Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de Gastroenterología del Perú
versión impresa ISSN 1022-5129
Rev. gastroenterol. Perú v.25 n.1 Lima ene./mar. 2005
ARTICULOS ORIGINALES
Radical surgical treatment for cystic neoplasms of the páncreas: Personal experience
Luciano A. Domingues1; Marcio F. Chedid1; Erico Lombardi1; Priscila F. Viero1, Aljamir D. Chedid1
1 Department of General Surgery Hospital Ernesto Dornelles and Postgraduate Program in Surgery, Federal University of Rio Grande do Sul, Porto Alegre (RS), Brazil
RESUMEN
Antecedentes: Las neoplasias quísticas del páncreas comprenden el 1-10% de todos los tumores pancreáticos. Hemos realizado un análisis del manejo quirúrgico radical de los pacientes con tumores quísticos del páncreas.
Métodos: Se evaluaron los registros de todos los pacientes con neoplasias pancreáticas operados por un mismo cirujano (A.D.C.). Se analizó la sobrevida postoperatoria y las complicaciones quirúrgicas.
Resultados: En el período comprendido entre agosto de 1983 y noviembre de 2003, se realizaron 77 resecciones pancreáticas oncológicas. En 9 (11.7%) de estas operaciones, la patología resecada consistió en neoplasias quísticas del páncreas. Todos los pacientes tratados con resecciones parciales fueron dados de alta. El paciente sometido a la pancreatectomía total falleció nueve días después de la operación. El promedio de seguimiento fue de 44 meses, falleciendo 1 paciente por recurrencia tumoral.
Conclusiones: Los tumores quísticos de páncreas representaron el 11,7 por ciento de todas las neoplasias de nuestra casuística. La resección de esos tumores puede ser realizada en forma segura a través de una pancreatectomía distal. La cirugía de Whipple es un procedimiento de mayor riesgo y su indicación debe ser individualizada. La pancreatectomía total es un procedimiento quirúrgico de alto riesgo y en nuestra opinión debe ser evitada.
Palabras clave: Pancreatectomía, cistadenoma mucoso pancreático, cistadenocarcinoma mucoso pancreatico.
SUMMARY
Background: Cystic neoplasms comprise 1-10% of all pancreatic neoplasms. Most authors recommend resection due to the difficulties in differentiating benign from malignant tumors prior to operation. We performed an analysis on the radical surgical handling of patients with cystic neoplasms of the pancreas.
Patients and Methods: We analyzed data of all patients with pancreatic neoplasms who were treated by potentially curative surgical treatment by a single surgeon (A.D.C.). Postoperative survival and complications were recorded.
Results: Between August 1983 and November 2003, the aforementioned surgeon performed 77 pancreatic resections for pancreatic tumors. Nine of those operations were performed for cystic neoplasms of the pancreas. All patients submitted to partial pancreatic resections were discharged in good health. The patient that underwent a total pancreatectomy died nine days after the procedure. At a mean follow up of 44 months, there has been one local recurrence with death.
Conclusions: Cystic tumors of the pancreas accounted for 11,7 % of the neoplasms studied in our series. Resection of those neoplasms attained by distal pancreatectomy is a safe procedure. PD is a risky procedure and should be viewed cautiously. Total pancreatectomy remains as a high-risk procedure and should be avoided.
Key-words: Pancreatic mucinous cystadenocarcinoma; Pancreatic mucinous cystadenoma; pancreatectomy.
INTRODUCTION
Cystic neoplasms comprise only about 10% of all pancreatic cystic lesions, since most of the latter are pancreatic pseudocysts (1-2). They also comprise 1-10% of all pancreatic neoplasms (1,3). Compagno and Oertel, in 1978, classified pancreatic cystic neoplasms histologically as benign, also known as microcystic (serous cystadenoma), and potentially malignant, also known as macrocystic (mucinous cystic neoplasm) (4, 5). Other uncommon types of pancreatic cystic neoplasms include intraductal papillary-mucinous neoplasm, papillary cystic neoplasm, endocrine cystic tumor, cystic teratoma, and acinar cystadenocarcinoma (3).
The treatment of pancreatic cystic neoplasms is still controversial. Most authors, however, recommend resection due to the difficulties in differentiating benign from malignant tumors prior to surgery (6). We performed an analysis of the short and long-term results after radical resection of pancreatic cystic neoplasms.
PATIENTS AND METHODS
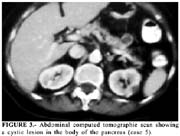
The charts of all patients submitted to pancreatic resections by a single surgeon (A.D. Chedid) between august 1983 and december 2003 were reviewed. Data from all patients submitted to pancreatic resections for cystic neoplasms of pancreas were recorded. Chest X-ray and abdominal computed tomography scan were performed in all patients in order to detect distant metastases. None of those patients had any evidence of disseminated neoplastic disease. The study was approved by the Hospital Ernesto Dornelles Institutional Review Board.
Because of the reduced sample size, the results were analyzed only by descriptive statistics.
RESULTS
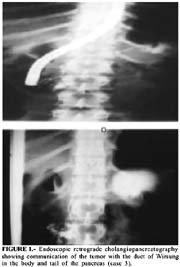
Between August 1983 and December 2003, the aforementioned surgeon performed 77 pancreatic resections for pancreatic tumors. Nine of these operations were performed for cystic neoplasms of pancreas (Table 1). Chest X-ray and abdominal computed tomography scan were performed in all patients in order to detect distant metastases. None of those patients had evidence of disseminated neoplastic disease. Abdominal cholangiopancreatography was performed in one patient (figure 1). None of these patients was submitted to either preoperative biopsy or percutaneous aspiration of the tumor.
All patients were Caucasian. There were seven females and two males. The mean age was 57 years-old (range 18 - 90). One total pancreatectomy, two partial pancreatoduodenectomies and six distal pancreatectomies plus "en bloc" splenectomies were performed.
The operative mortality for partial pancreatic resections (two partial pancreatectomies and six distal pancreatectomies) was null. The only patient submitted to total pancreatectomy died in the 8th postoperative day because of to hypoglycemia and sepsis. Mean postoperative stay length was 13 days (range=7-35 days) and 5 complications were found in 3 patients.
Mean follow-up was 44 months (range=8 days - 108 months), and all patients discharged were alive one year after surgery. One patient died twenty months after procedure secondary to the recurrence of the pancreatic tumor (a mucinous cystic adenocarcinoma).
DISCUSSION
Cystic masses of pancreas are an increasingly frequent diagnostic and therapeutic challenge faced by the surgeon. That is mainly due to the increasing use of abdominal US and CT scan for investigation of abdominal symptoms (6). They are more frequent in women (4:1), with an average age of 60 years at the initial diagnosis (7), which is similar to the characteristics of the herein described cases. The most important differential diagnosis is between cystic neoplasms and pancreatic pseudocysts, which are usually secondary to acute pancreatitis and have a locular appearance. Abdominal CT scan with the use of intravenous contrast is the most important diagnostic tool, rendering arteriography unnecessary most of the times. Clinically, the cystic lesions most likely to be malignant are those that have rapid growth, and symptoms such as weight loss and anorexia, especially in patients with no history of acute pancreatitis.
Sarr et al. recommend percutaneous aspiration of the content of the cyst when there is uncertainty about the diagnosis (8). Russell recommends avoiding preoperative biopsies of pancreatic cystic masses due to the risk of rupture of the cyst and spread of the tumor. The same author does not recommend drainage due to the risk of permanent pancreatic fistula (7). Machado et al. advise against biopsy (including intraoperative frozen section pathological examination) since mucinous cystadenocarcinomas have large parts covered by serous tissue or no epithelium, which could lead to an incorrect diagnosis of serous cystadenoma or pseudocyst (9). They recommend resection of all pancreatic cystic neoplasms, including the asymptomatic ones, except in patients with tumors located in the head of the pancreas and an unacceptable high operative risk. Therefore, we decided resect the tumor without any previous percutaneous aspiration or biopsy of the cyst.
The curative treatment of pancreatic cystic neoplasms is achieved by surgical resection. The resection may consist of enucleation, total or partial pancreaticoduodenectomy (for tumors located in the head of the pancreas), and distal pancreatectomy (for tumors located in the body or tail of the pancreas). We chose not to perform enucleation, since we felt it was an inappropriate approach to potentially malignant neoplasms.
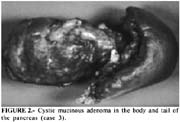
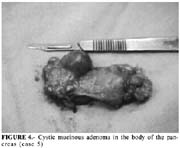
According to Fernandez Del Castillo, mucinous cystadenoma is the most common pancreatic cystic neoplasm, comprising 45% of them (1). Patients are usually females (80%), between 50 and 60 years. About 75% of the tumors are located in the body and/or tail of the pancreas (1). The presenting symptoms are usually abdominal or back pain, and/or weight loss. However, jaundice and bleeding are symptoms more frequently associated with mucinous cystadenocarcinoma, the most aggressive and malignant variant of the mucinous cystic neoplasm (12). ERCP is usually abnormal and may reveal a communication between the normal pancreatic duct and the cyst in the absence of findings suggestive of chronic pancreatitis (11).
Surgical treatment comprises surgical resection (12-13), although Talamini has suggested that enucleation of mucinous neoplasms is a reasonable approach, especially for tumors located on the head or uncinate process of the pancreas (14). He reported a low recurrence rate, but a high rate of pancreatic fistula. For that reason, we chose to perform total resection of the tumors through pancreatectomy in our patients.
Serous cystadenoma is a benign tumor, which predominates in females, and is asymptomatic in a third of the patients. When it become symptomatic, is usually detected by abdominal imaging exams in patients under investigation for a palpable mass and/or vague abdominal pain (15). Most patients undergo operation if the tumor causes symptoms or if there is uncertainty about the diagnosis (16-17), which led us to perform surgery on three of our described cases. The association of serous cystadenoma and acute pancreatitis such as in case 5 is rare (18).
It is rare for pancreatic endocrine tumors to present as cysts. Only about 50 cases are described in the English language literature. They are frequently associated with the multiple endocrine neoplasia type 1 syndrome (MEN-1) (19). According to Ligneau, that diagnosis must be considered in any patient found to have a pancreatic cystic lesion who has a family history of MEN-1 or clinical findings of this syndrome (young patients, multiple pancreatic tumors, evidence of hormone secretion, and endocrine functional syndromes) (19). Endocrine tumors of the pancreas should be treated by surgical resection due to their possible, yet rare, malignant evolution (1, 20- 23).
Papillary cystic neoplasm is a rare disease. Analyzing 508 cases of pancreatic tumors, Cubilla found only one case of pancreatic papillary cystic neoplasm (0.19%) (24). Compagno et al. (25) and Pezzi (26) confirmed the typical presentation of the papillary cystic neoplasm as a large tumor located most commonly in the body and tail of the pancreas, which affects young women in 90% of the cases, and has potential for local invasion, although it rarely metastasizes (2, 27- 28).
Horisawa analyzed cases of metastatic papillary cystic neoplasm and found a high rate of recurrence in patients who underwent local excision, as well as a low rate of morbidity and mortality for pancreatic resections performed by experienced surgeons (29). These data supports our choice to perform such an aggressive operation as the pancreaticoduodenectomy in an 18 years old patient.
Surgical resection is the only potentially curative procedure for the treatment of malignant pancreatic neoplasms. We found that total resection of those neoplasms attained by distal pancreatectomy is a safe procedure, with acceptable morbidity and no mortality in our series. On the other hand, pancreaticoduodenectomy seems to be a riskier procedure and should be opted cautiously, possibly risky preceded by biopsy or cystic fluid analysis in patients with high operative risk. We advise against total pancreatectomy for the treatment of cystic pancreatic neoplasms.
REFERENCES
1. FERNANDEZ-DEL CASTILLO C, WARSHAW AL. Cystic tumors of the pancreas. Surg Clin North Am 1995; 75: 1001-16. [ Links ]
2. MATSUNOU H, KONISHI F, YAMAMICHI N, et al. Solid infiltrating variety of papillary cystic neoplasm of the pancreas. Cancer 1990; 65: 2747-57. [ Links ]
3. LI BD, MINNARD E, NAVA H. Cystic neoplasms of the pancreas- a review. J La State Med Soc 1998; 150: 16-24. [ Links ]
4. COMPAGNO J, OERTEL JE. Microcystic adenomas of the pancreas (glycogen-rich cystadenomas): a clinicopathologic study of 34 cases. Am J Clin Pathol 1978; 69: 289-98. [ Links ]
5. COMPAGNO J, OERTEL JE. Mucinous cystic neoplasms of the pancreas with overt and latent malignancy (cystadenocarcinoma and cystadenoma). A clinicopathologic study of 41 cases. Am J Clin Pathol 1978; 69: 573-80. [ Links ]
6. BALCON IV JH, FERNANDEZ-DEL CASTILLO C, WARSHAW AL. Cystic lesions in the pancreas: when to watch, when to resect. Curr Gastroenterol Rep 2000; 2: 152-8. [ Links ]
7. RUSSELL RC. Unusual pancreatic tumours. In: Neoptolemos JP, Lemoine NR, ed. Pancreatic Cancer Molecular and Clinical Advances. London: Blackwell Science, 1996: 127-31. [ Links ]
8. SARR MG, KENDRICK ML, NAGORNEY DM, et al. Cystic neoplasms of the pancreas: benign to malignant epithelial neoplasms. Surg Clin North Am 2001; 81(3): 497-509. [ Links ]
9. MACHADO MC, MONTAGNINI AL, MACHADO MA et al. Cystic neoplasm of the pancreas: analysis of 24 cases. Rev Hosp Clin Fac Med S Paulo 1994; 49 (5): 208-12. [ Links ]
10. LE BORGNE J, DE CALAN L, PARTENSKY C. Cystadenomas and Cystadenocarcinomas of the pancreas. A Multiinstitutional Retrospective Study of 398 cases. French surgical Association. Ann Surg 1999; 230 (2): 152-61. [ Links ]
11. GAZELLE GS, MUELLER PR, RAAFAT N, et al. Cystic neoplasms of the pancreas: evaluation with endoscopic retrograde pancreatography. Radiology 1993; 188: 633-6. [ Links ]
12. THOMPSON LD, BECKER RC, PRZYGODZKI RM, et al. Mucinous cystic neoplasm (mucinous cystadenocarcinoma of low grade malignant potential) of the pancreas: a clinicopathologic study of 130 cases. Am J Surg Pathol 1999; 23: 1-16. [ Links ]
13. ZAMBONI G, SCARPA A, BOGINA G, et al. Mucinous cystic tumors of the pancreas. Am J Surg Pathol 1999; 23: 410-22. [ Links ]
14. TALAMINI MA, MOESINGER R, YEO CJ, et al. Cystadenomas of the pancreas: is enucleation an adequate operation? Ann Surg 1998; 227: 896-903. [ Links ]
15. LE BORGNE J. Cystic tumours of the pancreas. Br J Surg 1998; 85: 577-9. [ Links ]
16. PYKE CM, VAN HEERDEN JA, COLBY TV, et al. The spectrum of serous cystadenoma of the pancreas. Clinical, pathologic and surgical aspects. Ann Surg 1992; 215: 132-9. [ Links ]
17. WARSHAW A, RATTNER DW, FERNANDEZ-DEL CASTILLO C, Z'GRAGGEN K. Middle segment pancreatectomy: a novel technique for conserving pancreatic tissue. Arch Surg 1998; 133: 327-31. [ Links ]
18. FURUKAWA H, TAKAYASU K, MUKAI K, et al. Serous cystadenoma of the pancreas communicating with a pancreatic duct. Int J Pancreatol 1996; 19: 141-4. [ Links ]
19. LIGNEAU B, LOMBARD-BOHAS C, PARTENSKY C, et al. Cystic endocrine tumors of the pancreas: clinical, radiologic, and histopathologic features in 13 cases. Am J Surg Pathol 2001; 25 (6): 752-60. [ Links ]
20. NOJIMA T, KOJIMA T, KATO H, et al. Cystic endocrine tumor of the pancreas. Int J Pancreatol 1991; 10: 65-72. [ Links ]
21. RATTNER DW, FERNANDEZ-DEL CASTILLO C, WARSHAW AL. Cystic pancreatic neoplasms. Ann Oncol 1999; 10 Suppl. 4:104-6. [ Links ]
22. STAIR JM, SCHAEFER RF, MCCOWAN TC, BALACHANDRAN S. Cystic islet cell tumours of the pancreas. J Surg Oncol 1986; 32: 46-9. [ Links ]
23. THOMPSON NW, ECKHAUSER FE, VINIK AI, et al. Cystic neuroendocrine neoplasms of the pancreas and liver. Ann Surg 1984; 199: 158-64. [ Links ]
24. CUBILLA AL, FITZGERALD PJ. Tumours of the exocrine pancreas. In: Hartmann WH, ed. Atlas of Tumor Pathology. Washington, DC: Armed Forces Institute of Pathology, 1984: 201-7. [ Links ]
25. COMPAGNO J, OERTEL JE, KREMZAR M. Solid and Papillary epithelial neoplasm of the pancreas, probably of small duct origin: a clinicopathologic study of 52 cases. Lab Invest 1979; 40: 248-9. [ Links ]
26. PEZZI CM, SCHUERCH C, ERLANDSON RA, DEITRICK J. Papillary-cystic neoplasm of the pancreas. J Surg Oncol 1988; 37: 278-85. [ Links ]
27. KINGSNORTH AN, GALLOWAY SW, LEWIS-JONES H, et al. Papillary cystic neoplasm of the pancreas: presentation and natural history in two cases. Gut 1992; 33: 421-3. [ Links ]
28. YAMAGUCHI K, HIRAKATA R, KITAMURA K. Papillary cystic neoplasm of the pancreas: radiological and pathological characteristics in 11 cases. Br J Surg 1990; 77: 1000-3. [ Links ]
29. HORISAWA M, NIINOMI N, SATO T, et al. Frantz's tumor (solid and cystic tumor of the pancreas) with liver Metastasis: successful treatment and long-term-follow-up. J Pediatric Surg 1995; 30(5): 724-6. [ Links ]
CORRESPONDING AUTHOR:
Dr. Aljamir D. Chedid. Address: Rua Marechal Andréa 300/201. Bairro Boa Vista. Porto Alegre (RS), Brazil. Phone and Fax: 55 51 33282472. E-Mail: aljamir@terra.com.br