Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de Gastroenterología del Perú
versión impresa ISSN 1022-5129
Rev. gastroenterol. Perú vol.35 no.2 Lima abr. 2015
Artículos originales
Correlation between functional gastrointestinal disorders and gastric mucosa histopathology findings, including Helicobacter pylori infection, in Lima, Peru
Correlación entre síntomas gastrointestinales funcionales y la histopatología de la mucosa gástrica, incluyendo la infección por Helicobacter pylori, en Lima, Perú
Katharine McDonald1a, Samuel Shopinski1a, Ariana Wilkinson1a, Claudia Meza2b, Jaime Cok2,3c, Alejandro Bussalleu2,3d, Manuel Valdivieso1e
1 Medical School, University of Michigan. Míchigan, Estados Unidos.
2 Universidad Peruana Cayetano Heredia. Lima, Perú.
3 Hospital Nacional Cayetano Heredia. Lima, Perú.
a Undergraduate students; b Research Nurse; c Pathologist; d Gastroenterologist; e Oncologist, SWOG, Professor of Medicine
ABSTRACT
Objective: This study aims to investigate the relationship between functional gastrointestinal disorders and histopathology characteristics, including H. pylori infection, of gastric mucosa, at Cayetano Heredia National Hospital, Lima-Peru, in 2013. Materials and methods: 112 patients were interviewed prospectively between June and July 2013 in the gastroenterology service. Dyspepsia, irritable bowel syndrome, and postprandial distress syndrome were characterized using the Rome III Survey. Results: Pathology results were determined by gastric biopsies obtained by endoscopy. Of the patients interviewed, biopsy results were obtained for 101. 22.8% had atrophy, 24.8% had intestinal metaplasia, 57.4% presented with H pylori. Conclusions: Using chi-square analysis, no statistically significant relationship could be identified between clinical presentation and biopsy results.
Key words: Functional GI disorders, dyspepsia, irritable bowel syndrome, postprandial distress syndrome, gastric biopsy, Helicobacter pylori.
RESUMEN
Objetivo: El objetivo de este estudio fue investigar la correlación que existe entre la presencia de síntomas gastrointestinales y los hallazgos histopatológicos de la biopsia gástrica incluyendo la presencia de la infección por Helicobacter pylori. Materiales y métodos: Este estudio prospectivo comprendió a 112 pacientes que se incluyeron entre junio y julio de 2013 en el Servicio de Gastroenterología del Hospital Nacional Cayetano Heredia, Lima-Perú a los que se les hizo endoscopía y biopsia gástrica. Los síntomas de dispepsia, síndrome de intestino irritable y síndrome de distrés post prandial fueron obtenidos usando las encuestas de Roma III. Resultados: De los pacientes a los que se les hizo la encuesta sólo en 101 se les evaluó la biopsia. 22,8% tuvo atrofia, 24,8% presentó metaplasia intestinal, y en 57,4% se reportó la infección por Helicobacter pylori. Conclusiones: Usando el análisis con chi-cuadrado no se pudo establecer ninguna correlación estadísticamente significativa entre la presentación clínica y los resultados de las biopsias.
Palabras clave: Trastornos digestivos funcionales, dispepsia, síndrome de intestino irritable, síndrome de distrés post prandial, biopsia gástrica, Helicobacter pylori.
INTRODUCTION
Gastric cancer is the most common cause of cancer mortality in both men and women in Peru. In fact, 94% of gastric cancer cases in Peru are detected at an advanced stage (1). Infection with Helicobacter pylori has been associated with an increased risk for development of gastric cancer (2-4). This association has led the World Health Organization to classify H. pylori as a type I carcinogen (5,6). H. pylori has also been implicated in the development of a progressive series of precancerous lesions and other gastric pathology, including atrophic gastritis, intestinal metaplasia and gastric and duodenal ulcers (7-9). Lasting eradication of H. pylori infection from the gastric mucosa may be able to halt this progression to cancer and serve as a cancer prevention strategy (3 ).
As with all cancers, early detection is the key. If symptoms could be found to direct attention to the possible or likely presence of H. pylori and/or precancerous lesions, clinicians could better monitor patients at risk for developing gastric cancer. While numerous studies have failed to find a significant relationship between gastrointestinal symptoms of dyspepsia and the presence of H. pylori (10-14), others have found a direct association. Perri et al found an association between pain-prevalent dyspepsia and H. pylori infection (15). Vaira et al found that originally asymptomatic H. pylori-positive subjects were more likely to develop symptoms of dyspepsia than non-infected controls; additionally, H. pylori eradication was shown to prevent future development of both dyspepsia and peptic ulcers (16). The aim of our study is to correlate clinical presentations of dyspepsia, postprandial distress syndrome and irritable bowel syndrome with histological findings on biopsy, namely the presence of H. pylori and precancerous lesions.
MATERIALS AND METHODS
Subjects:
112 patients between the ages of 18-70 participated in this study. Eligible participants presented to the gastroenterology department at Cayetano Heredia National Hospital (HNCH) with indications for an endoscopy. Exclusion criteria included: TB infections, HIV/AIDS, and gastrointestinal bleeding. Patients who presented for screening purposes only were excluded.
Data collection:
Patients were interviewed face-to-face using two questionnaires: 1) risk factors for H. pylori infections and 2) Rome III of general GI symptomatology. Surveys were translated to Spanish prior to administration. The following variables were collected: age, gender, endoscopic diagnosis, and histologic findings of the biopsy. Patients were interviewed by non-native Spanish speakers with supervision by HNCH staff.
Endoscopy:
After obtaining signed informed consents, endoscopies were performed by residents and attending physicians in the gastroenterology department at HNCH. At endoscopy, an antral biopsy specimen was obtained using an Olympus GIF XP10, Olympus CF-VL, or Fujinon EPX-201 instrument with video and photo capture. Patients were sedated intravenously with midazolam and/or meperidine. Equipment was sterilized before each endoscopy. Diagnosis was made after visualizing the esophagus, antrum, stomach corpus, and duodenum.
Processing of biopsies:
Specimens obtained by endoscopy were analyzed for histological changes and the presence of H. pylori in the Department of Clinical and Anatomic Pathology at HNCH. Pathologists at HNCH used H&E staining to characterize intestinal metaplasia, dysplasia, atrophy, neutrophil activity, gastritis, and to identify the presence of H. pylori and lymphoid follicles. Chronic gastritis was considered as present if found in either the antrum or body of the stomach. This chronic gastritis was differentiated between superficial and profound based on presence of lymphoplasmocitary infiltrate above or below the glandular necks on the lamina propia. Intestinal metaplasia was considered absent-present on an incomplete or complete manner based on the presence of cells characteristic of the large intestine (incomplete) or small intestine (complete). Atrophy was classified as metaplastic, non metaplastic and mixed. Metaplastic when metaplasia is observed; non metaplastic when gland disappeared and there is not metaplasia. Neutrophil activity was characterized as present if the sample showed extravascular neutrophils.
Analysis:
Microsoft Excel was used to design the database collect information from risk surveys, Rome III surveys, endoscopy reports, and biopsy results. Rome III criteria were used to diagnose functional dyspepsia, irritable bowel syndrome, and postprandial distress syndrome (19,20). Data was transferred to SPSS for chi-square analysis by the University of Michigan Center for Statistical Consultation & Research.
RESULTS
A total of 112 patients were surveyed at the hospital. A total of 11 patients were excluded from our final analysis. One patient had a polyp biopsied in place of other stomach tissue, and the biopsy report did not include the parameters being analyzed. The other ten patients were excluded due to lack of a biopsy report. Demographic information for the excluded patients was available for ten of the eleven. Seven of the excluded patients were women, and the average age of the excluded patients was 46.2 years.
Our final analysis was done on the 101 patients who completed the survey and for whom biopsy reports were obtained. Of this group, 75 (74.3%) were female. The average age was 48.6 years old with a standard deviation of 12.7 years.
Three different clinical conditions could be diagnosed from the Rome III survey results obtained. These were irritable bowel syndrome (IBS), dyspepsia, and postprandial distress syndrome (PDS). Of 101 patients, 58 had dyspepsia (57.4%), 37 had IBS (36.6%), and 12 had PDS (11.9%). It was possible for patients to have more than one diagnosis, and 31 patients (30.7%) did not meet criteria for any of the syndromes studied.
The reported parameters selected to study were intestinal metaplasia, atrophy, and the presence of H. pylori. Of 101 patients, 7 (6.9%) had incomplete intestinal metaplasia, 10 (9.9%) had complete intestinal metaplasia, and 8 (7.9%) had both types, for a total of 25 (24.8%) with any type of intestinal metaplasia. Atrophy was present in 23 (22.8%), and H. pylori was present in 58 (57.4%) of patients.
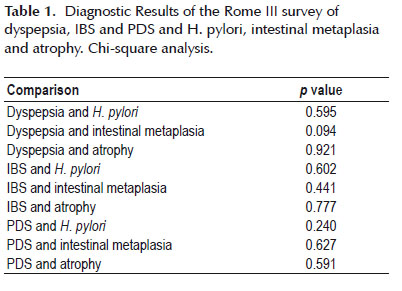
Chi-square analysis was conducted to compare the diagnostic results of the Rome III survey with three parameters reported from the biopsy samples. For each possible diagnosis of IBS, dyspepsia, and PDS, a chi square test was done for the presence of H. pylori, the presence and type of intestinal metaplasia, and the presence of atrophy in the biopsy sample. The p-values for these comparisons are compiled in Table 1.
Most of the p values obtained in chi-square analysis are well above the p <0.05 threshold for statistical significance. The comparison that came closest to yielding a significant result, dyspepsia and intestinal metaplasia, was complicated by the fact that some of the cross-tabulation boxes contained very low values. Attempts to correct this chi-square analysis with a Fisher’s Exact Test in SPSS were unsuccessful.
DISCUSSION
In this study, symptoms from clinical presentation according to the Rome III survey were compared to biopsy results. Previous studies demonstrated that H. pylori infection led to inflammation involved in atrophic gastritis, intestinal metaplasia, gastric and peptic ulcers (7-10). Given this mechanism, we expected clinical presentation to reflect these underlying processes. However, results from the chi-square cross tabulations were all statistically insignificant (Table 1) and did not yield any relationship between dyspepsia and intestinal metaplasia.
Of the clinical syndromes studied, two involve the upper gastrointestinal tract: dyspepsia and PDS. We expected these conditions would be more likely to correlate with H. pylori infection, which can occur only in the stomach. Though a neural connection between the stomach and colon is evidenced by the gastrocolic reflex, the upper and lower portions of the gastrointestinal tract are often distinguished in clinical practice. It was not surprising that a correlation between H. pylori or subsequent histological changes and IBS, thought to predominantly be an effect of colonic hypersensitivity, was not found.
Confounding factors contributing to our lack of significant results may have included verbal administration of the survey instrument, which was intended to be filled out in writing by the patients themselves. Verbal questionnaires may have led to more restricted disclosure about sensitive symptoms.
Another confounding factor could have been the length of H. pylori infection. Previous studies indicate that several decades are required to complete the pathogenetic sequence from H. pylori infection and chronic superficial gastritis to chronic atrophic gastritis and then to gastric cancer (21). Since it is expected that many years should elapse between initial infection and symptoms or histopathological findings of metaplasia or atrophy are manifest, our data may have been insignificant due to the inclusion of patients of all ages between 18 and 70. The younger patients in our cohort may not have been infected for a long enough period of time to develop the findings studied here. The length of H. pylori infection could not have been determined through our method of selecting study participants, who may have been infected since childhood or acquired their infections more recently. Choosing to include only an older subset of participants may have increased the chances that study subjects had longstanding infection for many years and might have yielded more significant findings.
Statistically, examining many categories of intestinal metaplasia decreases the number of subjects in any category and increases the threshold for significance. Using simple yes/no categories for metaplasia may have made the correlation more significant as well.
Numerous studies by other investigators also failed to find a correlation between any presenting symptoms and presence of H. pylori infection (12-18). Gastric cancer is difficult to diagnose in its early stages because it is often asymptomatic (19,20), and it is not surprising that the histological changes theorized to precede cancer similarly lack specific symptoms. Furthermore, our study was gender-biased with 74.3% women. Previous reports of gastric cancer in Lima have shown a greater prevalence of gastric cancer in men (1).
Attempts to find clinical markers for H. pylori infection and gastric cancer have consistently proved unfruitful. More recent attempts to find serum biomarkers for early gastric cancer suggest a more promising route. Assays of microRNA indicate that there may be consistent differences between early gastric cancer and normal controls (21), providing the basis for a future screening test. The benefits of a serological test include that fact that patients would not have to undergo endoscopy, which is time-consuming and requires anesthesia, for surveillance. A simple blood draw may improve surveillance because people would be more willing to undergo this procedure.
Acknowledgements
We are grateful for the support of physicians, residents, nurses, students, and other gastroenterology staff members in Hospital Nacional Cayetano Heredia.
This project was funded by the Global REACH Program at the University of Michigan Medical School. This project is part of a larger project through the Southwest Oncology Group with funding from the HOPE Foundation: "Clinical, Epidemiologic and Genomic Studies of Helicobacter Pylori in Lima, Peru: The role of contaminated water."
Conflictos de interés: None.
REFERENCES
1. Pilco PC, Payet E, Cáceres E. Cáncer gástrico en Lima Metropolitana . Rev Gastroenterol Peru. 2006;26(4):377-85. [ Links ]
2. Eslick GD, Lim L, Byles JE, Xia H, Talley NJ. Association of Helicobacter pylori infection with gastric carcinoma: a meta-analysis . Am J Gastroenterol. 1999 Sep;94(9):2373-9. [ Links ]
3. Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, et al. Helicobacter pylori infection and the development of gastric cancer . N Engl J Med. 2001 Sep 13;345(11):784-9. [ Links ]
4. Yamagata H, Kiyohara Y, Aoyagi K, Kato I, Iwamoto H, Nakayama K, et al. Impact of Helicobacter pylori infection on gastric cancer incidence in a general Japanese population: the Hisayama study . Arch Intern Med. Arch Intern Med. 2000 Jul 10;160(13):1962-8.
5. Ferlay J, Bray F, Parkin D. Cancer incidence, mortality and prevalence worldwide. Lyon, France: IARC; 2001. [ Links ]
6. Infection with Helicobacter pylori. In: IARC monographs on the evaluation of the carcinogenic risks to humans. Vol. 61. Schistosomes, liver flukes and Helicobacter pylori. Lyon, France: International Agency for Research on Cancer; 1994. p. 177-241.
7. Calam J, Baron JH. ABC of the upper gastrointestinal tract: Pathophysiology of duodenal and gastric ulcer and gastric cancer . BMJ. 2001 Oct 27;323(7319):980-2. [ Links ]
8. Collins JS, Knill-Jones RP, Sloan JM, Hamilton PW, Watt PC, Crean GP, et al. A comparison of symptoms between non-ulcer dyspepsia patients positive and negative for Helicobacter pylori . Ulster Med J. 1991 Apr;60(1):21-7. [ Links ]
9. Kuipers EJ, Uyterlinde AM, Peña AS, Roosendaal R, Pals G, Nelis GF, et al. Long-term sequelae of Helicobacter pylori gastritis . Lancet. 1995 Jun 17;345(8964):1525-8. [ Links ]
10. Ramirez-Ramos A, Gilman R, Leon-Barua R, Recavarren-Arce S, Watanabe J, Salazar G, et al. Rapid recurrence of Helicobacter pylori infection in Peruvian patients after successful eradication. Gastrointestinal Physiology Working Group of the Universidad Peruana Cayetano Heredia and The Johns Hopkins University . Clin Infect Dis. 1997 Nov;25(5):1027-31. [ Links ]
11. Danesh J, Lawrence M, Murphy M, Roberts S, Collins R. Systematic review of the epidemiological evidence on Helicobacter pylori infection and nonulcer or uninvestigated dyspepsia . Arch Intern Med. 2000 Apr 24;160(8):1192-8. [ Links ]
12. Fock KM. Functional dyspepsia, H. pylori and post infectious FD . J Gastroenterol Hepatol. 2011 Apr;26 Suppl 3:39-41. doi: 10.1111/j.1440-1746.2011.06649.x. [ Links ]
13. Sarnelli G, Cuomo R, Janssens J, Tack J. Symptom patterns and pathophysiological mechanisms in dyspeptic patients with and without Helicobacter pylori . Dig Dis Sci. 2003 Dec;48(12):2229-36. [ Links ]
14. Schubert TT, Schubert AB, Ma CK. Symptoms, gastritis and Helicobacter pylori in patients referred for endoscopy . Gastrointest Endosc. 1992 May-Jun;38(3):357-60. [ Links ]
15. Perri F, Clemente R, Festa V, Annese V, Quitadamo M, Rutgeerts P, et al. Patterns of symptoms in functional dyspepsia: Role of helicobacter pylori infection and delayed gastric emptying . Am J Gastroenterol. 1998 Nov;93(11):2082-8. [ Links ]
16. Vaira D, Vakil N, Rugge M, Gatta L, Ricci C, Menegatti M, et al. Effect of Helicobacter pylori eradication on development of dyspeptic and reflux disease in healthy asymptomatic subjects . Gut. 2003 Nov;52(11):1543-7. [ Links ]
17. Tack J, Talley NJ, Camilleri M, Holtmann G, Hu P, Malagelada JR, et al. Functional gastroduodenal disorders . Gastroenterology. 2006 Apr;130(5):1466-79. [ Links ]
18. Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disoders . Gastroenterology. 2006 Apr;130(5):1480-91.
19. Blaser MJ, Chyou PH, Nomura A. Age at establishment of Helicobacter pylori infection and gastric carcinoma, gastric ulcer, and duodenal ulcer risk . Cancer Res. 1995 Feb 1;55(3):562-5. [ Links ]
20. Layke JC, Lopez PP. Gastric cancer: diagnosis and treatment options . Am Fam Physician. 2004 Mar 1;69(5):1133-40. [ Links ]
21. Zheng G, Xiong Y, Xu W, Wang Y, Chen F, Wang Z, Yan Z. A two-microRNA signature as a potential biomarker for early gastric cancer . Oncol Lett. 2014 Mar;7(3):679-684 [ Links ]
Correspondence: Alejandro Bussalleu
E-mail: alejandro.bussalleu@upch.pe
Recibido: 14-12-2014
Aprobado: 22-03-2015