Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de Gastroenterología del Perú
versión impresa ISSN 1022-5129
Rev. gastroenterol. Perú v.33 n.1 Lima ene./mar. 2013
ARTÍCULO DE REVISIÓN
Improving early detection of gastric cancer: a novel systematic alphanumeric-coded endoscopic approach
Endoscopía sistemática alfanumérica codificada (SACE): un nuevo método para mejorar el diagnóstico del cáncer gástrico temprano
Fabian Emura 1,2,3,a,b,c, Ian Gralnek 4,a,c,d, Yasushi Sano 5,a,b, Todd H. Baron 6,a,c
1 Advanced Gastrointestinal Endoscopy, EmuraCenter LatinoAmerica, Bogotá DC, Colombia.
2 Emura Foundation for the Promotion of Cancer Research, Bogotá DC, Colombia.
3 Medical School, Universidad de La Sabana, Bogotá DC, Colombia.
4 GI Outcomes Unit, Technion-Israel Institute of Technology, Haifa, Israel.
5 Endoscopy Division, Sano Hospital, Kobe, Japan.
6 Division of Gastroenterology and Hepatology, Mayo Clinic, Rochester, Minnesota, USA.
a MD, b PhD, c FASGE, d MSHS
RESUMEN
A pesar del uso globalizado y masivo de la Esofagogastroduodenoscopia estándar (EGD), el cáncer gástrico (CG) sigue siendo uno de los cánceres más comunes y continúa posicionado como el tumor maligno más frecuente en el Este de Asia, Este de Europa y partes de América Latina. Las limitaciones actuales de usar un examen no sistemático durante la EGD pueden parcialmente ser responsables de la baja incidencia del diagnóstico de CG temprano en países con una alta prevalencia de la enfermedad. Originalmente propuesta por Emura et al., la Endoscopia Sistemática Alfanumérica Codificada (SACE) es un nuevo método que facilita el examen completo y sistemático del tracto gastrointestinal alto basado en la foto-documentación secuencial con imágenes parcialmente superpuestas y que, usando una nomenclatura alfanumérica endoluminal compuesta de ocho regiones y 28 áreas proporciona el cubrimiento del total de la superficie gastrointestinal durante una endoscopia alta. Para la localización precisa tanto de zonas normales o anormales, SACE incorpora un sistema sencillo de coordenadas basado en la identificación de ciertos ejes naturales, paredes, curvaturas y puntos de referencia anatómicos endoluminales. La efectividad de SACE fue recientemente demostrada en un estudio de tamizaje que diagnosticó CG temprano con una frecuencia de 0,30% (2/650) en voluntarios sanos con riesgo promedio. Este nuevo método de examen endoscópico si se aplica de manera uniforme en el mundo, podría considerablemente aumentar la detección temprana del cáncer gástrico y cambiar significativamente la manera en que realizamos endoscopia digestiva en en nuestra era.
Palabras clave: Endoscopía del sistema digestivo; Cáncer gástrico; Tamizaje; Anatomic landmarks (fuente: DeCS BIREME).
ABSTRACT
Despite extensive worldwide use of standard esophagogastroduodenoscopy (EGD) examinations, gastric cancer (GC) is one of the most common forms of cancer and ranks as the most common malignant tumor in East Asia, Eastern Europe and parts of Latin America. Current limitations of using non systematic examination during standard EGD could be at least partially responsible for the low incidence of early GC diagnosis in countries with a high prevalence of the disease. Originally proposed by Emura et al., systematic alphanumeric-coded endoscopy (SACE) is a novel method that facilitates complete examination of the upper GI tract based on sequential systematic overlapping photo-documentation using an endoluminal alphanumeric-coded nomenclature comprised of eight regions and 28 areas covering the entire surface upper GI surface. For precise localization or normal or abnormal areas, SACE incorporates a simple coordinate system based on the identification of certain natural axes, walls, curvatures and anatomical endoluminal landmarks. Efectiveness of SACE was recently demonstrated in a screening study that diagnosed early GC at a frequency of 0.30% (2/650) in healthy, average-risk volunteer subjects. Such a novel approach, if uniformly implemented worldwide, could significantly change the way we practice upper endoscopy in our lifetimes.
Key words: Endoscopy, digestive system; Gastric cancer; Screening; Anatomic landmarks (source: MeSH NLM).
BACKGROUND
Despite extensive worldwide use of standard esophagogastroduodenoscopy (EGD) examinations, gastric cancer (GC) continues to be one of the most common forms of cancer accounting for approximately 870,000 new cases, 650,000 deaths (1) and 10% of new cancer cases annually throughout the world (2). It ranks as the most common malignant tumor in East Asia, Eastern Europe and parts of Latin America (3,4) as well as being the main cause of cancer death in Colombia and other countries (5,6).
Although GC is a curable disease if diagnosed at an early stage (7,8) and several early detection endoscopic studies have demonstrated a significant quantitative impact on GC mortality (9,10), however it is noteworthy that there are only a few reports on endoscopic diagnosis of early GC in countries with a high prevalence of the disease.
In addition, progress in GC reduction has been not attributed entirely to early endoscopic detection, but at least in part to reduced exposure to known risk factors such as Helicobacter pylori infection, high intake of salt-preserved foods and dietary nitrite and low intake of fruit and vegetables (11).
Although several recommendations have previously been promulgated (12-14), currently performed standard EGD from the perspective of the authors lacks sequentiality, systematization, order, proper nomenclature and a protocol for completeness that could be partially responsible for the low incidence of early GC diagnosis in countries with a high prevalence of the disease in an advanced stage.
In light of continuing improvements in the early detection of GC, this review analyzes the limitations of standard EGD and highlights the potential benefit of a novel systematic alphanumeric-coded method for upper gastrointestinal endoscopy.
LIMITATIONS OF STANDARD EGD
There have not been any published reports on an effective protocol for completeness in upper GI examinations. The manner in which EGD is presently performed is based primarily on how professors and instructors have been teaching trainees for generations in accordance with their own individual perceptions and experience in various parts of the world resulting in EGD being very much of a non-standardized procedure.
Although the most commonly performed procedure in the GI arena, endoscopists do not follow a particular order, do are not lacks consistent in start and end points, do not use precise endoluminal nomenclature for describing regions or areas within the stomach and tends to record only abnormal mucosal findings. The concept of conducting a complete examination is not widely applied or required for practitioners. From a global point of view, the situation is even more disheartening because EGDs are now being performed by physicians from several different specialties including gastroenterologists, gastrointestinal surgeons, surgeons and internists in many countries making procedural standardization and definitions of quality indicators high priority issues.
Although there is an increasing demand for proper documentation of endoscopic procedures and a rapidly expanding availability of digital image storage for satisfying such documentation requirements (15), it is our perception that except for some Japanese referral centers recommending a set of at least 40 stomach images in many facilitiess only a relatively small number of endoscopic images are actually stored for each examination. As a consequence, EGD reports are far from being either complete or standardized in contrast to other diagnostic tests such as x-ray, CT scans and MRI.
Recently, a considerable amount of attention has been focused on high-cost equipment capable of producing high-definition images, but we believe performing a detailed and complete examination could have an even greater impact in terms of early diagnosis.
FUNDAMENTAL ENDOSCOPY ENDOLUMINAL ANATOMY
Esophageal anatomical landmarks
Besides the easily recognizable superior and inferior esophageal sphincters, we have identified other constant esophageal landmarks based on extensive observation specifically the main left bronchus/aortic arch groove and the left atrium groove.
Natural esophageal endoscopic axis
The esophagus is anatomically located in the posterior mediastinum immediately behind the main left bronchus and left atrium. Endoluminally, the portion of the circumference in contact with the main left bronchus/aortic arch and left atrium grooves, therefore, corresponds to the anterior esophageal wall. Thus, the natural esophageal endoscopic axis is easily obtainable by either positioning the endoscope with the main left bronchus/aortic arch or the left atrium grooves (anterior wall) at 9-12 oclock.
Esophageal walls
Having the anterior wall localized at 9-12 oclock, the posterior wall is located, therefore, on the opposite side at 3-6 oclock. With the patient in the left lateral position, water flushed through the endoscope channel flows downward filling a portion of the circumference. This portion of the esophagus then corresponds to the left lateral wall at 6-9 oclock and the right lateral wall is situated, therefore, directly opposite at 12-3 oclock.
Stomach anatomical landmarks
In addition to easily recognizable endoluminal landmarks such as the cardia, pyloric ring, angulus, duodenal bulb and second duodenal portion, we have identified several other anatomical features that are not as apparent, but believe these are key landmarks nonetheless for achieving a complete sequential examination. These include the "epithelium transition zone" at the greater curvature separating the lower and the middle body thirds of the stomach and the "gastric spur" which is an inflection of the greater curvature at the middle and upper bodies.
Natural stomach endoscopic axis
The stomach has two curvatures, the lesser and greater curvatures (16). For the lower and middle bodies, the natural stomach axis is easily obtainable by positioning the endoscope with the folds of the greater curvature in the lower half of the visual field.
Stomach walls
There are two gastric walls, the anterior and posterior walls (16). By observing the gastric lumen as the circumference for the antrum, lower and middle bodies, the anterior wall is located at 8-11 oclock, lesser curvature at 11-2 oclock, posterior wall at 2-5 oclock and greater curvature at 5-8 oclock.
SYSTEMATIC ALPHANUMERIC-CODED ENDOSCOPY
Originally proposed by Emura et al. (7,17,18), systematic alphanumeric-coded endoscopy (SACE) is a screening method that facilitates complete examination of the upper GI tract based on both sequential systematic overlapping photo-documentation and fundamental endoscopic endoluminal anatomy.
Similar to performing CT for a detailed study of an entire abdominal mass, SACE provides for a thorough endoscopic examination of the complete upper GI tract surface without any blind spots starting at the hypopharynx and continuing through the esophagus, stomach and second portion of the duodenum.
In order to perform such a complete examination, SACE utilizes a novel endoluminal alphanumeric-coded endoscopy nomenclature comprised of eight regions (Figure 1) and 28 areas (Figure 2). For precise localization or normal or abnormal areas, SACE incorporates a simple coordinate system based on the identification of certain natural axes, walls, curvatures and anatomical endoluminal landmarks. Regions, areas and the alpha-numeric codification is shown in Table 1.
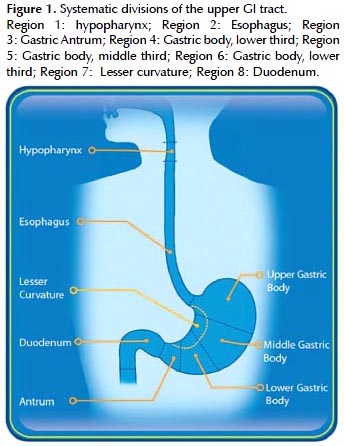
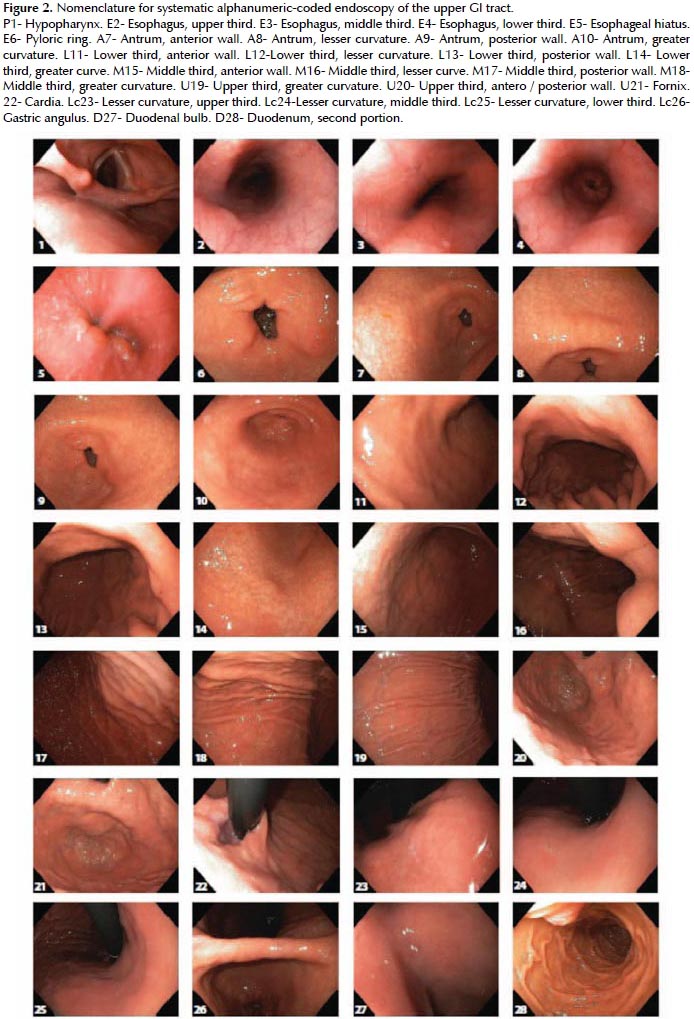
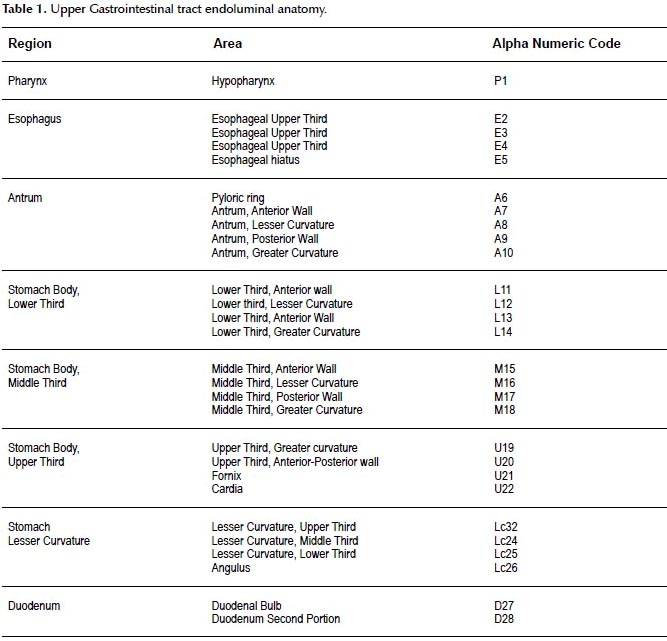
Pharynx – Region P
A complete hypopharyngeal examination includes photo documentation of the posterior wall, both pyriform sinuses and larynx with air inspiration facilitating vocal cord abduction and an unobstructed observation (P1). Endoscopic examination of the pharynx offers the best possibility for early diagnosis and curative treatment for high-risk pharyngeal cancer patients. Despite the current lack of a consensus, we believe examination of the pharynx in high-risk patients would be of considerable value as an integral part of a complete upper endoscopic examination (19).
Esophagus – Region E
Natural esophageal endoluminal landmarks divide the esophagus into four clearly distinct areas. The upper third (E2) extending from the upper esophageal sphincter to the esophageal main left bronchus/aortic arch groove. The middle esophagus (E3) from the esophageal main left bronchus/aortic arch groove to the left atrium groove. The distal esophagus (E4) from the left atrium groove downward to the distal esophagus and finally the esophageal hiatus (E5) by using the direct endoscope view.
Gastric antrum – Region A
Antrum examination includes evaluation of the pyloric ring area (A6), anterior wall area (A7), lesser curvature area (A8), posterior wall area (A9) and greater curvature area (A10). It should be noted that the pyloric ring is visible in all overlapping images of this region.
Lower gastric body – Region L
Lower body examination includes evaluation of the anterior wall area (L11), lesser curvature area (L12), posterior wall area (L13) and greater curvature area (L14) also known as the "epithelium transition zone". The pyloric ring is not visible in any overlapping images of this region.
Middle gastric – Region M
Middle third examination includes evaluation of the anterior wall area (M15), lesser curvature area (M16), posterior wall area (M17) also known as the "gastric spur" and greater curvature area (M18) which image is characterized by horizontal disposition of the gastric folds.
Upper gastric body – Region U
Upper third examination includes evaluation of the greater curvature area (U19) also which image is characterized by horizontal disposition of the gastric folds, anterior and posterior walls (U20), fornix (U21) and cardia using the retroflex view (U22).
Lesser curvature – Region Lc
Examination of this region includes the evaluation in retroflexion view of its upper third (Lc23), middle third (Lc24), lower third (Lc25) and angulus (Lc 26). Known to be a high risk region for early cancer, the lesser curvature is a very frequent location for superficial slightly elevated -IIa and slightly depressed -IIc cancers (20-22). Noteworthy, SACE examines not by chance but targeting early cancers detection, the middle and the lower thirds of the lesser curvature twice by both using the direct (L12, M16) and retroflex views (Lc24, Lc25).
Duodenum – Region D
Duodenal examination includes evaluation of the duodenal bulb (D27) and second duodenal portion (D28) in which the Vater ampoule is easily visible in the left superior quadrant.
SACE PROCEDURE
In brief, both piriform sinuses and the posterior wall of the hypopharynx are evaluated first. After passing the superior esophageal sphincter (the "no-swallowing" technique allows observation of the entire upper third of the esophagus), the endoscope is advanced distally in the natural esophageal axis (the anterior wall at 9-12 oclock) to the esophageal hiatus. A slight turn to the left allows easy entrance into the gastric lumen. After reaching the pyloric ring, the endoscope is gently pulled proximally and detailed observation of the gastric mucosa is performed by rotating the endoscope clock-wise while examining and photographing the gastric surface from the gastric antrum towards the middle and lower thirds using generous air insufflation. After reaching the union of the upper and middle bodies level with the esophageal hiatus, the endoscope is inverted and carefully reinserted proximally to evaluate the upper body and gastric fundus. The procedure continues using the retroflex view in examining the cardia, lesser curvature and all of its extension. Following careful evaluation of the gastric angulus, the endoscope is advanced through the pylorus to evaluate the duodenal bulb and the duodenum up to the second portion (7).
CLINICAL USEFULNESS OF SACE
In a recent Colombian screening study for GC, SACE successfully diagnosed early cancers at a frequency of 0.30% (2/650) in healthy, average-risk male and female volunteer subjects 40-70 years of age (7). Similar results had been achieved in an earlier endoscopic screening study in China that identified 743 (0.37%) early GCs in a high-risk male cohort of 199,000 subjects (23). The clinical benefits of SACE include establishing a standardized global reporting system, providing a more comprehensive endoscopic understanding of the anatomy of the upper GI tract, facilitating the precise location of a condition or early GC lesion so another examiner can easily locate it, designing or planning the most effective method for endoscopic removal and to control bleeding and for providing efficient endoscopic follow-up even when an examination is performed by a different examiner. It is estimated that SACE including biopsy samples if necessary can be completed within three minutes by a highly experienced endoscopist. Based on our observations following a hands-on training course over two full days, SACE can generally be performed within six minutes satisfactorily.
Early diagnosis of GC can have a positive impact in many ways including lower costs for less invasive treatment than extensive radical surgery as well as reduced use of mechanical sutures, procedure times, hospitalization stays, medical consultations, laboratory tests, complementary therapies, lost time from work, adverse effects on quality of life and negative psychological consequences.
CLEANNESS OF THE UPPER GI MUCOSA
Before endoscopy a cleaning solution to dissolve the overlying superficial mucus and for saliva bubbles removal is essential to assure optimal visualization of the mucosal surface avoiding the need of additional washing and suctioning. We do recommend a combination of mucolytic agent (e.g., Pronase), and de-foaming agent (e.g., Polydimethylpolyxiloxane-PDMS) administrated 15 minutes before SACE examination (7,24,25).
CHROMOENDOSCOPY
Once a suspicious lesions is detected using white light, indigo carmine staining has been useful to precisely determine the margin of a superficial lesion and to facilitate the depth estimation of early cancers (26). Although images of indigo carmine stained regions were not included in the current SACE protocol, we routinely use and recommend chromoendoscopy with indigo carmine 0.25% (Chromoendoscopia, Bogota DC, Colombia) for lesion characterization when either a suspicious lesion or cancer is detected or when examining a high risk gastric cancer patient once the protocol of completeness has been achieved (27). Images using electronic enhancement technologies (e.g., NBI, FICE) were not included in the current protocol. Noteworthy, the current SACE protocol is not only independent of any particular image-enhanced technology nor restricted to any endoscopic equipment but also designed as an universal platform from where different image-enhanced technologies could be applied for optimal lesion characterization.
CONCLUSIONS AND FUTURE DIRECTION
In certain respects, the way standard EGD is currently performed impedes rather than promotes early detection of GC and has several limitations to facilitate detection of the disease in an early stage. In contrast, SACE encompasses a completeness protocol that for the first time in medical endoscopy provides a systematic step by step examination protocol and a novel alphanumeric-coded nomenclature for regions and areas allowing precise description and anatomical location of any portion of the upper GI tract during upper endoscopy. Despite the current lack of a consensus
and relevant guidelines, we believe SACE constitutes an appropriate protocol for thorough examination of the upper GI tract and formal training under the direction of qualified experts should ideally become an important part of GI endoscopy curricula. Finally, SACE could help to improve patient care by reducing the risk of missed lesions, provide higher quality endoscopy and increase cost-effectiveness of the procedure by providing a complete examination. Such a novel approach, if proven and implemented on a widespread basis worldwide, could significantly change the way we practice upper endoscopy in our lifetimes.
REFERENCES
1. Ferlay J, Bray F, Parkin DM, Pisani P, eds. Gobocan 2000: Cancer Incidence and Mortality Worldwide. IARC Cancer Bases No. 5. Lyon: IARC Press; 2001. 2. Parkin DM. Epidemiology of cancer: global patterns and trends. Toxicol Lett. 1998;102-103:227-34. 3. Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12(3):354-62. 4. Guggenheim DE, Shah MA. Gastric cancer epidemiology and risk factors. J Surg Oncol. 2013;107(3) 230-6. 5. Piñeros M, Ferlay J, Murillo R. Cancer incidence estimates at the national and district levels in Colombia. Salud Publica Mex. 2006;48(6):455-65. 6. Yang L. Incidence and mortality of gastric cancer in China. World J Gastroenterol. 2006;12(1):17-20. 7. Emura F, Mejía J, Mejía M, Osorio C, Hernández C, González I, et al. Utilidad de la cromoendoscopia sistemática en el diagnóstico del cáncer temprano y lesiones gástricas premalignas. Resultado de dos campañas masivas consecutivas de tamización en Colombia (2006-2007). Rev Col Gastroenterol 2010;25(1):19-30. 8. Emura F, Oda I. Diagnóstico y tratamiento endoscópico del cáncer gástrico estado 0. ¿Qué hacer para que aumente más? [Editorial]. Rev Col Gastroenterol. 2009;24(4):333-5. 9. Hosokawa O, Miyanaga T, Kaizaki Y, Hattori M, Dohden K, Ohta K, et al. Decreased death from gastric cancer by endoscopic screening: association with a population-based cancer registry. Scand J Gastroenterol 2008;43(9):1112-5. 10. Longo WE, Zucker KA, Zdon MJ, Modlin IM. Detection of early gastric cancer in an aggressive endoscopic unit. Am Surg. 1989;55(2):100-4. 11. Brenner H, Rothenbacher D, Arndt V. Epidemiology of stomach cancer. Methods Mol Biol. 2009;472:467-77. 12. Cohen J, Safdi MA, Deal SE, Baron TH, Chak A, Hoffman B, et al. Quality indicators for esophagogastroduodenoscopy. Am J Gastroenterol. 2006;101(4):886-91. 13. The role of endoscopy in the surveillance of premalignant conditions of the upper gastrointestinal tract. American Society for Gastrointestinal Endoscopy. Gastrointest Endosc. 1998;48(6):663-8. 14. Rey JF, Lambert R; ESGE Quality Assurance Committee. ESGE recommendations for quality control in gastrointestinal endoscopy: guidelines for image documentation in upper and lower GI endoscopy. Endoscopy. 2001;33(10):901-3. 15. Asfeldt AM, Straume B, Paulssen EJ. Impact of observer variability on the usefulness of endoscopic images for the documentation of upper gastrointestinal endoscopy. Scand J Gastroenterol. 2007;42(9):1106-12. 16. Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14(2):101-12. 17. Emura F, Sakai P, Quintero I, Sobrino S, Navarrete C, Villa-Gómez G, et al. Adendum: CromoEndoscopia Sistemática (CES). In: Saenz R (ed.). Guías para mejorar la calidad de la endoscopia digestiva. Santiago de Chile; 2010. p. 82-84. 18. Emura F, Santacoloma M, Oda I. Diagnóstico y tratamiento del cáncer gástrico temprano. En: Gil F, Emura F, Santacoloma M (ed). Temas escogidos en endoscopia digestiva. 1 ed. Bogotá: Sociedad Colombiana de Endoscopia Digestiva; 2010. p. 45-60. 19. Emura F, Baron T, Gralnek I. The pharynx: examination of an area too often ignored during upper endoscopy. Gastrointest Endosc. 2013. In press. 20. Oda I, Gotoda T, H Hamanaka H, Eguchi T, Saito Y, Matsuda T, et al. Endoscopic submucosal resection for early gastric cancer: technical feasibility, operation time and complications from a large consecutive series. Digest Endosc. 2005;17(1):54-7. 21. You WC, Blot WJ, Chang YS, Li JY, Jin M, Zhao YX, et al. Comparison of the anatomic distribution of stomach cancer and precancerous gastric lesions. Jpn. J. Cancer Res. 1992;83(11):1150-3. 22. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003;58(6 Suppl):S3-43. 23. Dan YY, So JB, Yeoh KG. Endoscopic screening for gastric cancer. Clin Gastroenterol Hepatol. 2006;4(6):709-16. 24. Fujii T, Iishi H, Tatsuta M, Hirasawa R, Uedo N, Hifumi K, et al. Effectiveness of premedication with pronase for improving visibility during gastroendoscopy: a randomized controlled trial. Gastrointest Endosc. 1998;47(5):382-7. 25. Banerjee B, Parker J, Waits W, Davis B. Effectiveness of preprocedure simethicone drink in improving visibility during esophagogastroduodenoscopy: a double-blind, randomized study. J Clin Gastroenterol. 1992;15(3):264-5. 26. Kida M, Kobayashi K, Saigenji K. Routine chromoendoscopy for gastrointestinal diseases: indications revised. Endoscopy. 2003;35(7):590-6. 27. Emura F, Waxman I. Manejo endoscópico de las lesiones precursoras y la neoplasia temprana en estómago. En: Castaño R (ed.). Endoscopia Oncológica: diagnóstica y terapéutica. Bogotá: Universidad de Antioquia; 2011. p. 55-63.
Correspondence:
Fabian Emura
E-mail: fabian@emuracenter.org
Citar como: Emura F, Gralnek I, Sano Y, Baron TH.
Improving early detection of gastric cancer: a novel systematic alphanumeric-coded endoscopic approach. Rev Gatroenterol Peru. 2013;33(1):52-8.Recibido: 07/02/2013;













