Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de Gastroenterología del Perú
Print version ISSN 1022-5129
Rev. gastroenterol. Perú vol.34 no.3 Lima July 2014
Artículos Originales
Systematic review and meta-analysis of endoscopic ablative treatment of Barrett’s esophagus
Revisión sistemática y meta-análisis del tratamiento ablativo endoscópico del esófago de Barrett
Thiago Ferreira De Souza 1,a, Everson Luiz De Almeida Artifon 1,a, Luiz Henrique Mazzonetto Mestieri 1,a, Sílvia Mansur Reimão 1,b, Felipe Toyama Aires 1,b, Wanderley Marques Bernardo 1,a, Jose Pinhata Otoch 1,a, Eduardo Guimarães Hourneaux De Moura 1,a
1 Gastrointestinal Endoscopy Unit, Departmentof Gastroenterology, University of São Paulo. São Paulo, Brasil.
a Assistants and Associates Professors; b Residents of GI Endoscopy
ABSTRACT
Background: Barrett’s esophagus (BE) is the main risk factor for esophageal adenocarcinoma. Its therapeutic approach is controversial and surgical treatment in the presence of high-grade intraepithelial neoplasia may be indicated. Endoscopic approach is an alternative with lower mortality and morbidity rates and favorable results. Objective: To define the best option, according to literature, to treat Barrett’s Esophagus. Materials and methods: Design: Systematic review of PUBMED, EMBASE, LILACS, and Cochrane Library databases was conducted and articles of randomized, controlled studies on BE endoscopic ablative treatment were selected. The systematic review through PUBMED retrieved results with higher evidence level and available recommendation grade regarding BE ablative therapy. Nine articles on randomized, controlled studies classified as A or B according to the Oxford table were selected. Cryotherapy, laser, photodynamic therapy (PDT), multipolar electrocoagulation (MPEC), and ablation through argon plasma coagulation (APC) and radiofrequency were considered ablation therapies. Patients: 649 patients from 10 different studies were analysed. Results: PDT was found to present an increase in treatment failure compared with APC, NNH = -7. BE ablation through MPEC or APC was found to have similar risk for treatment failure in meta-analysis. PDT associated with proton pump inhibitor (PPI) is beneficial for BE ablation regarding PPI use alone, NNT = 2. Radiofrequency with PPI is an efficient method to reduce risk of treatment failure, NNT = 1. Conclusions: There are no studies demonstrating the benefit of indicating cryotherapy or laser therapy for BE endoscopic approach. APC ablation was found to have superior efficacy compared with PDT and ablation through APC and MPEC was found to present effective, similar results. Radiofrequency is the most recent approach requiring comparative studies for indication.
Key words: Barrett esophagus; Ablation techniques; Argon plasma coagulation; Meta-analysis; Review (source: MeSH NLM).
RESUMEN
Introducción: El esófago e Barrett (BE) es un factor de riesgo importante para adenocarcinoma de esófago.Su manejo terapéutico es controversial y el tratamiento quirúrgico en la presencia de neoplasia intraepitelial de alto grado puede estar indicado. El manejo endoscópico es una alternative con menores tasas de morbilidad y mortalidad y con resultados favorables. Objetivo: Definir la major opción de tratamiento del esófago de Barrett de acuerdo a la literatura. Materiales y métodos: Diseño: Se realize una revisión sistemática de PUBMED, EMBASE, LILACS yla librería Cochrane y los artículos randomizados, controlados en ablación endoscópica de BE fueron seleccionados. Esta revisión de PUBMED mostró resultados de una evidencia muy alta y recomendación alta para el uso de terapia ablativa. Nueve artículos de studios randomizados y controlados fueron catalogados grado A o B de aacuerdo a la table de Oxford y fueron seleccionados.Fueron consideradas como terapia ablativa, la crioterapia, laser,terapia fotodinámica (PDT),electrocoagulación multipolar (MPEC) ,ablación con coagulación por argón plasma (APC) y radiofrecuencia. Pacientes: 649 pacientes de 10 estudios diferentes fueron analizados. Resultados: El PDT se halló que tenía mas fallas que el APC , NNH=-7. La ablación del esófago de barrett por MPEC o APC tuvieron el mismo riesgo de fracaso terapéutico en los meta-análisis.La PDT asociado al uso de Inhibidores de bomba de protones (PPI) es beneficiosa versus el uso de, los PPI solos, NNT=2. La radiofrecuencia con PPI es un método eficiente para reducer el riesgo de fracas terapéutico, NNT=1. Conclusiones: No hay studios que demuestren el beneficio de la crioterapia o la terapia con laser para el esófago de Barrett, se encontró que tiene una eficacia superior comparada con el PDT y la ablación por APC y MPEC tenían resultados efectivos y similares.La radiofrecuencia es el manejo más reciente y requiere estudios comparativos para su indicación.
Palabras clave: Esófago de Barrett; Técnicas de ablación; Coagulación con plasma de argón; Metanálisis; Revisión (fuente: DeCS BIREME).
INTRODUCTION
Barrett’s esophagus (BE) is an acquired condition resulting from chronic aggression of esophageal squamous epithelium by gastroesophageal reflux, so that stratified epithelium is replaced by columnar epithelium with intestinalized cells, which are endoscopically identified and histologically confirmed, regardless of the extension involved (1-3) .
BE is the main risk factor for esophageal adenocarcinoma development and its stages of progression from low-grade to high-grade intraepithelial neoplasia and to adenocarcinoma are well established.
Risk of adenocarcinoma development in BE patients without dysplasia is 2%. In the presence of lowgrade intraepithelial neoplasia, this risk increases to 7% and high-grade intraepithelial dysplasia, to 22%. However, less than 5% of patients with esophageal adenocarcinoma had knowledge of BE and more than 40% did not present any gastroesophageal reflux disease (GERD) symptoms (4) .
BE patients periodically undergo endoscopic examinations to detect early dysplastic changes. Treatments currently accepted include clinical treatment with proton pump inhibitor (PPI) and fundoplication. Despite the efficacy of both treatments, these patients did not present metaplastic epithelium regression and adenocarcinoma risk reduction.
Therefore, a systematic literature review and metaanalysis aiming to establish a protocol for endoscopic ablation therapy is relevant for BE patients and this may postpone or prevent more aggressive treatments with higher complication risks.
MATERIAL AND METHODS
Study design
BE treatment aggregates different clinical, surgical and/or endoscopic procedures. The objective of this study is to identify randomized, controlled studies comparing various modalities of endoscopic therapy for BE or endoscopic ablation treatment vs. PPI in order to conduct an isolated analysis and meta-analysis. Ablation through cryotherapy, laser therapy, photodynamic therapy (PDT), multipolar electrocoagulation (MPEC), argon plasma coagulation (APC), and radiofrequency are considered endoscopic ablation interventions.
Literature search
Studies selected necessarily met the following criteria: patient selection based on results of confirmed and reviewed histopathologic analysis; controlled, randomized studies; one or more primary outcomes, such as BE histologic or endoscopic regression and reduction of progression risk for adenocarcinoma, but compulsorily reporting BE ablation with absolute data.
Systematic review was based on the search for publications in PUBMED, EMBASE, LILACS, and Cochrane Library databases. Restrictions, except for those linguistic and human, are circumscribed by Clinical Queries (CQ) in therapy review with narrow filter for randomized clinical trials. The terms Barrett Esophagus AND Cryotherapy, Barrett Esophagus AND Lasers OR Laser Therapy, Barrett Esophagus AND Photochemotherapy, Barrett Esophagus AND Diathermy OR Electrocoagulation OR Microwaves OR Ultrasonics, Barrett Esophagus AND Laser Coagulation, and Barrett Esophagus AND Catheter Ablation were used in PUBMED-MeSH.
Initial article selection was based on title. In case of doubt about content, evaluation relied on its abstract. All articles selected through their abstracts had their complete texts analyzed. Selected manuscripts were classified according to the Oxford table and JADAD. Randomized, controlled studies with evidence level 2A or 2B were considered for meta-analysis. References from selected studies were analyzed to verify other articles, but no additional publications were selected.
Studies were grouped according to ablation therapy methods: PDT vs. APC ablation (group 1), MPEC vs. APC ablation (group 2), and PDT ablation vs. PPI (group 3). For such studies critical analysis and meta-analysis were conducted. APC ablation vs. PPI and radiofrequency ablation vs. PPI were found to produce one article only and individual results were presented. All intervention and control groups received PPI.
Data extraction
Data were extracted by a single observer. Data regarding histological response and, in its absence, impression upon endoscopic examination were considered as basis to formulate tables and metaanalysis for all studies. Total number of randomized and assigned patients to each original group and losses of each group were considered events in intention-to-treat (ITT) analysis. Side effects considered were the most relevant and coincident in studies. Regarding followup period, the 4- to 12-month period was considered using the longest follow-up with loss smaller than 20%.
Statistical analysis
Meta-analysis was performed through Review Manager software (RevMan, version 5). The method of absolute risk difference (Mantel-Haenszel test) was used with confidence interval (CI) at 95%. Inconsistencies across clinical trials were estimated through chi-square test (Chi 2 ) of heterogeneity and quantified using I 2 test. Values over 50% were considered substantial. Studies yielded considerable heterogeneity were represented through funnel plots. CATmaker statistical software (Centre for Evidence-Based Medicine, Oxford, UK) about therapy was used to present results from isolated studies and absolute risk, number needed to treat (NNT) and number needed to harm (NNH) were considered.
RESULTS
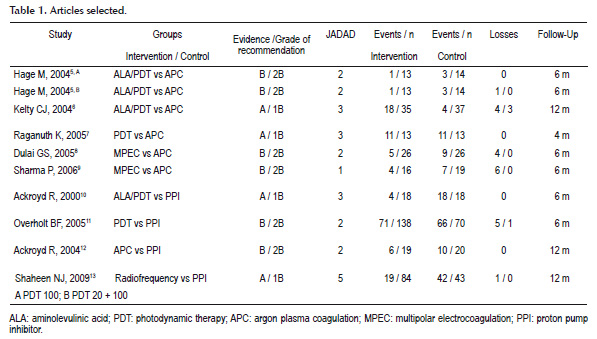
Performed search strategies yielded the following results of randomized, controlled studies. Table 1.
PDT vs. APC ablation
Three randomized, controlled studies evaluating endoscopic ablation therapies with PDT and APC were selected. Treatment with photodynamic therapy using aminolevulinic acid (ALA-PDT) compared with APC were found to result in similar number of treatment failure occurrences after ablation (no statistical difference) by Hage et al. (5) and Raganuth et al (6) . BE ablation through (PDT) intervention is reported to increase treatment failure risk compared with control (APC) by Kelty et al. (7) , absolute risk increase (ARI) = -0.41 and NNH = -2. Meta-analysis therefore results from increased treatment failure risk with ablation through PDT compared with APC, ARI = -0.14 (-0.02 to -0.27) and NNH = -7. Heterogenicity is 82% and results were influenced by Kelty et al 6 study presenting weight of 47.4%. Data extracted regarding these studies are presented in Figure 1.
MPEC vs. APC ablation
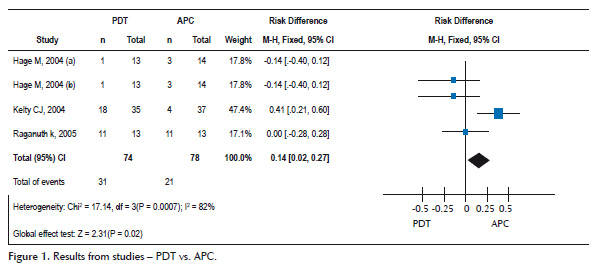
Two randomized, controlled studies evaluating endoscopic ablation therapies with APC and MPEC were selected. Treatment using MPEC compared with APC was found to result in similar number of treatment failure occurrences after ablation (no statistical difference) by Dulai et al. (8) and Sharma et al. (9) and the same is verified in meta-analysis. Data extracted regarding these studies are shown in Figure 2.
PDT ablation vs. PPI
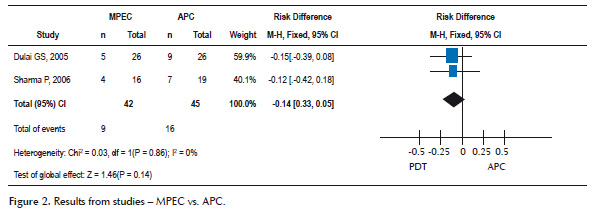
Three randomized, controlled studies evaluating endoscopic ablation therapies with PDT vs. PPI were selected. Two of them, Ackroyd et al (10) and Overholt et al. (11) , were considered for meta-analysis and Overholt et al. (12) was excluded for presenting the 5-year followup from Overholt et al. (11) study. Treatment with PDT compared with PPI alone was found to reduce treatment failure risk (10,11) absolute risk reduction (ARR) = 0.49 (0.58 to 0.39), p< 0.05 and NNT= 2. Meta-analysis heterogenicity is 89% and one study (11) weighted 83.8%. Extracted data regarding previously mentioned studies are presented in Figure 3.
APC ablation vs. PPI
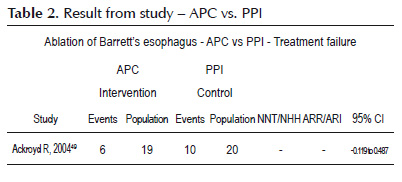
The randomized, controlled study conducted by Ackroyd et al (13) in patients with BE and antireflux barrier evaluated BE endoscopic treatment vs. PPI. In a 1-year follow-up, BE was found in 6 of 19 patients in intervention and in 10 of 20 in control group, with no significant difference, p>0.05 (Table 2). No severe complications were verified in APC-treated patients.
Radiofrequency ablation vs. PPI
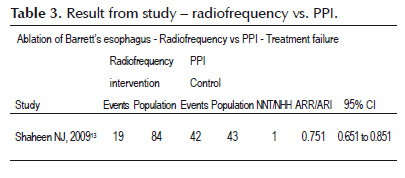
A randomized, controlled study conducted by Shaheen NJ et al. (14) in patients with BE and dysplasia evaluated BE endoscopic treatment using radiofrequency vs. PPI. ITT analysis considered 127 randomized patients and demonstrated 77.4% of all patients that underwent ablation by HALO system, after 12 months, were not found to present intestinal metaplasia compared with PPI (2.3%), demonstrating risk reduction (ARR = 0.751), p<0.001 and NNT= 1 (Table 3).
DISCUSSION
BE presence and possibility of its progression to low and high-grade intraepithelial neoplasia are determining factors of the need for regular endoscopic follow-up and biopsy surveillance due to adenocarcinoma risk (15) .
The relatively long evolution period from BE to adenocarcinoma allows specific treatments to be performed preventing malignant neoplasia outset (16) . Accordingly, the need to determine the best treatment form and approach to these patients arrises, so that ideal treatment is the safest and most effective, which is successful in the short and long term at low cost and available in most hospital units with short learning curve.
Ablation therapies face two major issues considerably important for involving adenocarcinoma possibility. Ablation does not provide material for histopathologic examination and, during reepithelization process, columnar epithelium site may be lined by regenerated epithelium turning adequate endoscopic evaluation and biopsy fragment collection more difficult.
These patients must be carefully followed up through multiple biopsies and guidance to the pathologist regarding performed intervention type. Presence of small islands and glands under squamous epithelium occur and present possibility of progression dysplasiacancer. The importance of this context resides in the fact that patients treated with ablation therapies may present normal upper GI endoscopy (UGE) and biopsies positive for BE, dysplasia or cancer (16) .
The natural history of high-grade intraepithelial neoplasias is still under discussion, making difficult and diverging conducts, and enables endoscopic follow-up with a series of biopsies or resection and/ or endoscopic ablation. Most patients with high-grade intraepithelial neoplasia were found not to progress to adenocarcinoma in some follow-up years by Flejou et al (17) . Therefore surgical procedure becomes a second option and endoscopic therapy or follow-up with a series of endoscopic examinations may happen (17) .
Two published meta-analyses (18,19) regarding BE treatment covering clinical treatment, surgical and endoscopic intervention were evaluated. Li et al. (18) used the same studies selected in this meta-analysis, except for the inclusion of three studies: Hage et al. (20) , which discussed molecular changes after BE ablation therapy and, hence, not compatible with outcomes considered for this meta-analysis; whereas Overholt et al. (14) and Bright et al. (21) present a 5-year follow-up in two studies included herein. Radiofrequency ablation was not included in the meta-analysis conducted by Li et al. (18) , which concludes that pharmacological therapy, antireflux surgery and endoscopic ablation therapies are promising alternatives for BE treatment, although studies are not currently sufficient to determine the benefit of prevention and reduction of adenocarcinoma occurrence in the long term (18).
Rees et al. (19) conducted a meta-analysis and considered the study by Luman et al (22) about BE treatment using laser with only eight patients, four in control and four in intervention group, restricting the possibility of conclusion. In a non-randomized study, complete regression occurred in 40% of patients (23) . A study conducted by Mackenzie et al (24) compared the same intervention with different substances, 5-ALA and porfimer sodium. Both studies were not included in this meta-analysis but used in discussion. Ablation therapies were found to present indications increasingly for managing patients with BE and dysplasia and currently indicate major benefits of radiofrequency relative to PDT. Nonetheless, long-term studies are required before its large-scale application to determine adenocarcinoma risk reduction (19) .
Isolated evaluation of three randomized, controlled studies using APC vs. PDT ablation therapies diverged regarding which would be the method of choice.
In a study conducted by Hage et al (5) and Raganuth et al. (7) , treatment with PDT compared with APC presented similar figures for treatment failure and no statistical difference. However, an increased risk of PDT use was reported by Kelty et al 6 implying sufficient influence in meta-analysis due to its heterogenicity (82%) and weight of this study (47.4%). In metaanalysis of three studies presented, total number for intervention group of 74 patients with 31 events and for control group of 78 patients with 21 events were obtained. Results demonstrated that although both ablation forms are effective, intervention presents increased risk of treatment failure, ARI = -0.14 (-0.02 to -0.27), NNH = -7 in 4- to 12-month follow-up.
Ablation with PDT requires less sessions when compared with APC, two and four sessions, respectively, in isolated studies. It is important to emphasize that APC use is less costly, easy to perform and widely available.
In variable follow-up, intestinalized gland presence in regenerated epithelium is lower in intervention group (PDT, 12%) relative to APC (19%). Favorable results for (PDT) intervention were verified by Raganuth et al 7 considering the outcome of dysplasia ablation with 77% vs. 67% resolution within 12 months. The clear mechanism is not established, but photosensitive substance is likely to accumulate in tissues with higher cellular activity, such as metaplastic epithelium.
Use of ablation through MPEC associated with PPI apparently promotes ablation and columnar reepithelization in most case series reported. In some patients, reepithelization does not occur completely and some islands of metaplastic columnar epithelium on the surface or beneath squamous epithelium may exist. Two studies with evidence level and recommendation grade B and 2B, respectively, evaluated BE endoscopic treatment with MPEC and APC. Results demonstrating no difference between MPEC and APC, p>0.05, were presented by Dulai et al (8) . The number of sessions necessary for ablation was lower in intervention group (mean 2.8) compared with control (mean 3.8) and no statistical difference. Similar results are reported by study conducted by Sharma et al (9) and no difference between intervention regarding control. Endoscopic treatment of both groups was performed in similar intervals (4-8 weeks) and 6 sessions at most. Meta-analysis of studies result in total number of 42 for intervention group with 9 events and 5 for control group with 16 events. Data obtained in meta-analysis reflect individual results from studies with similar number of post-ablation treatment failure, p>0.05 and null heterogenicity.
PDT vs. PPI ablation was evaluated through two randomized, controlled studies considered for metaanalysis. Although these studies represent different population sets (low- and high-grade dysplasia) and distinct intervention mediators (5-ALA and porfimer sodium), data for BE complete ablation and type of ablation therapy used (PDT) were considered. Endoscopic treatment with ALA-PDT and green light associated with PPI vs. PPI in patients with BE and low-grade intraepithelial neoplasia was evaluated by Ackroyd et al (10) . This study demonstrates the benefit of this intervention under control reducing treatment failure risk, ARR= 0.778, NNT= 1 and p<0.05. Ablation therapy benefits with PDT and porfimer sodium with red light associated with omeprazole vs. PPI was demonstrated by Overholt et al. (11) ARR= 0.429, NNT= 2 and p<0.05. In meta-analysis with total number of 156 patients in intervention group and 75 events, whereas in PPI group, 88 and 84, respectively, the intervention clearly presents reduction of treatment failure risk regarding PPI use alone in a 6-month follow-up, ARR= 0.49 (0.58-0.39), p<0.05 and NNT= 2. Meta-analysis heterogenicity is 98% in a study (11) weighted 83.8%. In a 5-year follow-up, 77% (106/138) of patients treated with PDT did not present any high-grade dysplasia vs. 39% (27/70) of PPI group, p<0.0001. Considering secondary outcome, evolution to cancer, the intervention was found to be effective, p<0.027 (14) .
Endoscopic treatment with APC vs. PPI was compared in a single study and does not allow metaanalysis. BE patients with antireflux barrier underwent endoscopic treatment with APC vs. PPI were evaluated (12) In a 1-year follow-up, BE was detected in 17 of 20 patients on endoscopic examination in control and partial regression occurred in 11 cases with BE extension increase in two patients. In APC-treated patients, BE was detected on endoscopic examination in 8 of 19 patients with significant difference between both groups and reduction in treatment failure risk (ARR = 0.429, 0.157 to 0.701) and NNT = 2. In those treated and without BE after one year, 10 of 11 did not present BE in 1-month follow-up after reccurrence within 12 months, GERD clinical manifestations were present and on UGE loss of fundoplication and small islands of columnar epithelium were detected.
On histopathologic examination, considered gold standard for BE diagnosis, 10 of 20 patients in control group did not present response to clinical treatment whereas six of 19 patients did not present BE despite disagreement with UGE examination in two cases. A chi 2 test was performed and no statistical difference demonstrating reduction or increase in treatment failure risk was found between groups. No BE case after 12 months was diagnosed with dysplasia or cancer in APCtreated group and partial response after one month from last session. Presence of subepithelial columnar glands occurred in 35% of patients after one month post-ablation therapy and in 5% (one patient) in the 12-month follow-up. Mean number of interventions necessary for ablation with APC was 3 (2-4 sessions). Surgical procedures associated with clinical treatment to reduce BE or dysplasia progression did not yield expressive results. In a 5-year follow-up, most patients did not present BE and dysplasia development occurred in two control patients (21) .
Radiofrequency ablation was evaluated in a randomized, controlled study conducted by Shaheen et al (13) aiming to assess efficacy and safety of radiofrequency use vs. PPI for eradicating dysplasia and intestinal metaplasia.
ITT analysis considering 127 randomized patients demonstrates that 77.4% of all patients that underwent ablation with HALO system (intervention), after 12 months, did not present intestinal metaplasia compared with PPI group (2.3%) with treatment failure reduction, ARR= 0.751 (0.651 to 0.851), p<0.001 and NNT= 1. Complete eradication of low- and high-grade dysplasia was also observed (90.5 and 81%) with radiofrequency relevant role in reducing cancer incidence regarding control (1 of 84 from intervention and 4 of 43 from control group). Three patients presented severe complications and needed to be admitted to hospital. Complications included hemorrhage in a patient with heart disease, chest pain immediately after procedure and chest pain with nausea eight days after procedure. All patients were clinically treated or through UGE and none required surgical intervention.
HALO system use presents efficacy and safety for ablation treatment, however long-term studies and follow-up are necessary, namely randomized, controlled studies with remaining existing methods to determine the real benefit of this new technology.
Systematic review, meta-analysis and evaluation of isolated studies present limitations. Most studies, although comprising better recommendation grades, strong evidence and JADAD currently available in the literature, may be partially considered high quality (recommendation grade A and JADAD ≥3). In most studies, endoscopists and patients cannot be unaware of the intervention or its absence and this might lead to subjective assessments, partially corrected by histologic analysis, in which prior knowledge from asigned groups is not required by the observer. Studies in the same group present variations in population, primary and secondary outcomes, technical differences (substance, dose, number of interventions, and performance), as well as follow-up leading to heterogenicity in the sample.
From the evaluation of various treatment modalities and comparison between each other it is possible to realize that ablative therapies, whenever indicated, must be performed to the detriment of isolated clinical treatment. The choice between various interventions requires caution because for most of them results are similar and long-term follow-up (five years) are scarce, but favorable to the intervention and adenocarcinoma decreased risk. Among therapeutic options, PDT is verified to present expressive results in relation to PPI use alone and efficient in BE ablation, although it is not superior to APC ablation in meta-analysis. Nonetheless, APC ablation was not found to present statistical difference regarding PPI isolated use in histologic evaluation, although there was statistical significance in endoscopic evaluation, but superior to PDT. MPEC Ablation is similar to APC ablation. Radiofrequency ablation compared with PPI isolated use, in one study, was found to be effective (p<0.01). From this set of information assessment, in isolated articles or metaanalysis, BE ablative therapies (PDT, MPEC, APC, and radiofrequency) are suggested to be potentially performed in a safe manner and results from BE ablation, intraepithelial neoplasia and adenocarcinoma risk to be expressive, despite scarce validation in the long term. The choice among techniques must take into consideration knowledge and acceptance by patients, method availability, ability to its performance, cost, follow-up possibility, and reintervention in the long term. The benefit of method combination remains to be established. Cryotherapy and laser must not be used, except for research protocol purposes.
Decision to indicate an ablative therapy for BE patients is difficult. Various studies indicate its long-term benefit, but the future of these patients is uncertain. Possibility of reccurrence, existence of columnar epithelium under regenerated epithelium and incomplete ablation are to be considered. Follow-up only, apparently insufficient or comfortable, may be a safe alternative. Not indicating ablative therapies may be safe, but ablation techniques are also safe. Currently, treatment of BE patients without dysplasia or with low-grade dysplasia, both intervention and follow-up are correct and, in case intervention is chosen, APC or MPEC is recommended based on this meta-analysis. In BE patients with highgrade dysplasia, intervention is indicated due to high risk of adenocarcinoma progression or its coexistence. Choice between endoscopic therapies of surgical resection must consider first and foremost the opinion of informed patients even supported by evidencebased medicine, on team confidence in respecting their desire to conduct their lives as decided and risks they are willing to take.
More randomized, controlled studies with follow-up of at least five years considering as endpoint evolution to adenocarcinoma are necessary to allow determination of efficacy, safety and benefits of different modalities of ablation therapies.
Conflict of interest: none.
REFERENCES
1. Dent J. Barrett’s esophagus: A historical perspective, an update on core practicalities and predictions on future evolutions of management . J Gastroenterol Hepatol. 2011 Jan;26 Suppl 1:11-30. doi: 10.1111/j.1440-1746.2010.06535.x.
2. Morales CP, Spechler SJ. Intestinal metaplasia at the gastroesophageal junction? Barrett’s, bacteria, and biomakers . Am J Gastroenterol. 2003 Apr;98(4):759-62.
3. Sampliner RE. Practice guidelines on the diagnosis, surveillance, and therapy of Barrett’s esophagus. The Practice Parameters Committee of the American College of Gastroenterology . Am J Gastroenterol. 1998 Jul;93(7):1028-32.
4. A New Horizon: Recommendations and Treatment Guidelines for Barrett’s Esophagus . Gastroenterol Nurs. 2009 MayJun;32(3):211-2. doi: 10.1097/SGA.0b013e3181acf1bd.
5. Hage M, Siersema PD, van Dekken H, Steyerberg EW, Haringsma J, van de Vrie W, et al. 5-aminolevulinic acid photodynamic therapy vs. argon plasma coagulation for ablation of Barrett’s oesophagus: a randomised trial . Gut. 2004 Jun;53(6):785-90.
6. Ragunath K, Krasner N, Raman VS, Haqqani MT, Phillips CJ, Cheung I. Endoscopic ablation of dysplastic Barrett’s oesophagus comparing argon plasma coagulation and photodynamic therapy: a randomized prospective trial assessing efficacy and cost-effectiveness . Scand J Gastroenterol. 2005 Jul;40(7):750-8.
7. Kelty CJ, Ackroyd R, Brown NJ, Stephenson TJ, Stoddard CJ, Reed MW. Endoscopic ablation of Barrett’s oesophagus: a randomized-controlled trial of photodynamic therapy vs. argon plasma coagulation . Aliment Pharmacol Ther. 2004 Dec;20(11-12):1289-96.
8. Dulai GS, Jensen DM, Cortina G, Fontana L, Ippoliti A. Randomized trial of argon plasma coagulation vs. multipolar electrocoagulation for ablation of Barrett’s esophagus. Gastrointest Endosc. 2005 Feb;61(2):232-40.
9 . Sharma P, Wani S, Weston AP, Bansal A, Hall M, Mathur S, et al. A randomised controlled trial of ablation of Barrett’s oesophagus with multipolar electrocoagulation vs. argon plasma coagulation in combination with acid suppression: long term results . Gut. 2006 Sep;55(9):1233-9.
10. Ackroyd R, Brown NJ, Davis MF, Stephenson TJ, Marcus SL, Stoddard CJ, et al. Photodynamic therapy for dysplastic Barrett’s oesophagus: a prospective, double blind, randomised, placebo controlled trial . Gut. 2000 Nov;47(5):612-7.
11. Overholt BF, Lightdale CJ, Wang KK, Canto MI, Burdick S, Haggitt RC, et al. Photodynamic therapy with porfimer sodium for ablation of high-grade dysplasia in Barrett’s esophagus: international, partially blinded, randomized phase III trial . Gastrointest Endosc. 2005 Oct;62(4):488-98.
12. Overholt BF, Wang KK, Burdick JS, Lightdale CJ, Kimmey M, Nava HR, et al. Five-year efficacy and safety of photodynamic therapy with Photofrin in Barrett’s high-grade dysplasia . Gastrointest Endosc. 2007 Sep;66(3):460-8.
13. Ackroyd R, Tam W, Schoeman M, Devitt PG, Watson DI. Prospective randomized controlled trial of argon plasma coagulation ablation vs. endoscopic surveillance of patients with Barrett’s esophagus after antireflux surgery. Gastrointest Endosc. 2004 Jan;59(1):1-7.
14. Shaheen NJ, Sharma P, Overholt BF, Wolfsen HC, Sampliner RE, Wang KK, et al. Radiofrequency ablation in Barrett’s esophagus with dysplasia . N Engl J Med. 2009;360(22):2277-88. doi: 10.1056/NEJMoa0808145.
15. Birkmeyer JD, Siewers AE, Finlayson EVA, Stukel TA, Lucas FL, Batista I, et al. Hospital volume and surgical mortality in the United States . N Engl J Med. 2002;346:1128-37.
16. Barr H, Stone N, Rembacken B. Endoscopic therapy for Barrett’s oesophagus . Gut. 2005 Jun;54(6):875-84.
17. Fléjou JF. Barrett’s oesophagus: from metaplasia to dysplasia and cancer . Gut. 2005 Mar;54 Suppl 1:i6-12.
18. Li YM, Li L, Yu CH, Liu YS, Xu CF. A systematic review and meta analysis of the treatment for Barrett’s esophagus . Dig Dis Sci. 2008 Nov;53(11):2837-46. doi: 10.1007/s10620-008-0257-3.
19. Rees JR, Lao-Sirieix P, Wong A, Fitzgerald RC. Treatment for Barrett’s oesophagus . Cochrane Database Syst Rev. 2010 Jan 20;(1):CD004060. doi: 10.1002/14651858.CD004060.pub2.
20. Hage M, Siersema PD, Vissers KJ, Steyerberg EW, Haringsma J, Kuipers EJ, et al. Molecular evaluation of ablative therapy of Barrett’s oesophagus . J Pathol. 2005 Jan;205(1):57-64.
21. Bright T, Watson DI, Tam W, Game PA, Astill D, Ackroyd R, et al. Randomized trial of argon plasma coagulation vs.endoscopic surveillance for barrett esophagus after antireflux surgery: late results . Ann Surg. 2007 Dec;246(6):1016-20.
22. Luman W, Lessels AM, Palmer KR. Failure of Nd-YAG photocoagulation therapy as treatment for Barrett’s oesophagus--a pilot study . Eur J Gastroenterol Hepatol. 1996 Jul;8(7):627-30.
23. Norberto L, Polese L, Angriman I, Erroi F, Cecchetto A, D´Amico DF. High-energy laser therapy of Barrett´s esophagus: preliminary results . World J Surg. 2004 Apr;28(4):350-4.
24. Mackenzie GD, Dunn JM, Selvasekar CR, Mosse CA, Thorpe SM, Novelli MR, et al. Optimal conditions for successful ablation of high-grade dysplasia in Barrett’s oesophagus using aminolaevulinic acid photodynamic therapy . Lasers Med Sci. 2009 Sep;24(5):729-34. doi: 10.1007/s10103-008-0630-7.
25. Parrilla P, Martínez de Haro LF, Ortiz A, Munitiz V, Molina J, Bermejo J, ET al. Long-term results of a randomized prospective study comparing medical and surgical treatment of Barrett’s esophagus . Ann Surg. 2003 Mar;237(3):291-8.
Correspondencia:
Thiago Ferreira de Souza Av. Dr. Enéas de Carvalho Aguiar, 255 PAMB 6 ° andar, Sala 3 São Paulo, Brasil. 05403-000 E-mail: tsouzahc@gmail.com
Recibido: 26-03-2014
Aprobado: 31-05-2014