mechanism of the hypoglycemic effect of stevioside, a glycoside of Stevia rebaudiana. Planta Med. 2005 Feb; 71(2):108-13.
Curi R., M. Alvarez, R.B. Bazotte, et al. 1986. Effect of Stevia rebaudiana on glucose tolerance in normal adult humans. Braz J Med Biol Res. 19(6):771-4.
Das S., A.K. Das, R.A. Murphy, et al. 1992. Evaluation of the cariogenic potential of the intense natural sweeteners stevioside and rebaudioside A. Caries Res. 26: 363-366.
Defronzo R.A. Lilly lecture 1987. The triumvirate: beta-cell, muscle, liver. A collusion responsible for NIDDM. Diabetes. 1988 Jun; 37(6):667-87.
Donalisio M.G.R., F.R. Duarte, A. Pinto & C.J. Souza. 1982. Stevia rebaudiana. Agronómico 34: 65-68.
Fors A. 1995. A new character in the sweetener scenario. Sugar J. 58: 30.
Geuns J.M. Stevioside. Phytochemistry. 2003 Nov; 64(5):913-21.
Goenadi D.H. 1983. Water tension and fertilization of Stevia rebaudiana Bertoni on Oxic Tropudalf . Menara Perkebunan. 51: 85-90.
Gregersen S., P.B. Jeppesen, J.J. Holst & K. Hermansen.2004. Antihyperglycemic effects of stevioside in type 2 diabetic subjects. Metabolism. Jan; 53(1):73-6.
Haebisch E.M.A.B. 1992. Pharmacological trial of a concentrated crude extract of Stevia rebaudiana (Bert.) Bertoni in healthy volunteers. Arquivos de Biologia e Tecnologia Curitiba 35(2): 299-314.
Hsu Y.H., J.C. Liu & P.F. Kao, et al. 2002. Antihypertensive effect of stevioside in different strains of hypertensive rats. Zhonghua Yi Xue Za Zhi (Taipei). Jan; 65(1):1-6.
Jan M.C. Geuns. 2004. REVIEW: The safety of stevioside used as a sweetener. Proceedings of the first symposium The Safety of Stevioside. Chapter 9, pages 85-127, kuleuven.
Jeppesen P.B., S. Gregersen, K.K. Alstrup & K. Hermansen. 2002. Stevioside induces antihyperglycaemic, insulinotropic and glucagonostatic effects in vivo: studies in the diabetic Goto-Kakizaki (GK) rats. Phytomedicine. 2002 Jan; 9(1):9-14.
Jeppesen P.B., S. Gregersen, C.R. Poulsen &, K. Hermansen. 2000. Stevioside acts directly on pancreatic beta cells to secrete insulin: actions independent of cyclic adenosine monophosphate and adenosine triphosphate-sensitive K+-channel activity. Metabolism. Feb; 49(2):208-14.
Jeppesen P.B., S. Gregersen & S.E. Rolfsen, et al. 2003. Antihyperglycemic and blood pressure-reducing effects of stevioside in the diabetic Goto-Kakizaki rat. Metabolism. 2003 Mar;52(3):372-8.
Kinghorn A.D. & D.D. Soejarto. 1985. Current status of stevioside as a sweetening agent for human use. In Wagner, H., Hikino, H., and Farnsworth, N.R. (eds.). Economic and medical plant research. Academic Press. London.
Lee J.I., K.K. Kang & E.U. Lee. 1979. Studies on new sweetening resource plant stevia (Stevia rebaudiana Bert.) in Korea. I. Effects of transplanting date shifting by cutting and seeding dates on agronomic characteristics and dry leaf yields of stevia . Res. Rep. ORD 21: 171-179.
Mazzei-Planas G. & J. Kuc. Contraceptive properties of Stevia rebaudiana. Science 162:1007, 1968.
Melis M.S. 1999. Effects of chronic administration of Stevia rebaudiana on fertility in rats. J Ethnopharmacol. 1999 Nov 1; 67(2):157-61.
Melis M.S. 1995. Chronic administration of aqueous extract of Stevia rebaudiana in rats: renal effects. J Ethnopharmacol. 1995 Jul 28; 47(3):129-34.
Melis M.S. 1992. Stevioside effect on renal function of normal and hypertensive rats. J Ethnopharmacol. 1992 Jun;36(3):213-7.
Mitsuhashi H. 1976. Safety of Stevioside. In Tama Biochemical Co. Ltd. Report on Safety of Stevia pp. 9-10.
Mori N., M. Sakanoue, M. Takeuchi, et al. 1981. Effect of stevioside on fertility in rats. J. Food Hyg. Soc. Jpn., 22, 409-414.
Portella N.B. & N. Álvares. 1982. Influência do chá da caá-heê, Stevia rebaudiana (Bert.) Bertoni, sobre o peso dos órgãos sexuais isolados de camundogos jovens. Acta Amazónica 18 (1-2), 181-185.
Prophet E.B., B. Mills, J.B. Arrington & L.H. Sobin. 1995. Métodos Histotecnológicos. IPFA, USA 280 pp.
Saxena N.C. & L.S. Ming. 1988. Preliminary harvesting characteristics of stevia. Phys. Prop. Agric. Mat. Prod. 3: 299-303.
Oliveira-Filho R.M., O.A. Uehara, C.A.S.A. Minett & L.B.S. Valle. 1989. Chronic Administration of Aqueous Extract of Stevia rebaudiana (Bert.) Bertoni in Rats: Endocrine Effects. Gen. Pharmac 20(2), 187-191.
Schvartzman J.B., D.B. Krimer & R. Moreno Azorero. 1977. Cytological effects of some medicinal plants used in the control of fertility. Experientia. 1977 May 15; 33(5):663-5.
Silva A.R., C.M. Sladanha, R. Boelter, A.M. Chagas. 1986. Fertilidade de Ratos: Extrato Aquoso de Stevia rebaudiana (Bert.) Bertoni e Steviosideo. III Seminario Brasileiro sobre Stevia rebaudiana (Bert.) Bertoni. 34-1-2.
Soejarto D.D., A.D. Kinghorn & N.R. Farnsworth. 1982. Potential sweetening agents of plant origin. III. Organoleptic evaluation of Stevia leaf herbarium samples for sweetness. J Nat Prod. Sep-Oct; 45(5):590-99.
Tomita T., N. Sato, T. Arai, et al. 1997. Bactericidal activity of a fermented hot-water extract from Stevia rebaudiana Bertoni towards enterohemorrhagic Escherichia coli O157:H7 and other food-borne pathogenic bacteria. Microbiol Immunol. 1997; 41(12):1005-9.
Yabu M., M. Takase, K. Toda, K. Tanimoto & A. Yasutake. 1977. Studies on Stevioside, natural sweetener. Effect on the growth of some oral microorganisms. Hiroshima Daigaku Shigaku Zasshi.; 9(1):12-7.
Yamada A., S. Ohgaki, T. Noda & M. Shimizu. 1985. Chronic toxicity study of dietary Stevia extracts in F344 rats. J Food Hyg Soc Japan; 26:169-183.
Yasukawa K., S. Kitanaka & S. Seo. 2002. Inhibitory effect of stevioside on tumor promotion by 12-O-tetradecanoylphorbol- 13-acetate in two-stage carcinogenesis in mouse skin. Biol Pharm Bull. Nov; 25(11):1488-90.
Yodyingyuad V.& S. Bunyawong.1991. Effect of stevioside on growth and reproduction. Hum Reprod. Jan; 6(1):158-65.
____________________
Presentado: 04/10/2007
Aceptado: 18/03/2008