INTRODUCTION
One of the most severe neurological manifestations of acute respiratory syndrome coronavirus 2 (SARS- COV-2) is cerebrovascular disease (CVD). Reports suggest an association between COVID-19 and CVD, especially ischemic strokes, and less frequently cerebral venous thrombosis and hemorrhagic strokes. This can be explained by the association of coronavirus with a transient hypercoagulable state and disruption of endothelial function, especially in severe cases 1,2,3.
During the pandemic, different studies have shown that not all patients develop a severe form of the disease and stroke during infection with COVID-19. It has been reported an incidence of acute ischemic stroke in COVID-19 patients that ranges from 0.9% to 2.7%, with a moderate severity, high prevalence of large vessel occlusion (40.9%) and a high mortality rate (38%) 4,5. Similarly, healthcare racial/ethnic inequities have been unveiled during the pandemic 6. Elevated D-dimer, fibrinogen and the presence of antiphospholipid antibodies are prominent in COVID-19 patients with concomitant stroke. In Latin America, a study done in Bolivia found that age (>60y) and hypertension were independent risk factors for mortality 6. Finally, a systematic review found that patients with COVID-19 who experienced stroke, were more likely to be older, have pre-existing cardiovascular comorbidities, and a severe infection 7.
The determination of these risk factors in Latin American countries is important because it will allow prioritizing patients and deriving the necessary resources, since equipment and resources are scarce. For instance, even though the Peruvian government set up rules to contain the infection, like mandatory quarantine, Peru had one of the highest mortality rates in the world 8. It is necessary to have information on the current situation in Latin America on how the COVID-19 pandemic has influenced hospital care for stroke.
This study aims to assess the frequency and mortality of stroke on patients hospitalized for COVID-19 in two major referral centers in Peru. Additionally, to assess the associated factors to mortality in these patients. Finally, to compare the frequency of stroke admissions and reperfusion treatments during pandemic time with the same period in 2019.
MATERIAL AND METHODS
Study Design and Participants
An observational, retrospective cohort study was conducted in Guillermo Almenara Irigoyen and Edgardo Rebagliati Martins National Hospitals. These two medical centers are considered as national referral centers for patients with COVID-19, especially for the most severe COVID-19 cases.
All consecutive adult patients (18 years of age or older) who were admitted with stroke and COVID-19 between April and August 2020 were eligible for inclusion. We excluded patients who had the infection after a stroke.
The patients were considered COVID-19 cases following the recommendations of the Ministry of Public Health. (Ministerial resolution N 905-2020-MINSA Sanitary directive 122) Requiring a positive antigenic test or diagnostic serological test (ELISA, immunofluorescence, chemiluminescence) reactive to IgM or IgM/IgG to SARS-CoV-2 infection or a positive polymerase chain reaction test with nasopharyngeal swab (RT-PCR) to be considered positive cases of COVID-19. The standard test to confirm cases of COVID-19 was the molecular test, in situations where this test is not available, the SARS-CoV2 antigen detection test was considered a confirmatory test.
Data Collection
The information was collected from the electronic health care records including all the COVID-19 patients with stroke. A database was elaborated using Excel, and data entry was performed by neurologists and neurology residents trained in responsible conduct for research from both hospitals. We collected the following information: demographics, clinical, radiological findings and laboratory data (hemogram, coagulation, and inflammatory markers), and reason for admission. The type of stroke, affected cerebrovascular territory, severity of the stroke (estimated by the National Institute of Health Stroke Scale, NIHSS), and the reperfusion modality were recorded. We reported the reperfusion treatment, in-hospital complications, mortality, and length of hospital stay.
We use the NIHSS to assess stroke severity which includes 15 elements, and ranges from 0 to 42. We categorized as: minor stroke with a scale of 1-5; moderate stroke, 6 -14 and severe stroke > 15.
In order to estimate the number of stroke patients during 2019 and 2020, we included patients with ICD-10 codes 10 U07.1 and G45.8, G45,9, I63.0, I63.2, I63.3, I63.4, I63.5, I63.6, I63.9, I64, I67.9.
The mortality rate was corroborated with the national information system of deaths of Perú.
The outcome was the functional status estimated by the modified Rankin scale (mRS) at discharge or the last evaluation for patients still admitted. This is a scale that measures the degree of disability in patients who have had a stroke. It ranges from 0 (No symptoms at all) to 6 (dead), and we considered scores greater than 2 as functional dependency.
Statistical analysis
Absolute and relative frequencies were used to describe categorical variables, while central tendency and dispersion measures were used for quantitative variables. To evaluate associated factors to mortality and dependency (mRS> 2) we fitted Poisson regression models with robust variance and adjusted for potential confounders (age and sex). The analyses were carried out using a level of significance of 5%. The mortality rate was calculated by estimating the frequency of deaths in COVID patients during the period of time of our study.
The statistical analysis was performed using Stata v14 (StataCorp LP, 2015. College Station, Texas, USA).
Ethical aspects
The study was approved by the Research Ethics Committee for COVID-19 of the Health Social Security - Essalud. Informed consent was not obtained because the information was collected from electronic clinical records and patients were not intervened. The principle of confidentiality was guaranteed in the whole process of the study, keeping the identity of the patients anonymous.
RESULTS
There were 101 patients with COVID-19 and stroke among the 7342 patients admitted with COVID-19 in the study sites (stroke incidence:1.4%). The median age was 75 years, ranging from 25 to 96 years, and 56.4% were males (Table 1). About reperfusion treatments, only 6 patients received intravenous thrombolysis and none thrombectomy.
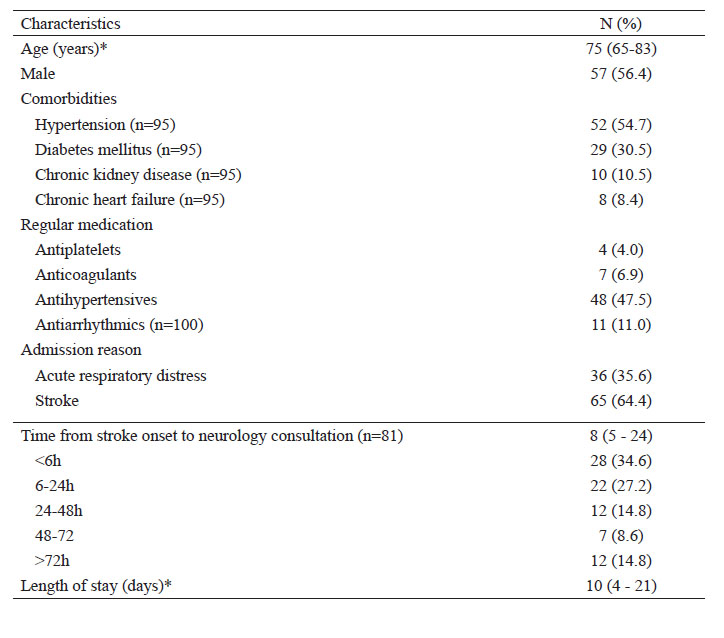
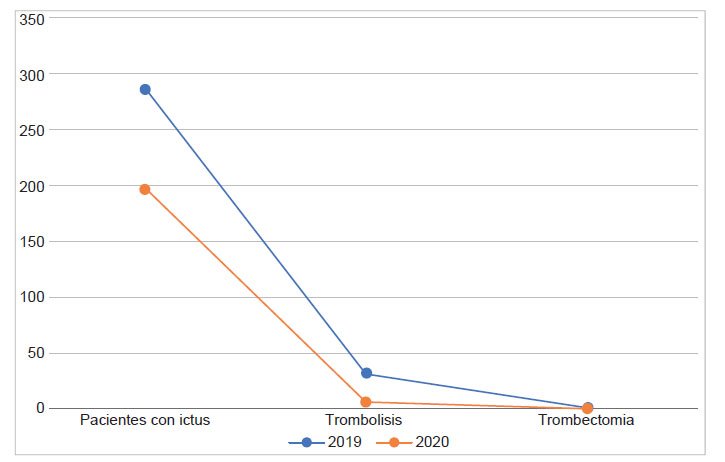
Before pandemic, between April and August 2019, there were 286 stroke admissions in both hospitals. Compared both periods of time, there was a 35% decrease in admissions of stroke before and after pandemic.
Regarding intravenous reperfusion treatment with rtPA, in 2019 there were 32 patients and in 2020 in the same period of time there were 6 with a reduction of 81% and no cases of thrombectomy were reported in 2020 and 1 the previous year (Figure 1), 93% of the patients receive anti aggregating treatment and 5% anticoagulation.
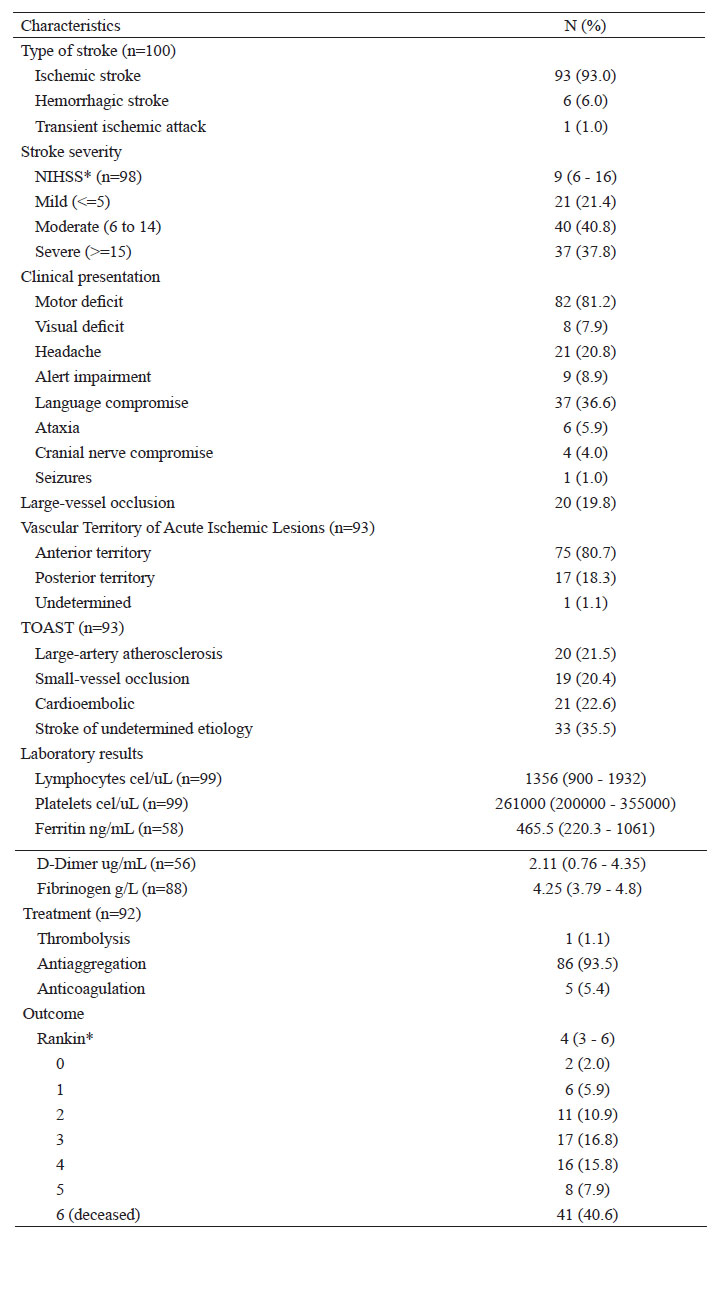
The majority of patients (64.4%) with stroke and COVID-19 were hospitalized only for stroke without respiratory symptoms and 35.6% were admitted for acute respiratory distress. 13.9% of the patients that were admitted with respiratory symptoms developed stroke during hospitalization. Most of the patients had a confirmed diagnosis of COVID19 (84.7%) (Table 2).
Ischemic stroke was the most frequent (93%), followed by hemorrhagic stroke (6%) and transient ischemic attack (1%). The median number of hours passed between the onset of stroke symptoms and the first medical assessment of stroke was 8 hours (IQR: 5 to 24) and 13.9% of the patients developed stroke during hospitalization. Moderate to severe stroke (NIHSS >= 6) was found in 78.6% of the participants and mild strokes (NIHSS <5) was found in 21.4%.
According to the TOAST classification (acute ischemic stroke), 35.5% were of undetermined etiology, 22.6% were cardioembolic, 22.5% were large artery atherosclerosis, and small-vessel disease accounted for 20.4%. The most frequently affected vascular territory was the middle cerebral artery (79.6%), followed by vertebral and basilar territories (6.5% each).
The SMASH-U etiological classification was used for hemorrhagic stroke: 1 patient had a structural vascular lesion (rupture of an aneurysm of the posterior cerebral artery territory), 2 due to hypertensive cause and the other 4 had an indeterminate etiology. All of the patients with hemorrhagic stroke died.
During hospitalization, one-third of patients developed at least one complication (31/84). The most common complications were infectious (14/84), metabolic (12/84), and neurological (10/84). Ten patients presented neurological complications including: hemorrhagic transformation (4/10), cerebral edema, intracranial hypertension and increased cerebral infarction.
We found an overall mortality rate of 40.6% and a dependency rate at discharge of 68.3%. The mortality in the ischemic stroke group of patients was 36.6%.
When we explored the association between factors, we found that an increase in 10 years of age was associated with a 29% increase in the risk of mortality when adjusting by sex. In addition, after adjusting for age and sex, having hypertension and chronic kidney disease was associated with a 1.79 and 1.82-fold increased risk of mortality compared to those who did not have these conditions, respectively. The severity of stroke measured using NIHSS was associated with a 1.70-fold increased risk of mortality compared to those with non-severe stroke. Inflammatory markers such as D-dimer and Ferritin were also associated with higher mortality (Table 3).
Table 3 Associated factors to mortality in patients with stroke and COVID-19 (n=101)
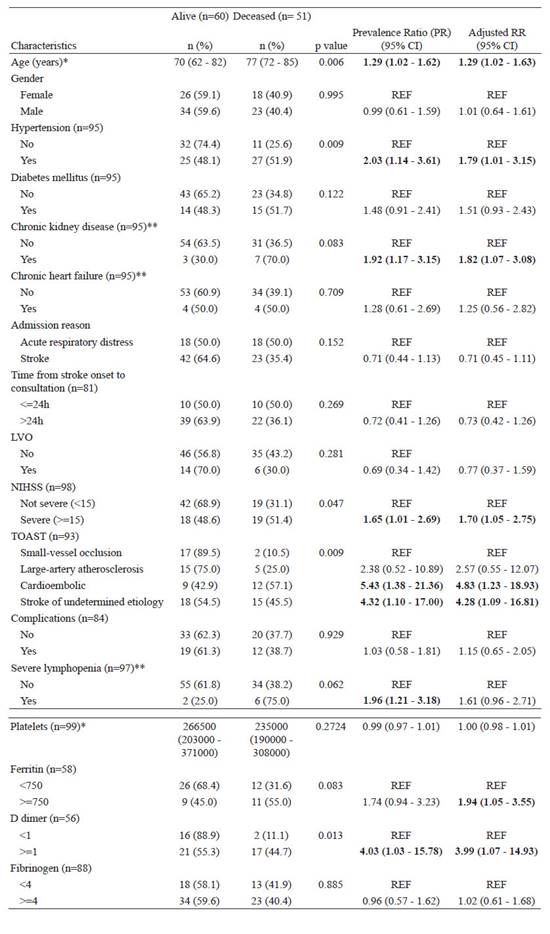
*Ranksum test
**Fisher Exact test
Models adjusted by age and sex. Age was scaled to 10 for the regression models.
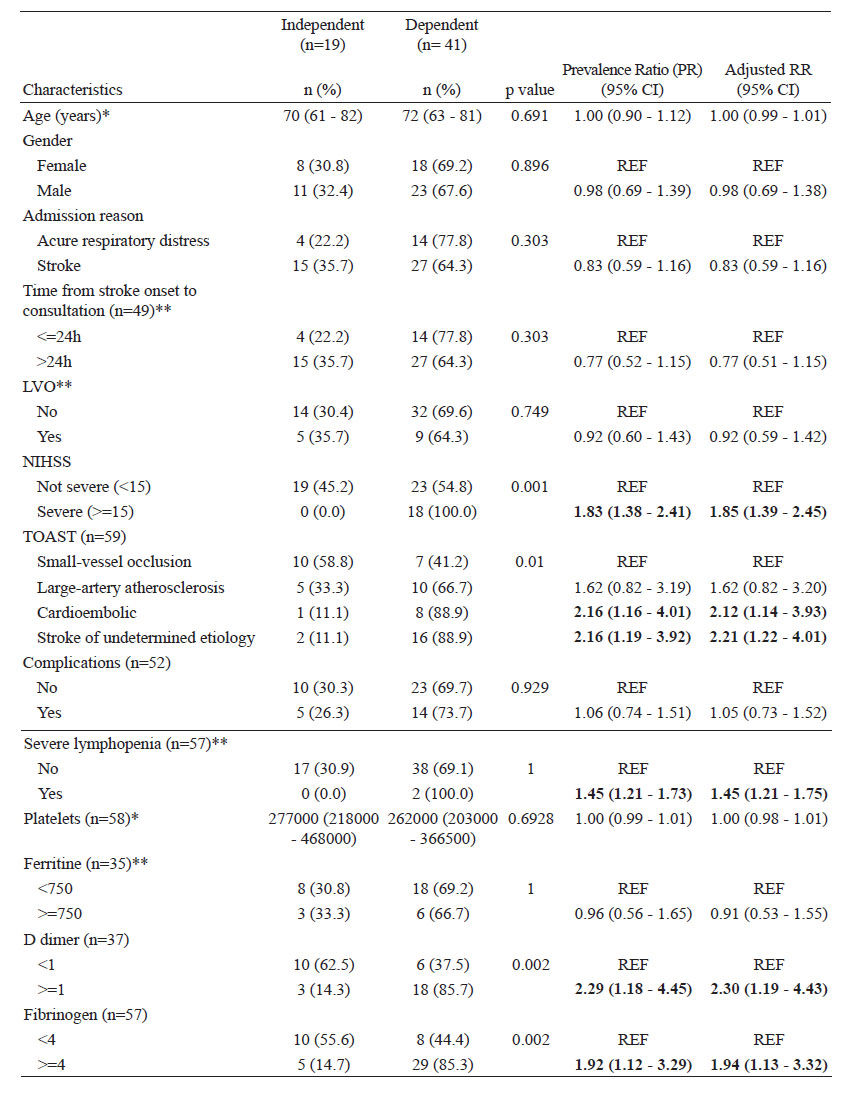
With regards to dependency, the severity of stroke was associated with a 1.85-fold increased risk of dependency compared to those with non-severe stroke when adjusting for age and sex. Also, lymphopenia and inflammatory markers (D-dimer and fibrinogen) were associated with greater risk of dependency (Table 4).
DISCUSSION
In our two-center observational study we described the characteristics of stroke patients with COVID-19. We observed a reduction of 31% in stroke patients admitted by emergency during the pandemic time (April - August) as compared to the previous year. Similarly, in Catalonia, Chicago, and Italy the number of code stroke activations decreased by 18%, 20% and 50% respectively during the pandemic 9. Also, Latin American countries like Brazil and Chile reported a decrease in the admission of all acute strokes. This can be explained by patients’ reluctance to seek medical care during the pandemic due to the adaptation of social distancing practices, the fear of contagion of the population and subsequent avoidance of the overcrowded emergency departments. Additionally, due to the prioritization of the management of patients with COVID-19 over other conditions with the subsequent reallocation of resources (e.g., specialists such as neurologist providing care to patients with COVID-19 or reallocation of beds) and the reduced capacities of emergency services due to the burden of COVID-19 patients 10,11,12.
In the period of our study, 1.4% of the patients with COVID-19 presented a stroke. There have been reported an incidence of stroke in COVD-19 patients from 0.19% to 5.4% and by Klok et al. of 1.6% and 2.5% by Lodigiani et al., 13,14,15. Our study reported most of the cases were ischemic strokes.
These results are similar to the previously reported literature. Ghannam et al. that studied neurological manifestations in COVID 19 patients, reported 87.5% ischemic stroke, 5% cerebral venous thrombosis, 5% intraparenchymal hemorrhage, and 2.5% subarachnoid hemorrhage 16. It is hypothesized that patients with COVID-19 are at increased risk of cerebrovascular events, due to a systematic response by SARS- COV2 that lead to arterial and venous thrombotic complications, hypercoagulability, endothelial injury and venous stasis 17.
Only 32% of the patients arrived at the emergency room within 6 hours of having symptoms, 25% 6 to 24 hours and 36% after 24 hours to 92 hours, losing the possibility of intravenous or endovascular reperfusion treatment in the early window without the need of perfusion imaging. This might reflect a well-known old problem. In Peru, we lack an adequate pre-hospital system for stroke and there is poor recognition of stroke symptoms. Moreover, in Latin America it has been reported that only 3-15% of the population and 50% of health workers recognize at least one warning symptom of stroke. We have no data of the onset time of symptoms to CT scan and other times but it is likely that it has increased, because of the restriction of the transportation during the quarantine and COVID-19 screening procedures (18). During the pandemic, triage system underwent modifications, prioritizing if the patient had respiratory symptoms. The patients usually underwent brain and lung tomography in addition to taking a sample for COVID antigen or PCR. Every patient was treated as COVID positive until the results were available which prolonged treatment times.
A large-vessel occlusion was found in 19.8% of cases, these results are lower than other studies carried out worldwide, in some of them this percentage is even doubled 19,20. However, we were able to perform complete vascular imaging studies in less than 40% of our patients. Also, based on the TOAST classification the predominant subtype was cryptogenic stroke in 33%, this predominance coincides with the studies carried out worldwide, some of them give even higher percentage like the 60% of their total cases 21.
Although the efficacy of mechanical thrombectomy has been demonstrated in South American countries 22, the only available treatment in public hospitals in Peru is intravenous thrombolysis. Despite this, we evidenced a significant decrease in the number of intravenous thrombolysis performed compared to 2019. The reduction of this reperfusion treatment was 81% (32 vs. 6), these percentages are much higher compared to similar studies carried out in other parts of the world, like in France, USA (Ohio, Kentucky, and Indiana) and Lyon where they observed 33.3%, 31% and 25% respectively fewer acute revascularization treatments during the month of March 2020 vs. their similar one in 2019, primarily related to reduced thrombolysis rates (23,24,25). Before the pandemic in Peru there were studies that reported 1-2% thrombolysis rates showing a problem that with the pandemic only got worse.
It has been described that COVID-19 patients have a greater stroke severity and poorer outcomes compared to historical series prior to the pandemic, 40.8% of patients had moderate impairment (NIHSS 10-15) and 37.8% severe compromise (NIHSS >15). 5 A preliminary report of a study carried out in a reference hospital in Peru showed an association between the presence of stroke and mortality in patients admitted for COVID-19 (RR = 1.49). They concluded that the presence of stroke is a poor prognostic factor in patients with COVID-19 (26). Previous studies conducted in Peru before the pandemic, reported a rate of 42-68% of patients with independence (mRS0- 1) after thrombolysis. In our study only 18.8% of the patients were independent (mRS 0-2). There are many reasons that can explain the poor functional outcomes in our patients; one of them is the decrease rates of thrombolysis and the co-infection with COVID-19. The increased stroke severity at admission in COVID-19 associated stroke patients compared with the non- COVID-19 cohort may explain the worse outcomes. In the global COVID-19 registry, they found 51% of the patients had severe disability at discharge and patients with COVID-19 had higher risk of severe disability and death compared with patients with no COVID19 27,28,29.
The overall mortality found in our study was high (40.6%) and consistent with some international cohorts such as those carried out in Wuhan and Philadelphia where they report a mortality rate of 54.5% and 39.29% respectively 21. In the subgroup of patients with ischemic stroke, we reported a mortality of 36.6% similar to that reported in The Global COVID-19 Stroke Registry in which information was collected from 28 sites in 16 countries reporting a mortality of 27.6% 29.
Regarding risk factors, we found that age, comorbidities (arterial hypertension and chronic kidney disease) and stroke severity are factors associated with mortality in patients with stroke and COVID-19, as proposed in a study as a phenotype clinical associated with high in-hospital mortality (30). In addition, an association was found between mortality and pro-inflammatory markers (D-dimer and ferritin), which are early markers of severity in COVID-19 31. The authors propose the need for reorganization strategies of the care system and thus be able to direct a differentiated management in these patients with risk factors for mortality.
There are widely studied pro-inflammatory and hypercoagulability biomarkers that determine a worse prognosis in general in patients with COVID-19 infection 32. In the present study, an association of severe lymphopenia (<500 lymphocytes) and D-dimer > 1 gr / dl (Table 5) was found with greater disability at medical discharge in patients with stroke and COVID-19, these same markers have also been related to a higher mortality rate (Table 4). It has been proposed that the inflammatory process and exaggerated immune response can precipitate destabilization of atherosclerotic plaques causing acute thromboembolic events in people with chronic atherosclerosis, and also a high level of proinflammatory cytokines, such as IL-6 whose action is widely recognized in the called “cytokine storm”, are associated with greater infarction and poor clinical outcome. All these factors could be associated with more serious cerebrovascular events and therefore cause a greater degree of dependence as evidenced in 37% of our patients in whom a NIHSS score greater than 15 was found on the emergency room 33.
This study is the first to describe the COVID-19 and stroke patients characteristics and outcomes in two of the most important referral centers in Peru. With the largest hospital capacity in the country, most of the patients had all the laboratory tests and the outcome variables.
However, our findings should be interpreted with caution due to the limitations of our study. First of all, due to the inherent retrospective study design, there is a potential for selection bias. In addition, we only included the social security health system, so the results cannot be extrapolated to the general population.
CONCLUSIONS
This is the largest cohort of stroke and COVID-19 patients reported in Peru. The stroke admissions decrease during the pandemic when compared to 2019. However, greater stroke onset time and ED consultation, stroke severity, and mortality were encountered.
This shows the necessity to educate the population in the early recognition of stroke and the reorganization of acute stroke care in order to carry out a timely treatment to improve the prognosis in these patients.