Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de Gastroenterología del Perú
versión impresa ISSN 1022-5129
Rev. gastroenterol. Perú v.31 n.4 Lima oct./dic. 2011
ARTÍCULO DE REVISIÓN
Endoscopy Ultra- Sonography –Guided Biliary Drainage in the Surgical –Endoscopy Era
Everson L. A. Artifon ; Jose Pinhata Otoch ; Luiz F. Poli-de-Figueiredo ; Paulo Sakai ; Samir Rasslan
* GI Endoscopy Service, Department of Surgery; University of Sao Paulo-Sao Paulo, Brazil
RESUMEN
ANTECEDENTES: La Ecoendoscopia guiada para hepático - gastrostomía, colédoco duodenostomía y colédoco antrostomía, son procedimientos avanzados de la endoscopia biliar y pancreático, y juntos forman el drenaje biliar eco-guiada. La Hepático - gastrostomía está indicada en casos de obstrucción hiliar, mientras que en las lesiones distales el procedimiento de elección es la colédoco -duodenostomía. Ambos procedimientos se deben hacer solamente después de la CPRE sin éxito.
OBJETIVOS: Para aclarar a los lectores sobre la indicación de estos procedimientos, que deben ser realizados conforme a un punto de vista multidisciplinaria, con un intercambio de información con el paciente ó su representante legal.
RESULTADOS: Por separado se indicó en la definición de los procedimientos de drenaje biliar EUS e incluye los detalles de las técnicas y análisis crítico.
CONCLUSIÓN: La hepático-gastrostomía y colédoco duodenostomía-son factibles cuando es realizada por endoscopistas con experiencia en endoscopia pancreática biliar y de eco-endoscopia y se debe realizar en la actualidad bajo un protocolo riguroso en las instituciones educativas.
PALABRAS CLAVE: Ultrasonografia endoscópica, Drenaje biliar.
ABSTRACT
BACKGROUND: US-guided hepatico -gastrostomy, choledocho-duodenostomy and choledocho-antrostomy are advanced procedures on biliary and pancreatic endoscopy and together make up the echo-guided biliary drainage. Hepatico - gastrostomy is indicated in cases of hiliar obstruction, while the procedure of choice is choledocho - duodenostomy in distal lesions. Both procedures must be done only after unsuccessful ERCP.
AIMS: To clarify to the readers about indication of these procedures, they must be made under a multidisciplinary view while sharing information with the patient or legal guardian.
METHODS: All series cases report and selected cohort studies were selected according to the DDTS system in which key words were EUS biliary drainage, choledocho-duodenostomy, hepatico-gastrostomy, EUS, palliation and pancreatic biliary advanced cancer.
RESULTS: Separately it was stated definition on the EUS biliary drainage procedures and it includes the techniques details and critical analysis.
CONCLUSION: Hepatico- gastrostomy and Choledocho- duodenostomy are feasible when performed by endoscopists with expertise in bilio pancreatic endoscopy and advanced echo-endoscopy and should be performed currently under rigorous protocol in educational institutions.
KEY WORD: Endoscopic Ultrasonography, Biliary Drainage.
Endoscopic biliary stenting at ERCP is a well established therapy for both benign and malignant biliary obstruction [1-3]. To overcome ERCP failures and improve outcomes over those afforded by more invasive alternatives -percutaneous trans hepatic biliary drainage (PTBD) and surgery-EUS-guided ductal access techniques paired with standard ERCP drainage techniques have been developed in the last decade. This hybrid procedure is given a variety of names, but the more encompassing one is endosonographic cholangio pancreatography (ESCP) [4]. Based on the combination of the three possible access routes (intra hepatic bile duct, extra hepatic bile duct and pancreatic duct) with the three possible drainage routes (trans mural, trans papillary ante grade and trans papillary retrograde), ESCP admits nine variant approaches, six for the bile duct and three for the pancreatic duct [5,6]. The six ESCP variant approaches to bile duct drainage are also referred as EUS-guided biliary drainage (EUSBD). Trans mural bile duct drainage under EUS effectively creates a bilio -digestive anastomosis, since the stent is placed across the GI tract wall and the bile duct.
This article discusses first the EUSBD technique that provides trans mural drainage from an extra hepatic bile duct access route, and is most commonly termed EUS-guided choledocho duodenostomy (EUS-CDS) and the Trans mural intra hepatic EUSBD (hepatico gastrostomy) is discussed below.

I. RATIONALE
As stated above, EUSBD is divided by access route into EUSguided intrahepatic bile duct drainage, where the intrahepatic bile duct is punctured from a trans esophageal, trans gastric or trans jejunal approach, and EUS-guided extra hepatic bile duct drainage, where the common bile duct (CBD) is punctured from a trans duodenal or trans gastric approach (usually from the distal antrum). The overall rationale for EUS-CD is shared by the alternative EUSBD techniques, and it is threefold: 1) logistic advantage (it can be performed in the same session as the originally failed ERCP without further delay); 2) physiologic advantage (it provides immediate internal biliary drainage without the need for external drains); and 3) anatomic advantage (it can be tailored to the individual patients anatomy; the precise imaging afforded by EUS resulting in a potentially less invasive procedure than PTBD).
In addition to the underlying common rationale for EUSBD implicit in EUS-CDS, there is a specific rationale for it. The CBD is more easily imaged under EUS than the intrahepatic bile ducts, in contrast to what happens under trans abdominal US. This means that it can be imaged and accessed under EUS without added risks even in patients with minimal or no bile duct dilation. In those patients with dilated bile ducts, the CBD is a much more obvious target for puncture than the intrahepatic ducts. This results in faster, cleaner access without repeated puncture attempts, thereby minimizing risks. The retroperitoneal location of the CBD makes it also an attractive access site for patients with as-cites, in whom fluid around the liver makes trans hepatic access (whether percutaneous or trans gastric under EUS) more difficult and hazardous.
Besides the advantages of extra hepatic access over intra hepatic access, the specific rationale for EUS-CDS is also derived from the trans mural drainage route, as opposed to trans papillary EUSBD (ante grade or rendezvous). As explained in more detail, ante grade stent insertion from an extra hepatic access site is challenging and has only been reported in two exceptional cases [7,8]. The real choice between trans mural and trans papillary drainage after extra hepatic bile duct access under EUS therefore lies between EUS-CDS and rendezvous. Proponents of rendezvous argue that it may be less invasive than EUS-CDS, since trans mural intervention is usually limited to puncture and guide wire passage, then drainage is accomplished retrograde via ERCP without the need for puncture tract dilation [9]. However, EUSBD rendezvous carries a 20% failure rate – even in expert centers-because guide wire passage across the stricture and the papilla is often unsuccessful. The needle allows virtually no interplay with the guide wire, which cannot be manipulated across the stricture through a needle in the same way as it can be done at ERCP using flexible catheters. EUSBD needlerendezvous (that is, without creating a fistula to allow passage into the bile duct through the puncture tract of flexible devices to help manipulate the guide wire ante grade) may require repeat punctures with different angles or trying different types of guide wires, often resulting in a prolonged, labor-intensive procedure. The second part of rendezvous following ante grade guide wire passage involves scope exchange and guide wire retrieval, and it is also cumbersome and plagued with difficulties. In summary the advantages of EUS-CDS over trans papillary rendezvous are its higher success rate and relative simplicity, which appear to make it a more reproducible approach, despite being perhaps more invasive. Nonetheless, both EUSBD variant approaches can be considered complementary inasmuch as these procedures are used in a heterogeneous patient population. As we will discuss below, some indications are better suited for EUS-CDS, whereas in other cases EUSBD rendezvous is clearly advantageous. Similarly, even if rendezvous is the intended drainage technique, EUS-CDS can be used as a second line approach to salvage the significant proportion of failed rendezvous cases[10, 11]. This open-ended approach to EUSBD (i.e. inclusive of both rendezvous and EUS-CDS) results in comparatively higher success rates than that of EUSBD series limiting their approach to just rendezvous [9].It is important to know that choledocho antrostomy, described by Artifon et al. (54), is a new technique that is useful for those patients with duodenal bulb infiltration and should be a new and feasible tool as a variant of choledocho duodenostomy.
II. TECHNICAL DATA, DISCUSSION OF POSSIBLE THERAPIES AND RECOMMENDATION OF THE PROSTHESIS, PRACTICAL RECOMMENDATIONS FOR PROPOSED ENDOSCOPIC TECHNIQUES
a. Indication
In common with other EUSBD techniques, EUS-CDS should only be considered in patients with confirmed (not just suspected) biliary obstruction after failed ERCP despite maximal attempts by experienced operators. General patient, operator and equipment requirements are the same as for other EUSBD techniques. However, EUS-CDS has specific anatomic requirements differing from other EUSBD alternatives. The first anatomic requirement is distal biliary obstruction. In other words, EUS-CDS is not suitable for proximal (hilar) biliary obstruction, where intrahepatic EUSBD approaches are clearly required. The second anatomic requirement is the ability to image under EUS the CBD. Since the CBD is typically imaged from the distal stomach or the duodenal bulb, this is difficult to impossible in patients with prior gastrectomy and gastro jejunostomy (e.g., Roux-en-Y)[12].
Finally, as with most other EUSBD approaches, EUSCDS is predominantly used in patients with malignant biliary obstruction. But whereas alternative approaches such as rendezvous may rightly be considered after failed cannulation in patients with documented benign causes of biliary obstruction (e.g., CBD stones or papillary stenosis), EUS-CDS is less adequate in these distinct settings, where biliary drainage is usually accomplished by means of sphincterotomy (with or without stone removal) as opposed to stenting.
b. Procedure
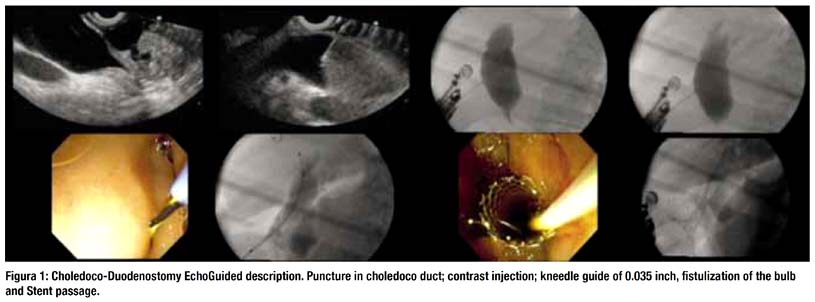
As stated above, puncture of the CBD from the duodenum (EUS-CDS) is the most common approach. A similar approach from the stomach (EUS-choledocho gastrostomy or EUS-choledocho antrostomy) may also be used in selected instances depending on the patients anatomy (see below). The CBD is visualized from the duodenal bulb by using a curved linear array echo endoscope in a long or a short scope position. The direction of the needle in the long scope position is toward the hiliar (proximal) bile duct. The direction of the needle in the short scope position is toward the lower (distal) bile duct. The correlation between scope position and needle orientation is not always straightforward. Anatomic distortion may make necessary additional fine adjustments involving torque of the echo endoscope shaft and/or the control wheels. The orientation of the needle can be checked with fluoroscopy before the puncture is actually carried out. It is relevant to do so, because an upward needle orientation makes EUS-CDS easier, since it tends to decrease the angle for trans mural stent advancement over the guide wire into the bile duct. Conversely, a downward needle orientation is sought when rendezvous is intended as the initial drainage choice.
Two types of needle devices are available for access. Conducting flexible needles, commonly used at ERCP for pre-cut and pseudo cyst drainage, using electrocautery (Endo Cut ICC200, ERBE ELEKTROMEDIZIN GmbH, Tübingen, Germany). The so called needle-knife (Zimmon papillotome, Cook Endoscopy, Winston-Salem, NC), used for precut, produces axial cutting with a thin wire extending 2 mm beyond the tip of the catheter. The so called cystotome or fistulotome (Cook Endoscopy, Winston-Salem, NC; Endoflex, Tubingen, Germany), traditionally used for pseudo cyst drainage, has a blunt, round cutting piece at the tip that produces circumferential cutting. Cystotomes are slightly stiffer than needle-knifes and produce a larger burn on the duodenal and CBD walls. This larger, round cutting reduces the need for dilation before stent insertion. Cystotomes are therefore particularly useful in cases where resistance to the advancement of flexible devices over the wire into the duct is met. Thinner caliber cystotomes (6-Fr) are preferable to larger caliber ones (10-Fr). Needle-knives, on the other hand, being more flexible, can be used free-hand under EUS as the initial access device. There are also non-conducting stiff cutting needles, commonly used for EUS-guided fine needle aspiration (EUS-FNA). EUS-FNA needles are available in several calibers. The two most commonly used are the large 19-gauge needle and the thin 22-gauge needle (EchoTip, Cook Endoscopy). A specific needle for EUSBD has recently been developed by Cook. Whatever the needle choice, it is inserted Trans duodenaly into the bile duct under EUS visualization. To confirm needle ductal access, the stylet is removed and bile is aspirated. If there is a bile return, contrast medium is injected into the bile duct for cholangiography, then, a 450 cm long, 0.035-inch, 0.021-inch, or 0.018inch guide wire is inserted through the outer sheath and its position is confirmed fluoroscopically. We will comment below on differential guide wire features. If there is no return of bile or a bloody aspirate, the needle is removed, flushed with saline inside the gastrointestinal lumen to prevent clogging, and a repeat puncture attempted. Nonetheless, the problem of a needle apparently inside the duct under EUS but in actual fact on a different plane usually occurs when accessing very small ducts,
which is hardly ever the case during EUS-CDS. After guide wire access into the bile duct, some dilation of the puncture track is usually necessary, using either a dilating biliary catheter (Soehendra biliary dilator, Cook Endoscopy), a papillary balloon dilator (Maxpass, Olympus medical systems, Tokyo, Japan) or both sequentially (axial dilator first, then balloon dilator). This is aimed at dilating the duodeno choledochal fistula to facilitate stent insertion. The need for dilation is maximal when no cautery is used for initial entry under EUS, a stiffer (metal) or larger caliber plastic (10-Fr) stent is intended, and when the distance to the CBD or the resistance felt during the initial advancement of the needle are greater. Finally, a 5-Fr to 10-Fr biliary pig-tail or straight plastic stent or a fully covered self-expandable metal stent (SEMS) [Zeon Medical Co. Ltd. Tokyo, Japan] is inserted through the choledocho duodenostomy site into the CBD. Care should be taken to monitor by fluoroscopy the intraductal placement of the proximal end of the stent and to monitor by endoscopy the intra duodenal (or intra gastric) position of the distal (closer to the scope) end of the stent. This latter aspect is of particular relevance when using SEMS. SEMS tend to foreshorten upon full expansion, which takes place a few hours after the procedure. Early SEMS dislodgment may be caused by foreshortening towards the CBD beyond the GI wall. To prevent this serious complication an adequate length of SEMS (15-20 mm) should be left inside the GI lumen. This is longer than what is customarily done when placing SEMS Trans papillary at ERCP. Additional anchorage techniques to prevent dislodgment are forceful balloon dilation of the SEMS up to 8-10 mm after initial deployment, or the use of a coaxial double pig-tail through the SEMS, as reported for pseudo cyst drainage using trans mural SEMS [13].
Despite the seemingly simple sequence of duct imaging and puncture under EUS, guide wire advancement and track dilation under fluoroscopy, and eventually stent insertion and deployment under combined fluoroscopic and endoscopic monitoring, EUS-CDS is an invasive, complex procedure. Knowledge about the full array of needle devices, guide wires, dilators and stents as well as about the subtle variations in scope position (gastric or duodenal), scope orientation (upward and downward), and stent anchoring techniques is highly recommended to increase success rates and minimize complications. Operator confidence with specific devices also plays a role. Some authors feel that access without cautery is less prone to complications. These authors favor initial non-conducting needle access and then use cautery only selectively after failed mechanical dilation over the guide wire of the puncture tract [6, 14]. Mechanical dilation without cautery requires a stiffer 0.035-inch guide wire for support, which in turn involves the use of a 19-gauge EUS-FNA needle. Other authors find the stiffer 19-gauge EUS-FNA needles cumbersome to use in the relatively long position of the echo endoscope in the duodenum, and resort to either initial direct needle-knife access under EUS [15], or needleknife access under a thinner 0.018 guide wire passed into the CBD after puncture with a 22-gauge EUS-FNA needle [16]. Finally, some other authors resort to both needle-knife and EUS-FNA needle access [17]. These procedural variations as reported in the literature are listed in Table I, and will be discussed next.
III. LITERATURE FINDINGS BASED ON THE PERS-PECTIVE OF EVIDENCE-BASED MEDICINE [5-27]
EUS-guided choledocho duodenostomy was first reported by Giovannini et al.[18]. Some authors exchanged the echo endoscope over a catheter-protected guide wire for a duodenoscope, through which the stent was eventually inserted. As detailed earlier, the puncture needles available are conducting needles and non conducting needles. About half the number of each has been used in published reports. This is in contrast to what is reported for intrahepatic EUSBD, where no conducting needle access is clearly preferred. The reason why cautery access (conducting needle) is favored during EUS-CDS is probably fourfold. Firstly, for EUS-CDS the echo endoscope is in a longer, curved position in the duodenum in comparison with the shorter distance to the subcardial region from where intrahepatic access is typically gained. This long position increases friction between the stent delivery system and the endoscope working channel, which impairs the transmission of the pushing force, thereby making trans mural stent insertion more difficult. Secondly, the thicker, fibrous wall of the CBD is harder to penetrate mechanically than the relatively soft liver parenchyma (except in cases with underlying cirrhosis) and the wall of smaller bile ducts. Thirdly, the tendency to create a space by pushing until the bile duct wall yields is greater between the duodenal wall and the CBD than between the gastric wall and the liver. Finally, the CBD is larger and has the nearest vessels at a greater distance than the intrahepatic bile ducts (where vessels run closely in parallel), which offers some protection against severe bleeding, a feared complication of cautery access.
In most reported cases, a plastic stent has been placed. However, recently, the use SEMS is increasingly been reported [14]. The success rate for the 61 cases reported to date is as high as 95%, with excellent results in all successfully drained patients (100% per-protocol clinical response rate). There were some cases where stent insertion was too difficult and a naso biliary drainage tube was placed instead [17,24]. Another interesting variation on EUS-CDS is illustrated by a few cases where the extra hepatic bile duct was punctured from the stomach rather than the standard trans duodenal approach[14,20]. Although only 6 cases were reported, all were successful.
IV. EXPECTED COMPLICATIONS AND TREATMENT OPTIONS
Complications can be divided into procedure-related complications and stent-related complications. Definitions of procedural complications are not well standardized. Most are related to bile (or just air) leakage into the retro peritoneum (with trans duodenal access) or the peritoneum (with trans gastric access to the CBD), with or without added infection. The severity ranges from a self-limiting condition that resolves within 48-72 hours with conservative measures, to full-blown peritonitis requiring emergency surgery. Most reported complications are mild. The need for emergency surgery is exceedingly rare. Other interventional measures that may be required in the event of complications, such as percutaneous drainage, are however not all that uncommon.
Peri-procedural leakage of bile into the abdominal cavity is most likely due to poor drainage. Poor drainage can be caused by factors such as too large a fistula, early stent clogging, and inappropriate positioning of the stent (including foreshortening of SEMS).
Late stent-related complications, that is, once a mature fistula is formed, are similar to those seen with trans papillary stents placed at ERCP, namely, migration and stent occlusion. Stent migration or occlusion is managed in the same way as in stents placed at ERCP, by inserting a new stent. The technique for repeat stent placement differs from what is commonly done at ERCP. If a clogged plastic stent is in place across the fistula, a guide wire is advanced through the stent and the stent is grasped with a snare passed over-the-wire and removed over it. This somewhat more complex maneuver is aimed at keeping guide wire access to the duct after stent removal. After plastic stent removal, a SEMS may be placed using a duodenoscope. If clogging of a SEMS occurs, the debris occluding its lumen may be cleaned up. But just cleaning is probably not long-lasting in this setting. A new coaxial stent needs to be placed inside the clogged one, either a plastic stent, or a SEMS, the so called stent-in-stent approach.
Distal stent migration into the GI tract lumen with a mature fistula only involves repeat biliary drainage, since migrated stents usually pass out spontaneously. Repeat biliary drainage may be attempted in several ways. The simplest one is placing a new stent through the same fistula, if it is still visible. If the fistula can not be identified endoscopically, either repeat EUS-CDS through a new puncture site or PTBD are required. If proximal stent migration to the retro peritoneum or the peritoneum occurs, recovery of the stent as well as emergency surgery should be considered. This serious complication, however, has not yet been reported for EUS-CDS. Finally, even if the less serious distal migration occurs but the fistula is still immature (a fibrous track not yet formed), this may cause bile leakage into the abdomen. In the event of stent migration and leakage with an immature fistula, repeat EUS-guided biliary drainage (perhaps using a SEMS), or PTBD need to be considered. Surgery should also be considered depending on the patients condition.

General patient, equipment and operator requirements for EUSBD are listed in further. We will further describe here the equipment and devices required for EUS-HG, common to most other EUSBD approaches. A step-by-step description of EUS-HG will be presented next. Finally, the specific place of EUS-HG within the context of other EUSBD approaches will be discussed and the published literature on it briefly reviewed.
EQUIPMENT AND DEVICES
A. INTERVENTIONAL ECHO ENDOSCOPES
Around 1990, the Pentax-Corporation developed an electronic convex curved linear array echo endoscope (FG32UA) with an imaging plane in the long axis of the endoscope and aligned with the instrumentation plane. This echo endoscope, equipped with a 2.0mm working channel, enabled fine-needle aspiration biopsy under EUS guidance (EUSFNA). However, the relatively small working channel of the FG32UA was a drawback for therapeutic intervention. As an example, drainage of a non-bulging pseudo cyst using this early instrument was soon reported, but it required exchanging the echo endoscope for a therapeutic duodenoscope in order to insert a stent31. To enable stent placement using an echo endoscope, interventional echo endoscopes (FG 38X, EG 38UT and EG 3870UTK) were developed by Pentax-Hitachi. The FG 38X has a working channel of 3.2 mm, which allows the insertion of a 8.5 French stent or nasocystic drain. The EG38UTand EG3870UTK have larger working channels of 3.8mm and are equipped with an elevator, thereby allowing the placement of a 10 French stent32,33.
The Olympus Corporation has also developed convex linear array echo endoscopes. The GFUC 30P has a biopsy channel of 2.8mm, which enables the placement of 7 French stents or nasocystic catheters. This echo endoscope is also equipped with an elevator. A new prototype, the GFUCT 30, has a larger working-channel of 3.7 mm allowing the placement of a 10 French stent . The main drawback of convex linear array echo endoscopes is the more limited imaging field (120° using the Pentax and 180° using the Olympus) produced by an electronic transducer. The Olympus instruments are coupled with the Aloka processor or with a smaller processor (Suzie).
B. NEEDLES AND ACCESSORIES FOR DRAINAGE
As already described, needles used for bile duct access under EUS can be categorized into flexible, cautery needles (needleknives or fistulotomes) and stiff, cutting needles (EUS-FNA needles). Needle knives can be difficult to visualize endosonographically. The "Zimmon" needle-knife (Wilson-Cook Corporation, Winston Salem, North Carolina, USA) has a large gauge needle that is relatively easy to visualize compared to other needle-knives. Cautery is usually required to penetrate through the intervening structures into the bile duct when a needle-knife is used29. A cystotome is a more stable diathermic sheath and has a round cutting tip instead of a needle. Cystotomes are commonly used during pancreatic pseudo cyst drainage 32. The calibre used for pseudo cyst drainage is usually 8.5 to 10 French. A modified small calibre cystotome (6 French), also referred to as "fistulotome" (Endolflex, Voerde, Germany) is more convenient for EUSBD.
Standard EUS-FNA needles are well visualized endosonographically and can be used for non-cautery access to the bile duct. The drawback of the most commonly used EUSFNA needles is their small calibre (22 or 23 G) allowing only 0.018 inch guide wires. Using a larger 19G FNA needle (Wilson-Cook Corporation), a 0.0035 inch guide wire can be inserted through the needle into the dilated bile duct. As explained in Chapters 13-14 and 16-17, one of the main problems with EUS-FNA needle access to the duct is the difficulty in manipulating the guide wire through the needle. The main trouble is the "stripping" of the wire coating, which in turn risks leaving part of it into the patient. Furthermore, a strip-off or cut-off wire usually prevents stent insertion over it, which results in procedural failure unless a repeat puncture is attempted. As the intrahepatic bile duct rapidly collapses upon initial puncture, and the subsequent contrast or bile extravasation may substantially impair the endosonographic view, repeat puncture is not always feasible when EUS-HG is the approach to EUSBD pursued.
To solve the problem of guide wire damage with standard EUS-FNA needles, we worked with Cook Medical to design a new special needle called the EchoTip® Access Needle. This needle is original because it has a sharp stylet that makes it relatively easy to insert the needle into the bile duct, the pancreatic duct or a pseudo cyst. When the stylet is withdrawn, the outer needle sheath is left in place with a blunt, non cutting tip..Manipulation of the guide wire without incurring the risk of damaging the guide wire is easy with this blunt tipped needle sheath.
TECHNIQUE OF EUS-GUIDED HEPATICO-GAS-TROSTOMY (EUS-HG)
As in the alternative extra hepatic access EUSBD technique for trans mural drainage (i.e., choledocho duodenostomy), EUS-HG is closely related to EUS-guided drainage of pancreatic pseudocysts32. In all these cases, the target is imaged under EUS and punctured with a needle. The puncture tract is then dilated (using cautery, mechanical devices, or both), and a stent is placed across the puncture tract to drain the duct or the pseudo cyst into the GI tract lumen.
EUS-guided hepatico gastrostomy was first reported in 2003. Burmester and coworkers used EUS-HG in a Billroth II patient with unresectable pancreatic cancer and failed ERCP because of tumor infiltration of the papilla. In the same series, another patient with recurrent gastric cancer and total gastrectomy had a trans mural stent placed across the jejunal wall below the gastro jejunostomy, i.e. EUS-guided hepaticojejunostomy34. We also reported in 2003 EUS-HG in a patient with subtotal gastrectomy and recurrent malignancy. The left biliary system was inaccessible, because a metal stent had been previously placed percutaneously in the right hepatic duct across the confluence35.
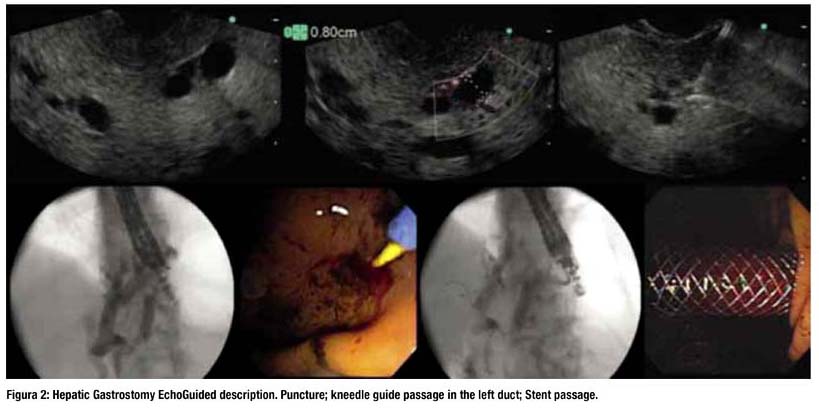
The procedural steps of EUS-HG are as follows. Using an interventional echo Endoscope, the dilated left hepatic duct (usually segment III) is well visualized. EUS-HG is then performed under combined fluoroscopic and ultrasound guidance, with the tip of the echo endoscope positioned such that the ultrasound transducer is either in the middle part of the small curvature of the stomach or slightly upwards, closer to the cardia. A needle (19 G, EchoTip® Access Needle, Cook Ireland Ltd., Limerick, Ireland) is inserted transgastrically into a peripheral branch of the left hepatic duct, and contrast medium is injected. Before contrast is injected, bile can be aspirated through the needle in order to confirm the intraductal position of the needle tip. Opacification delineates fluoroscopically the dilated biliary tree down to the point of obstruction. The needle is exchanged over a guide wire (0.02 inch diameter, Terumo Europe, Leuven, Belgium) for a 6.0 French diathermic sheath (Cysto-Gastro set, Endo-Flex, Voerde, Germany) , which is then used to enlarge the channel between the stomach (or jejunum in patients with total gastrectomy) and the left hepatic duct. The diathermic sheath is advanced across the intervening liver parenchyma by using cutting current. After removing over a guide wire (TFE-coated 0.035 inch diameter, Cook Europe, Bjaeverskov, Denmark) the diathermic sheath, an 8.5 French, 8-cmlong hepatico-gastric stent) or an 8 cm long covered self-expandable metal stent (SEMS) (partially covered Wallstent or fully covered Wallflex, Boston-Scientific, Nattick, Massachusetts, USA) is placed transmurally. Fluoroscopy confirms adequate stent placement and function by showing contrast drainage through the stent into the stomach.
Bile leakage into the peritoneum is the major risk of EUS-HG. Several strategies are used by different authors to minimize this risk. A 6 or 7 French naso-biliary drain with mild aspiration or gravity drainage can be left in place through the metal stent during 48 hours, even if this is somewhat inconvenient to the patient. More recently we have developed a more patient-friendly approach to minimize the risk of leakage, by combining an uncovered metal stent with a covered metal stent inside. The uncovered stent is deployed initially, so as to provide anchorage and prevent migration, and then the covered stent is inserted coaxially and deployed within the first stent. Finally, in cases where the guide wire crosses the downstream stricture ante gradely, hepaticogastrostomy can be combined with ante grade placement of an additional metal stent bridging the distal stricture, which further decreases the pressure gradient across the trans mural stent by providing additional downstream decompression of the bile duct36.Alternative strategies used by other authors to prevent migration include the used of fully covered SEMS with both ends flared37 or forceful balloon expansion upon stent deployment (as opposed to gradual spontaneous selfexpansion over several hours) -to monitor foreshortening- plus insertion of a double pig-tail stent through the expanded SEMS -to provide additional anchorage38.
EUS-HG IN COMPARISON WITH OTHER EUSBD APPROACHES.
As discussed in the previous two chapters of this book, the rationale for all variant EUSBD approaches as a second-line option in select difficult cases where ERCP is not feasible is threefold. EUSBD may be potentially more convenient (performed in the same session), more physiologic (allowing immediate internal biliary drainage) and less invasive (affording more accurate control as well as more access sites to the bile duct) than the classic alternatives of percutaneous biliary drainage (PTBD) or surgery.
The specific anatomic features of patients that may make EUS-HG preferable to other EUSBD are based on the intrahepatic access route and the trans mural drainage route. Intrahepatic access is the only choice in patients with proximal (hilar) biliary obstruction and is usually more convenient in patients with distal gastrectomy, since imaging the CBD under EUS is not always possible in the setting of postoperative altered anatomy.39 One advantage of trans mural drainage after intrahepatic bile duct access over trans papillary drainage is that the challenging step of ante grade guide wire passage (required for both rendezvous and antegrade stenting) is avoided. In addition to guide wire passage, rendezvous requires an accessible papilla, which is usually not the case in patients with surgically altered anatomy or tight duodenal stenoses. Ante grade stent insertion does not require an accessible papilla, but involves dilation of the puncture tract, just as EUS-HG. In patients with postoperative anatomy, ante grade trans papillary stenting without combined hepatico-gastrostomy is less convenient for stent revisions, since HG provides easy repeat access to the bile duct without the need for a repeat puncture. Stent revisions are not uncommonly required during follow-up. As illustrated in Fig.3, the advantages of EUS-HG over rendezvous or ante grade stent insertion are particularly relevant in patients with prior duodenal or biliary SEMS who experience recurrent biliary obstruction40.
These variant EUSBD approaches must, however, be viewed as complementary rather than mutually exclusive. For example, as mentioned when discussing strategies to minimize the risk of bile leakage in EUS-HG, ante grade trans papillary stents can be combined with trans mural stenting36. Puspok et al performed ante grade trans papillary SEMS insertion in a patient with recurrent gastric cancer after Roux-en-Y gastrectomy. They then left a transmural plastic stent across the puncture tract both to minimize the risk of leakage and to preserve access41. Dual drainage (ante grade and trans mural) has also been used serially. Fujita et al performed trans oesophageal EUSBD by inserting a 7 French plastic stent into a peripheral left bile duct branch in a patient with advanced gastric cancer42. Ten days later, the plastic stent was cannulated with a guide wire and removed over it with a snare43. Then, using flexible devices through the mature fistula, the guide wire was manipulated under fluoroscopy across the malignant distal bile duct stricture, and a SEMS passed ante gradely over the wire was subsequently deployed across the stricture above the papilla.
Patients with distal bile duct obstruction without prior gastrectomy who have both intra and extra hepatic bile duct dilation (and no gross ascites) are the only ones in whom there is an issue about which access site for EUSBD might be preferable, intra hepatic or extra hepatic. If the selection criteria for EUSBD versus PTBD are broad (i.e. EUSBD is favoured as the initial second-line approach after failed ERCP), this type of patients may represent just 20% of the candidate population44. Operator preference plays a part in this small patient subset. As highlighted in Chapter 14, the CBD offers a more obvious target for EUS puncture, the echoendoscope is in a more anchored position, and probably access to the CBD makes rendezvous easier than it is with intrahepatic access. On the other hand, intrahepatic EUSBD is performed with the echoendoscope in a more straight position, which favours transmission of the pushing force during stent insertion. It is also probably easier to penetrate a small intrahepatic bile duct surrounded by liver parenchyma than the fibrotic, hard wall of the CBD.
LITERATURE REVIEW
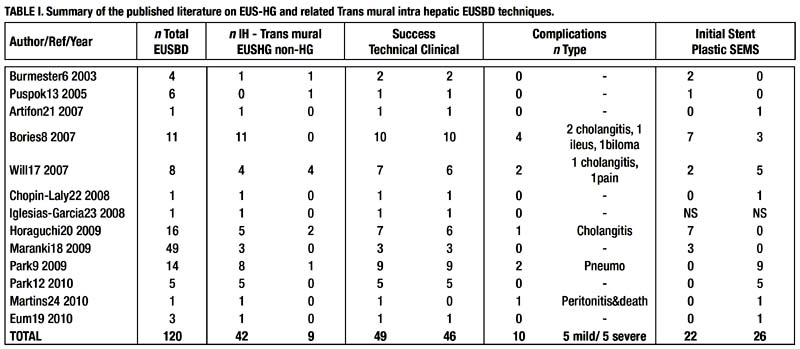
To date, trans mural intra hepatic EUSBD has been reported in 51 patients, EUS-HG in 42 and other closely related variant approaches through a trans jejunal or a trans oesophageal route in 9. In five patients with total gastrectomy, the left bile duct was similarly accessed under EUS from below the cardia and trans mural stents were placed across the jejunal wall. In the remaining four patients a cephalic peripheral left bile duct branch was selected for puncture, so that eventually the stent pierced the wall of the intra-abdominal oesophagus slightly above the cardia. Approximately half of these patients come from three small series specifically dealing with trans mural intra hepatic EUSBD36,40,45, whereas the other half comes from either mixed series in which EUS-HG is reported along extra hepatic EUSBD34,37,41,46-48 or individual case reports35,42,49-52 (Table 1).
EUS-HG (or its variants) was technically successful in 49 out of these 51 patients, with clinical resolution of biliary obstruction in 46 cases. Therefore EUS-HG had a 94% per-protocol success rate and a 90.2% success rate on an intention-to-treat basis. These success rates are very high, considering the difficult patient population in which EUS-HG was attempted. However, three facts deserve consideration. First, these results come from highly experienced operators at referrals centres. Secondly, there is definitely a significant publication bias, i.e. since positive studies are more likely to be published, and this patient cohort is derived from small series and individual case reports, in real practice outcomes are probably somewhat less favourable. Finally, success was achieved at the expense of an overall 20% complication rate, twice as high as that of ERCP. Most complications were accounted for by inadequate biliary drainage, resulting in either peritoneal bile leakage or cholangitis (Table 1). Plastic stents caused cholangitis due to early migration48 or early clogging36. Foreshortening of trans mural SEMS led to bile peritonitis or biloma, requiring percutaneous drainage and repeat EUSBD36, and caused the only reported death to date52. Half of the complications were nonetheless mild, manifested by transient abdominal pain with or without neumo peritoneum that settled on conservative measures.
There is great consistency across all reports on EUSHG regarding technical details. FNA needle access was used initially in all but two cases, in which cautery access using a prototype fistulotome was used instead34. Boogie or balloon dilation was performed before stent insertion in all but four cases, the two just mentioned in which a fistulotome was used, a case in which the tract was dilated after FNA-needle guide wire placement with the tapered tip of a wallstent49, and finally another case in which apparently just cautery was used for access, since no mention of dilation is made52 The one technical aspect in which there is less uniformity is the use of cautery, be it needle-knives or fistulotomes. Overall, any diathermy use was reported in just 39.5% of cases. Whereas some authors use it routinely36, others resort to it selectively37 (only after failure to advance a mechanical dilator over the guide wire) or do not use it at all45.
From a clinical standpoint, however, the most relevant technical choice appears to be the type of stent. As detailed in Table I, 7 to 8.5 plastic stents were placed in 46% of cases, whereas uncovered, partially covered or fully covered SEMS were placed initially in 54%. It is difficult to draw significant conclusions from the published reports, since no formal comparisons have been made between the two types of stents. SEMS are appealing for three reasons. Firstly, upon full expansion SEMS effectively seal the puncture/dilation tract, which would in theory prevent leakage more effectively. Secondly, their larger diameter provides better long-term patency, which would decrease the need for stent revisions. Finally, if dysfunction by ingrowth or clogging occurs, management is somewhat less challenging than with plastic stents, since a new stent (plastic or SEMS) can easily be inserted through the occluded SEMS in place. In contrast, exchanging a clogged plastic trans mural stent usually requires over-the-wire replacement, because free-hand removal involves the risk of track disruption with subsequent guide wire passage into the peritoneum, hence requiring repeat EUSBD (or PTBD) to re-establish drainage43. These presumed advantages of SEMS must be balanced against the fact that trans mural SEMS insertion and deployment are somewhat more demanding than they are at ERCP. In particular, the serious risk of foreshortening and bile peritonitis should be prevented with careful attention to detail52.
Bories et al. had recently reported their experience in 38 patients (11 with benign disease and 27 with malignancy) using trans gastric EUSBD with trans mural, trans papillary (ante grade) or combined stent insertion. The technical success rate was 97%, and all successfully stented patients improved clinically. However, the complication rate was 25% (5 bile peritonitis, 3 stent migration, 1 liver abscess). There was one death caused by bile peritonitis, and the rest resolved under conservative management53.
EUSBD: EUS-guided biliary drainage, HG: hepaticogastrostomy; SEMS: Self-expandable metal stent; Pneumo: pneumoperitoneum. Case reports from Giovannini et al35 and Fujita et al42 not tallied because already included in case series by Bories et al36 and Horaguchi et al48, respectively. (From Giovannini & Bories, Gastroenterology and research practice 2011).
CONCLUSIONS
These techniques are invasive procedures that require careful patient selection and experienced operators backed by a multidisciplinary team. Further technical improvements are likely to reduce number of adverse events and will probably contribute to the more widespread adoption of these procedures as a second-line approach to biliary drainage after failed ERCP. Although multicenter trials aimed at standardizing the technique for performing EUS-guided biliary drainage would be desirable, the relatively few patient candidates for it and the wide spectrum of technical variations reported to date make this endeavor difficult to accomplish in the near future. Detailed prospective studies with homogeneous inclusion criteria and careful follow-up and dedicated hands-on training models will probably be more effective in advancing this burgeoning field of interventional endoscopy.
REFERENCES
1. FOGEL EL, SHERMAN S, DEVEREAUX BM, LEHMAN GA. Therapeutic biliary endoscopy [review]. Endoscopy 2001; 33:31-8.
2. SMITH AC, DOWSETT JF, RUSSELL RC, et al. Randomised trial of endoscopic stenting versus surgical bypass in malignant low bile duct obstruction. Lancet 1994; 344:1655-60.
3. LAI EC, MOK FP, TAN ES, et al.
Endoscopic biliary drainage for severe acute cholangitis. N. Engl. J. Med. 1992; 24:1582-6.4. WIERSEMA, M. J., et al.
Endosonography-guided cholangiopancreatography. Gastrointest.Endosc. 43.2 Pt 1 (1996): 102-06.5. UPTA, K., et al. Endoscopic ultrasound and percutaneous access for endoscopic biliary and pancreatic drainage after initially failed ERCP. Rev.Gastroenterol. Disord. 7.1 (2007): 22-37.
6. PEREZ-MIRANDA M, DE LA SERNA C, DIEZ-REDONDO P, VILA JJ. Endosonography-guided cholangiopancreatography as a salvage drainage procedure for obstructed biliary and pancreatic ducts. World J. Gastrointest. Endosc. 2010; 16:2:212-2.
7. PUSPOK A, LOMOSCHITZ F, DEJACO C, et al.
Endoscopic ultrasound guided therapy of benign and malignant biliary obstruction: A case series. Am. J. Gastroenterol. 2005; 100: 1743-1747.8. NGUYEN-TANG, T., et al.
"Endoscopic ultrasound (EUS)-guided transhepatic anterograde self-expandable metal stent (SEMS) placement across malignant biliary obstruction. Endoscopy 42.3 (2010): 232-36.9. KIM, Y. S., et al. "Endoscopic ultrasound rendezvous for bile duct access using a trans duodenal approach: cumulative experience at a single center. A case series. Endoscopy 42.6 (2010): 496-502.
10. MARANKI J, HERNANDEZ AJ, ARSLAN B, et al.
Interventional endoscopic ultrasound-guided cholangiography: Long-term experience of an emerging alternative to percutaneous trans hepatic cholangiography. Endoscopy 2009; 41: 532-538.11. BRAUER BC, CHEN YK, FUKAMI N, SHAH RJ. Single-operator eus-guided cholangiopancreatography for difficult pancreaticobiliary access (with video). Gastrointest. Endosc. 2009; 70: 471-479.
12. WILSON, J. A., et al. "EUS in patients with surgically altered upper GI anatomy." Gastrointest.Endosc. 72.5 (2010): 947-53.
13. TALREJA JP, SHAMI VM, KU J, et al. Trans enteric drainage of pancreatic-fluid collections with fully covered self-expandable metallic stents (with video). Gastrointest Endosc 2008;68:1199-203.
14. PARK DH, KOO JE, OH J, et al. EUS-guided biliary drainage with one-step placement of a fully covered metal stent for malignant biliary obstruction: A prospective feasibility study. Am. J. Gastroenterol. 2009; 104:2168-74.
15. YAMAO K, SAWAKI A, TAKAHASHI K, et al. EUSguided choledochoduodenostomy for palliative biliary drainage in case of papillary obstruction: report of 2 cases. Gastrointest. Endosc. 2006;64:663-714.
16. TARANTINO I, BARRESI L, REPICI A, TRAINA M. EUS-guided biliary drainage: A case series. Endoscopy 2008; 40: 336-339.
17. ITOI T, ITOKAWA F, SOFUNI A, et al. Endoscopic ultrasound-guided choledochoduodenostomy in patients with failed endoscopic retrograde cholangiopancreatography. World J. Gastroenterol. 2008; 14: 6078-6082.
18. GIOVANNINI M, MOUTARDIER V, PESENTI C, et al. Endoscopic ultrasound-guided bilioduodenal anastomosis: A new technique for biliary drainage. Endoscopy 2001; 33: 898-900.
19. BURMESTER E, NIEHAUS J, LEINEWEBER T, HUETTEROTH T. EUS-cholangio-drainage of the bile duct: Report of 4 cases. Gastrointest. Endosc. 2003; 57: 246-251.
20. KAHALEH M, HERNANDEZ AJ, TOKAR J, et al.
Interventional EUS-guided cholangiography: Evaluation of a technique in evolution. Gastrointest. Endosc. 2006; 64: 52-59.21. FUJITA N, NODA Y, KOBAYASHI G, et al.
Histological changes at an endosonography-guided biliary drainage site: A case report. World J Gastroenterol. 2007; 13: 5512-5515.22. ANG TL, TEO EK, FOCK KM. Eus-guided trans duodenal biliary drainage in unresectable pancreatic cancer with obstructive jaundice. JOP 2007; 8: 438-443.
23. YAMAO K, BHATIA V, MIZUNO N, et al.
Eus-guided choledochoduodenostomy for palliative biliary drainage in patients with malignant biliary obstruction: Results of long-term follow-up. Endoscopy 2008; 40: 340-342.24. HORAGUCHI J, FUJITA N, NODA Y, et al.
Endosonography-guided biliary drainage in cases with difficult transpapillary endoscopic biliary drainage. Dig. Endosc. 2009; 21:239-44.25. HANADA K, IIBOSHI T, ISHII Y. Endoscopic ultrasound-guided choledochoduodenostomy for palliative biliary drainage in cases with inoperable pan-creas head carcinoma. Dig. Endosc. 2009; 21(Suppl. 1):S75-S78.
26. IWAMURO M, KAWAMOTO H, HARADA R, et al.
Combined duodenal stent placement and endoscopic ultrasonography-guided biliary drainage for malignant duodenal obstruction with biliary stricture. Dig. Endosc. 2010; 22:236-40.27. ITOI T, YAMAO K. EUS 2008 Working Group document: evaluation of EUS-guided choledochoduodenostomy (with video). Gastrointest. Endosc. 2009;69 (Suppl.): S8-S12.
28. ITOI T, SOFUNI A, ITOKAWA F, et al. Endoscopic ultrasonography-guided biliary drainage. J. Hepato biliary Pancreat. Surg. 2010; 17:611-6.
29. GIOVANNINI M, PESENTI C, BORIES E, CAILLOL F. Interventional EUS: difficult pancreaticobiliary access. Endoscopy 2006; 38 Suppl. 1:S93-S95.
30. KAHALEH M. EUS-guided cholangio-drainage and rendezvous techniques. Tech. Gastrointest. Endosc. 2007; 9:39-45.
31. GRIMM H, BINMOELLER KF, SOEHENDRA N. Endosonography-guided drainage of a pancreatic pseudo cyst. Gastrointest. Endosc. 1992; 38:170-171.
32. GIOVANNINI M. EUS-guided pancreatic pseudo cyst drainage. Tech Gastrointest. Endosc. 2007; 9:32-38.
33. GIOVANNINI M. Ultrasound-guided endoscopic surgery. Best Pract. Res. Clin. Gastroenterol. 2004; 18:183-200.
34. BURMESTER E, NIEHAUS J, LEINEWEBER T, HUETTEROTH T. EUS-cholangio-drainage of the bile duct: report of 4 cases. Gastrointest. Endosc. 2003;57:246-251.
35. GIOVANNINI M, DOTTI M, BORIES E, MOUTARDIER V, PESENTI C, DANISI C, DELPERO JR. Hepaticogastrostomy by echo-endoscopy as a palliative treatment in a patient with metastatic biliary obstruction. Endoscopy 2003; 35:1076-1078.
36. BORIES E, PESENTI C, CAILLOL F, LOPES C, GIOVANNINI M. Transgastric endoscopic ultrasonography-guided biliary drainage: results of a pilot study. Endoscopy 2007; 39:287-291.
37. PARK DH, KOO JE, OH J, LEE YH, MOON SH, LEE SS, SEO DW, LEE SK, KIM MH. EUS-guided biliary drainage with one-step placement of a fully covered metal stent for malignant biliary obstruction: a prospective feasibility study. Am. J. Gastroenterol. 2009; 104:2168-2174.
38. PEREZ-MIRANDA M, DE LA SERNA C, DIEZ-REDONDO P, VILA J. Endosonography-guided cholangio pancreatography as a salvage drainage procedure for obstructed biliary and pancreatic ducts. World J. Gastrointest. Endosc. 2010; 2:212-222.
39. WILSON JA, HOFFMAN B, HAWES RH, ROMAGNUOLO J. EUS in patients with surgically altered upper GI anatomy. Gastrointest. Endosc. 2010; 72:947-953.
40. PARK DH, SONG TJ, EUM J, MOON SH, LEE SS, SEO DW, LEE SK, KIM MH. EUS-guided hepatico gastrostomy with a fully covered metal stent as the biliary diversion technique for an occluded biliary metal stent after a failed ERCP (with videos). Gastrointest. Endosc. 2010; 71:413-419.
41. PUSPOK A, LOMOSCHITZ F, DEJACO C, HEJNA M, SAUTNER T, GANGL A. Endoscopic ultrasound guided therapy of benign and malignant biliary obstruction: a case series. Am. J. Gastroenterol. 2005; 100:1743-1747.
42. FUJITA N, NODA Y, KOBAYASHI G, ITO K, OBANA T, HORAGUCHI J, TAKASAWA O, SUGAWARA T. Temporary endosonography-guided biliary drainage for trans gastrointestinal deployment of a self-expandable metallic stent. J Gastroenterol. 2008; 43:637-640.
43. FUJITA N, SUGAWARA T, NODA Y, KOBAYASHI G, ITO K, OBANA T, HORAGUCHI J, TAKASAWA O. Snare-over-the-wire technique for safe exchange of a stent following endo sonography-guided biliary drainage. Dig. Endosc. 2009; 21:48-52.
44. PEREZ-MIRANDA M, DE LA SERNA C, DIEZ-REDONDO MP, GOMEZ DE LA CUESTA S., GIL-SIMON P., ALCAIDE N., CARO-PATON A. Endosonographyguided cholangio pancreatography (ESCP) as the primary approach for ductal drainage after failed ERCP. Gastrointest. Endosc. 2010; 71:AB136.
45. WILL U, THIEME A, FUELDNER F, GERLACH R, WANZAR I, and Meyer F. Treatment of biliary obstruction in selected patients by endoscopic ultrasonography (EUS)-guided trans luminal biliary drainage. Endoscopy 2007; 39:292-295.
46. MARANKI J, HERNANDEZ AJ, ARSLAN B, JAFFAN AA, Angle JF, Shami VM, Kahaleh M. Interventional endoscopic ultrasound-guided cholangiography: long-term experience of an emerging alternative to percutaneous trans hepatic cholangiography. Endoscopy 2009; 41:532-538.
47. EUM J, PARK DH, RYU CH, KIM HJ, LEE SS, SEO DW, LEE SK, KIM MH. EUS-guided biliary drainage with a fully covered metal stent as a novel route for natural orifice trans luminal endoscopic biliary interventions: a pilot study (with videos). Gastrointest. Endosc. 2010; 72:1279-1284.
48. HORAGUCHI J, FUJITA N, NODA Y, KOBAYASHI G, ITO K, OBANA T, TAKASAWA O, KOSHITA S, KANNO Y. Endosonography-guided biliary drainage in cases with difficult transpapillary endoscopic biliary drainage. Dig. Endosc. 2009; 21:239-244.
49. ARTIFON EL, CHAVES DM, ISHIOKA S, SOUZA TF, MATUGUMA SE, SAKAI P. Echo guided hepaticogastrostomy: a case report. Clinics (Sao Paulo) 2007; 62:799-802.
50. CHOPIN-LALY X, PONCHON T, GUIBAL A, ADHAM M. Endoscopic bilio gastric stenting: a salvage procedure. Surgery 2009; 145:123.
51. IGLESIAS-GARCIA J, LARINO-NOIA J, SEIJO-RIOS S, DOMINGUEZ-MUNOZ JE.
Endoscopic ultrasound for cholangio carcinoma re-evaluation after Wall stent placement. Rev Esp. Enferm. Dig 2008; 100:236-237.52. MARTINS FP, ROSSINI LG, FERRARI AP. Migration of a covered metallic stent following endoscopic ultrasound-guided hepatico-gastrostomy: fatal complication. Endoscopy 2010; 42 Suppl. 2:E126-E127.
53. BORIES E, PESENTI C, CAILLOL F, GIOVANNINI M. Endoscopic ultrasound-guided biliary procedures: report of 38 cases [abstract]. Endoscopy 2008;40 (Suppl 1):A55.
54. ARTIFON ELA, OKAWA L, TAKADA J, et al EUS guided choledochoamtrostomy :an alternative for biliary drainage in unresectable pancreatic cancer with duodenal invasion.Gastrointest. Endoscopy 2011,73(6)1317-20.













