Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de Gastroenterología del Perú
versión impresa ISSN 1022-5129
Rev. gastroenterol. Perú v.24 n.2 Lima abr./jun. 2004
Update on the Pathogenesis and Immunotherapy of Esophageal Squamous Cell Carcinoma
Jorge Cervantes, M.D., Ph.D.(1,2)
1 Scientia Pro Hominem (Non-Governmental Organization), Lima, Peru. and
2 Department of Microbiology and Immunology, Hamamatsu University School of Medicine, Hamamatsu, Japan.
RESUMEN
El carcinoma de células escamosas de esófago (CCEE) es el subitpo histólogico predominante de cáncer esofágico, y se caracteriza por su alta mortalidad y diferencias geográficas en cuanto a su incidencia. A pesar de que se ha dedicado mucha investigación en esta área, aún no se conoce la causa exacta de esta neoplasia. Nuestro entendimiento de la patogénesis, epidemiología y comportamiento del CCEE continúa en desarrollo con los avances en el campo de la biología molecular. Algunos de estos avances incluyen la investigación en la etiopatogénesis (virus –como el papilomavirus humano-, y genes susceptibles a cáncer), genes relacionados a tumores (oncogenes, genes supresores de tumores), así como nuevas formas de inmunoterapia neoadyuvante para el tratamiento de esta neoplasia.
PALABRAS CLAVE: Carcinoma esofágico, células escamosas, HIV, oncogenes, inmunoterapia.
SUMMARY
The esophageal squamous cell carcinoma (ESCC) is the prevailing histology subtype of esophageal cancer and is distinguished by its high mortality and its geographic differences in regards to its incidence. The exact cause of this neoplasia is still unknown in spite of all the research made in this area. Our understanding about pathogenesis, epidemiology and behaviour of the ESCC is still in progress thanks to the advances on the field of molecular biology. Some of these advances include the research of etiopathogenesis (virus, as the human papillomavirus, and the genes susceptible to cancer), genes associated with tumors (oncogenes, tumor suppressor genes), as well as new forms of neoadjuvant immunotherapy for the treatment of this neoplasia.
KEY WORDS: Esophagus carcinoma, squamous cells, HIV, oncogenes, immunotherapy.
INTRODUCTION
Cancer of the esophagus exists in 2 main forms with different etiological and pathological characteristics, squamous cell carcinoma (SCC) and adenocarcinoma (ADC) (1). Regardless of the cell type (squamous or adenocarcinoma), esophageal carcinoma is an uncommon but agressive malignancy that usually presents in a locally advanced stage (2). Esophageal squamous cell carcinoma (ESCC) is the predominant histological subtype of esophageal cancer and is characterized by poor prognosis and a wide incidence variation in different geographical regions (3, 4).
I. PATHOGENESIS
Human papillomavirus
A possible role for Human papillomavirus (HPVs) in ESCC has been suspected since the infection of this DNA virus in the epithelium plays a crucial role in the development of cervical SCC (5, 6). However, the role of HPV in the pathogenesis of esophageal SCC is still conflicting, with regional susceptibility differences among populations around the world (7). Studies mainly from China, were the initial association was found (8), report HPV to be present in relatively high percentages of ESCC cases (9-15). A different picture seems to occur in several European countries, were studies failed to detect HPV in ESCC (16-22). A rare involvement of HPV in ESCC has been reported in Belgium (23) and Brazil (24). In Japan HPV may be absent (25) or infrequent, and what is more surprising HPV has been found even in DNA from non-cancerous esophageal mucosa (26). I recently reported absence of HPV in a group of samples from Papua Guinea (27) and a a few cases from Peru, assessed by three different high sensitive PCR based detection systems (28, 29, 30).
Tumor suppressor genes (p53, pRB)
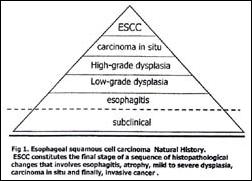
Genetic changes associated with the development of ESCC include mutation of the p53 gene, disruption of cell-cycle control in G1 by several mechanisms, including alterations in the retinoblastoma protein (RB), activation of oncogenes, and inactivation of several tumor suppressor genes. HPV early genes, E6 and E7, are important in cancer development functioning as transforming genes. E6 protein binds to and promotes degradation of the tumor suppressor protein, p53, while E7 protein complexes and inactivates the RB protein; together, they disrupt cell cycle regulation (31, 32). Genetic changes associated with the development of ESCC include mutation of the p53 gene and disruption of cell-cycle control by several mechanisms (including alterations of RB). ESCC constitutes the final stage of a sequence of histopathological changes that involves esophagitis, atrophy, mild to severe dysplasia, carcinoma in situ and finally, invasive cancer (Fig 1). Focal accumulation of p53 protein mutations in esophagitis areas at the margins of tumors have been observed (33). Mutations of the p53 gene are also involved in the pathogenesis of adenocarcinomas in Barretts esophagus. Assessment of p53 mutations status may be clinically important as a parameter for the definition of risk groups after potentially curative resections (34).
Overexpression and p53 mutations occurs frequently in both HPV negative and HPV positive ESCC lesions (35). Overexpression of p53 and loss of pRB is considered abnormal (36). RB expression can be even found in high frecuencies in ESCC (higher than p53)(37-39). Immunohistochemically determined loss of RB protein expression may indicate loss of heterozygosity of the RB gene (40).
Amplification of several other oncogenes have been detected in esophageal squamous cell carcinomas (41). The alterations observed in tumor suppressor genes or oncogenes in the esophagus can be, in any case, due to exposure to other carcinogens, such as aflatoxin B1, benzopyrene produced by fungi and bacteria, and nitrosamines caused by cigarette smoking (42, 43). Chronic mucosal irritation due to hot beverage drinking has been also considered as an etiological factor (44, 45). Smoking, alcohol consumption, and low fruit and vegetable consumption have been recently reported as risk factors for ESCC in a multicenter population-based case-control study (46), pointing out the impact of lifestyle in the pathogenesis of this neoplasia.
II. IMMUNOTHERAPY
Immune response against tumors
Local infiltration of T-cells, B-cells and macrophages has been found to be a useful prognostic factor for 5-year survival in esophageal SCC cases without preoperative radiotherapy, chemotherapy or immunotherapy, indicating that this local immunocyte infiltration, in and around the cancer stroma, is a manifestation of the host defense against cancer (47). In fact immunosuppression, which is associated with a variety of tumors, most commonly lymphoma, may also lead to the development of squamous cell carcinoma of the esophagus (48).
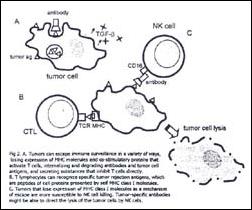
T cells are critical mediators of tumor immunity. T lymphocytes can recognize specific antigens on human tumors (not displayed on the surface of normal cells), these tumor rejection antigens are peptides of tumor-cell proteins presented to T cells by MHC class I molecules. The anti-tumor response is unable to spontaneously eliminate an established tumor, either because the tumor-specific antigens are not immunogenic enough, or because tumor cells can escape recognition and killing by cytotoxic T cells (Fig. 2). The aim of tumor immunotherapy is to enhance and augment such T cell response.
Use of activated lymphocytes stimulated with tumor-pulsed dendritic cells (to enhance the cytotoxity of the activated lymphocytes) showed disappearance of skin metastatsis of ESCC and might be useful for local treatment or postoperative adjuvant therapy (49).
Interleukin-2 (IL-2) is a T-cell growth factor, it mediates the in vivo expansion of T-cells with specific immunological functions as well as expand non-Major Histocompatability Complex (MHC) restricted lymphokine activated killer (LAK) cells. Administration of IL-2 could mediate the regression of established human cancer like metastatic melanomas, metastatic kidney cancers and B-cell lymphomas (50). In fact, use of IL-2 preoperative as neoadjuvant immunochemotherapy for locally advanced esophageal cancer may cause significant tumor regression in both size and shape (with clear surgical margins and absence of metastasis) (51).
Clinically significant tumor regression of solid metastatic lesions from esophageal cancer has been achieved through the use of locoregional adoptive immunotherapy (AIT). Locoregional administration (either endoscopically injected into primary tumor site or directly injected into metastatic lymph nodes) of autologous lymphocytes stimulated with autologous tumor cells and IL-2 in vitro, could achieve tumor regression in a considerable percentage of patients with advanced and recurrent esophageal cancer, with moderate and tolerable toxicity (52-54). It may be benefitial especially as postoperative adjuvant therapy in esophageal cancer (55). These studies suggest that CTLs rather than LAK cells are needed to achieve the tumor regression. However, tumors that lose expression of MHC class I molecules as a mechanism of escape from immune surveillance are more susceptible to killing by natural killer cells (NK) (Fig 2 C).
Furthermore, various cancers, including human esophageal carcinomas express Fas ligand (FasL) and can kill lymphoid cells (tumor-infiltrating lymphocytes (TIL) by Fas-mediated apoptosis, thereby contributing to the immune privilege of the tumor (56). Another approach involves the use of cis-dichlorodiammineplatinum (CDDP) as a Fas inducer to make esophageal tumors susceptible to Fas antigen and LAK cytotoxic effector cells (57).
Tumor-specific antibodies might be able to direct the lysis of the tumor cells by NK cells via their Fc receptors (Fig 2 B). The use of a monoclonal antibody of murine origin (KIS1) shown to react specifically with an antigen of human squamous cell carcinoma (SCC) had the problem of inducing the generation of human anti-mouse antibody (HAMA). The use of the KIS1 F(ab)2 fragment, i.e. the part of the antibody that interacts with its antigen, may not only overcome this difficulty, but has been shown to be superior to intact KIS1. Furthermore, it may be clinically useful for radioimmunodetection followed by tumor targeting therapy for patients with SCC of the esophagus (58).
The oncolytic herpes simplex-1 virus (NV1066), is a virus that has been engineered to infect and lyse tumor cells selectively. Due to its oncolytic activity in vitro and in vivo, which can be tracked endoscopically as it expresses the gene for green fluorescent protein (GFP), may be a useful therapy against esophageal cancer (59).
Recent progress in gene technology has identified some cancer-rejection genes and peptides such as MAGE, MART, etc. Effective HPV vaccines constitute our most promising weapon in the battle against cervical cancer (60), nevertheless its usefulness would be debatable as we previously discussed that the role of this virus in ESCC is controversial. Since the clinical efficacy of HLA class I-restricted peptide vaccines is still poor, many researchers are mainly administering immuno-cell therapies. As the number of clinicals trials of cancer-specific immunotherapy for esophageal carcinomas continues to increase, we hope that new apects on the way of how to use the immune response to attack this neoplasia will be enlighted.
REFERENCES
1. MONTESANO R, HOLLSTEIN M, HAINAUT P.Genetic alterations in esophageal cancer and their relevance to etiology and pathogenesis: a review. Int J Cancer. 1996 69(3):225-35 [ Links ]
2. HEITMILLER RF.. Epidemiology, diagnosis, and staging of esophageal cancer. Cancer Treat Res. 2001;105:375-86 [ Links ]
3. PARKIN DM, BRAY F, FERLAY J, PISANI P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001 ;94(2):153-6 [ Links ]
4. MATHERS CD, SHIBUYA K, BOSCHI-PINTO C, et al. Global and regional estimates of cancer mortality and incidence by site: I. Application of regional cancer survival model to estimate cancer mortality distribution by site. BMC Cancer. 2002 ;2(1):36. [ Links ]
5. ZUR HAUSEN H. Papillomaviruses and cancer: from basic studies to clinical application. Nat Rev Cancer. 2002; 2(5): 342-50. [ Links ]
6. BOSCH FX, LORINCZ A, MUNOZ N, MEIJER CJ, SHAH KV. The causal relation between human papillomavirus and cervical cancer. J Clin Pathol. 2002 ;55(4):244-65. [ Links ]
7. SYRJANEN KJ. HPV infections and oesophageal cancer. J Clin Pathol. 2002 (10):721-8. [ Links ]
8. SYRJANEN K, PYRHONEN S, AUKEE S, KOSKELA E. Squamous cell papilloma of the esophagus: a tumour probably caused by human papilloma virus (HPV). Diagn Histopathol. 1982 ;5(4):291-6. [ Links ]
9. LAM KY, HE D, MA L, et al. Presence of human papillomavirus in esophageal squamous cell carcinomas of Hong Kong Chinese and its relationship with p53 gene mutation. Hum Pathol. 1997 ;28(6):657-63. [ Links ]
10. HE D, ZHANG DK, LAM KY, et al. Prevalence of HPV infection in esophageal squamous cell carcinoma in Chinese patients and its relationship to the p53 gene mutation. Int J Cancer. 1997 ;72(6):959-64. [ Links ]
11. DE VILLIERS EM, LAVERGNE D, CHANG F, et al. An interlaboratory study to determine the presence of human papillomavirus DNA in esophageal carcinoma from China. Int J Cancer. 1999 ;81(2):225-8. [ Links ]
12. CHANG F, SYRJANEN S, SHEN Q, et al. Evaluation of HPV, CMV, HSV and EBV in esophageal squamous cell carcinomas from a high-incidence area of China. Anticancer Res. 2000 ;20(5C):3935-40. [ Links ]
13. LI T, LU ZM, CHEN KN, et al. Human papillomavirus type 16 is an important infectious factor in the high incidence of esophageal cancer in Anyang area of China. Carcinogenesis. 2001 ;22(6):929-34 [ Links ]
14. SHEN ZY, HU SP, LU LC, et al. Detection of human papillomavirus in esophageal carcinoma. J Med Virol. 2002 ;68(3):412-6. [ Links ]
15 ZHOU XB, GUO M, QUAN LP, et al. Detection of human papillomavirus in Chinese esophageal squamous cell carcinoma and its adjacent normal epithelium.World J Gastroenterol. 2003 ;9(6):1170-3. [ Links ]
16. SMITS HL, TJONG-A-HUNG SP, TER SCHEGGET J, et al. Absence of human papillomavirus DNA from esophageal carcinoma as determined by multiple broad spectrum polymerase chain reactions. J Med Virol. 1995 ;46(3):213-5 [ Links ]
17. KOK TC, NOOTER K, TJONG-A-HUNG SP, et al. No evidence of known types of human papillomavirus in squamous cell cancer of the oesophagus in a low-risk area. Eur J Cancer. 1997 ;33(11):1865-8 [ Links ]
18. VAN DOORNUM GJ, KORSE CM, BUNING-KAGER JC, et al. Reactivity to human papillomavirus type 16 L1 virus-like particles in sera from patients with genital cancer and patients with carcinomas at five different extragenital sites. Br J Cancer. 2003 ;88(7):1095-100. [ Links ]
19. BENAMOUZIG R, JULLIAN E, CHANG F, et al. Absence of human papillomavirus DNA detected by polymerase chain reaction in French patients with esophageal carcinoma. Gastroenterology. 1995 ;109(6):1876-81 [ Links ]
20. ASHWORTH MT, MCDICKEN IW, SOUTHERN SA, NASH JR. Human papillomavirus in squamous cell carcinoma of the oesophagus associated with tylosis. J Clin Pathol. 1993 ;46(6):573-5 [ Links ]
21 MORGAN RJ, PERRY AC, NEWCOMB PV, HARDWICK RH, ALDERSON D. Human papillomavirus and oesophageal squamous cell carcinoma in the UK. Eur J Surg Oncol. 1997 ;23(6):513-7 [ Links ]
22. LAGERGREN J, WANG Z, BERGSTROM R, DILLNER J, NYREN O. Human papillomavirus infection and esophageal cancer: a nationwide seroepidemiologic case-control study in Sweden. J Natl Cancer Inst. 1999 ;91(2):156-62. [ Links ]
23. LAMBOT MA, HAOT J, PENY MO, FAYT I, NOEL JC. Evaluation of the role of human papillomavirus in oesophageal squamous cell carcinoma in Belgium. Acta Gastroenterol Belg. 2000 ;63(2):154-6 [ Links ]
24. WESTON AC, PROLLA JC. Association between esophageal squamous cell carcinoma and human papillomavirus detected by Hybrid Capture II assay. Dis Esophagus. 2003; 16(3): 224-8. [ Links ]
25. SAEGUSA M, HASHIMURA M, TAKANO Y, OHBU M, OKAYASU I. Absence of human papillomavirus genomic sequences detected by the polymerase chain reaction in oesophageal and gastric carcinomas in Japan. Mol Pathol. 1997 ;50(2):101-4. [ Links ]
26. MIZOBUCHI S, SAKAMOTO H, TACHIMORI Y, et al. Absence of human papillomavirus-16 and -18 DNA and Epstein-Barr virus DNA in esophageal squamous cell carcinoma. Jpn J Clin Oncol. 1997 ;27(1):1-5. [ Links ]
27. CERVANTES J, KORIYAMA C, SHUYAMA K, et al. Absence of Human Papillomavirus in Esophageal Squamous Cell Carcinoma Cases from Papua New Guinea. Int J Oncol 2004, submitted. [ Links ]
28. GRAVITT PE, PEYTON CL, ALESSI TQ, et al. Improved amplification of genital human papillomaviruses. J Clin Microbiol. 2000 ;38(1):357-61. [ Links ]
29. KLETER, B., L. J. VAN DOORN, J. TER SCHEGGET, L. et al. A novel short-fragment PCR assay for highly sensitive broad-spectrum detection of anogenital human papillomaviruses. Am. J. Pathol. 1998 153:1731-1739 [ Links ]
30. FUJINAGA, Y., SHIMADA, M., OKAZAWA, K., et al. Simultaneous detection and typing of genital human papillomavirus DNA using the polymerase chain reaction. Journal of General Virology,1991, 72, 1039-1044. [ Links ]
31. SCHEFFNER M, WERNESS BA, HUIBREGTSE JM, et al. The E6 oncoprotein encoded by human papillomavirus types 16 and 18 promotes the degradation of p53. Cell. 1990 ;63(6):1129-36. [ Links ]
32. BOYER SN, WAZER DE, BAND V. E7 protein of human papilloma virus-16 induces degradation of retinoblastoma protein through the ubiquitin-proteasome pathway. Cancer Res. 1996 ;56(20):4620-4. [ Links ]
33. MANDARD AM, HAINAUT P, HOLLSTEIN M. Genetic steps in the development of squamous cell carcinoma of the esophagus. Mutat Res. 2000; 462(2-3):335-42 [ Links ]
34. SCHNEIDER PM, HOLSCHER AH, WEGERER S, et al. Clinical significance of p53 tumor suppressor gene mutations in adenocarcinoma in Barrett esophagus Langenbecks Arch Chir Suppl Kongressbd. 1998;115(Suppl I):495-9 [ Links ]
35. CHANG F, SYRJANEN S, TERVAHAUTA A, et al. Frequent mutations of p53 gene in oesophageal squamous cell carcinomas with and without human papillomavirus (HPV) involvement suggest the dominant role of environmental carcinogens in oesophageal carcinogenesis. Br J Cancer. 1994 ;70(2):346-51. [ Links ]
36. IKEGUCHI M, OKA S, GOMYO Y, et al. Combined analysis of p53 and retinoblastoma protein expressions in esophageal cancer. Ann Thorac Surg. 2000; 70(3):913-7 [ Links ]
37. KATO H, YOSHIKAWA M, FUKAI Y, TAJIMA K, MASUDA N, TSUKADA K, KUWANO H, NAKAJIMA T.An immunohistochemical study of p16, pRb, p21 and p53 proteins in human esophageal cancers. Anticancer Res. 2000 ;20(1A):345-9. [ Links ]
38. SHINOHARA M, AOKI T, SATO S, et al. Cell cycle-regulated factors in esophageal cancer. Dis Esophagus. 2002;15(2):149-54. [ Links ]
39. MATHEW R, ARORA S, KHANNA R, et al. Alterations in p53 and pRb pathways and their prognostic significance in oesophageal cancer. Eur J Cancer. 2002;38(6):832-41. [ Links ]
40. SARBIA M, TEKIN U, ZERIOUH M, et al. Expression of the RB protein, allelic imbalance of the RB gene and amplification of the CDK4 gene in metaplasias, dysplasias and carcinomas in Barretts oesophagus. Anticancer Res. 2001;21(1A):387-92. [ Links ]
41. ARAI H, UENO T, TANGOKU A, et al. Detection of amplified oncogenes by genome DNA microarrays in human primary esophageal squamous cell carcinoma: comparison with conventional comparative genomic hybridization analysis. Cancer Genet Cytogenet. 2003 ;146(1):16-21 [ Links ]
42. STEMMERMANN G, HEFFELFINGER SC, NOFFSINGER A, et al. The molecular biology of esophageal and gastric cancer and their precursors: oncogenes, tumor suppressor genes, and growth factors. Hum Pathol. 1994 ;25(10):968-81. [ Links ]
43. CHANG F, SYRJANEN S, WANG L, SYRJANEN K. Infectious agents in the etiology of esophageal cancer. Gastroenterology. 1992 ;103(4):1336-48. [ Links ]
44. WAHRENDORF J, CHANG-CLAUDE J, LIANG QS, et al. Precursor lesions of oesophageal cancer in young people in a high-risk population in China. Lancet. 1989; 2(8674):1239-41. [ Links ]
45. KINJO Y, CUI Y, AKIBA S, et al. Mortality risks of oesophageal cancer associated with hot tea, alcohol, tobacco and diet in Japan. J Epidemiol. 1998 ;8(4):235-43 [ Links ]
46. ENGEL LS, CHOW WH, VAUGHAN TL, et al. Population attributable risks of esophageal and gastric cancers. J Natl Cancer Inst. 2003 ;95(18):1404-13. [ Links ]
47. MA Y, XIAN M, LI J, et al. Interrelations of clinicopathological variables, local immune response and prognosis in esophageal squamous cell carcinoma. APMIS. 1999 ;107(5):514-22. [ Links ]
48. ATREE SV, CRILLEY PA, CONROY JF, et al. Cancer of the esophagus following allogeneic bone marrow transplantation for acute leukemia. Am J Clin Oncol. 1995 ;18(4):343-7 [ Links ]
49. NAGAO N, KATOH M, KUMAZAWA I, et al.A recurrent case of esophageal cancer in which metastatic skin tumor disappeared after local injection of activated lymphocytes with tumor-pulsed dendritic cells. Gan To Kagaku Ryoho. 1999 ;26(12):1937-9. [ Links ]
50. ROSENBERG SA. Progress in the development of immunotherapy for the treatment of patients with cancer. J Intern Med. 2001 ;250(6):462-75. [ Links ]
51. OKUNO K, TANAKA A, YOSHIKAWA H, et al. A new preoperative immunochemotherapy for the treatment of locally advanced esophageal cancer. Hepatogastroenterology. 1998 ;45(22):950-3 [ Links ]
52. TOH U, YAMANA H, SUEYOSHI S, et al. Locoregional cellular immunotherapy for patients with advanced esophageal cancer. Clin Cancer Res. 2000 ;6(12):4663-73 [ Links ]
53. TOH U, SUDO T, KIDO K, et al. Locoregional adoptive immunotherapy resulted in regression in distant metastases of a recurrent esophageal cancer.Int J Clin Oncol. 2002 ;7(6):372-5 [ Links ]
54. UEDA Y, SONOYAMA T, ITOI H, et al. Locoregional adoptive immunotherapy using LAK cells and IL-2 against liver metastases from digestive tract cancer Gan To Kagaku Ryoho. 2000 ;27(12):1962-5. [ Links ]
55. TOGE T, YAMAGUCHI Y. Lymphokine-activated killer cell adoptive immunotherapy for cancer treatment and its significance. Hum Cell. 1992 ;5(3):218-25 [ Links ]
56. BENNETT MW, OCONNELL J, OSULLIVAN GC, et al.The Fas counterattack in vivo: apoptotic depletion of tumor-infiltrating lymphocytes associated with Fas ligand expression by human esophageal carcinoma. J Immunol. 1998 1;160(11):5669-75. [ Links ]
57. MATSUZAKI I, SUZUKI H, KITAMURA M, et al. Cisplatin induces fas expression in esophageal cancer cell lines and enhanced cytotoxicity in combination with LAK cells. Oncology. 2000 ;59(4):336-43. [ Links ]
58. FUJII T, YAMANA H, TOH Y, et al. The effect of radioimmunotherapy using murine monoclonal antibody KIS1 on esophageal squamous cell carcinoma-bearing nude mice. Surg Today. 1997;27(11):1026-34. [ Links ]
59. STILES BM, BHARGAVA A, ADUSUMILLI PS, et al. The replication-competent oncolytic herpes simplex mutant virus NV1066 is effective in the treatment of esophageal cancer. Surgery. 2003 ;134(2):357-64 [ Links ]
60. GALLOWAY DA. Papillomavirus vaccines in clinical trials. Lancet Infect Dis. 2003 ;3(8):469-75. [ Links ]