Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de Gastroenterología del Perú
Print version ISSN 1022-5129
Rev. gastroenterol. Perú vol.36 no.1 Lima Jan./Mar. 2016
ARTÍCULOS ORIGINALES
Local staging of rectal carcinoma: endorectal ultrasound versus magnetic resonance imaging. Systematic review of literature and meta-analysis
Estadiaje local del carcinoma rectal: imágenes de ecografía versus resonancia magnética. Revisión sistemática de la literatura y metaanálisis
Katia F. Guenaga1, Jose P. Otoch1, Everson L. A. Artifon1
1 Departamento de Cirurgia, Universidade de São Paulo. São Paulo, Brasil.
ABSTRACT
New surgical techniques in the treatment of rectal cancer have improved survival mainly by reducing local recurrences. A preoperative staging method is required to accurately identify tumor stage and planning the appropriate treatment. MRI and ERUS are currently being used for the local staging (T stage). In this review, the accuracy of MRI and ERUS with rigid probe was compared against the gold standard of the pathological findings in the resection specimens. Five studies met the inclusion criteria and were included in this meta-analysis. The accuracy was 91.0% to ERUS and 86.8% to MRI (p=0.27). The result has no statistical significance but with pronounced heterogeneity between the included trials as well as other published reviews. We can conclude that there is a clear need for good quality, larger scale and prospective studies.
Key words: Rectal cancer; Magnetic resonance imaging; Ultrasonography (source: MeSH NLM).
RESUMEN
Las nuevas técnicas quirúrgicas en el tratamiento del cáncer de recto han mejorado la supervivencia, principalmente mediante la reducción de las recidivas locales. Se requiere un método de estadificación preoperatoria para identificar con precisión el estádio del tumor y planificar el tratamiento adecuado. RNM y EAR actualmente están siendo utilizados para la estadificación local (estadio T). En esta revisión, La exactitud de la RNM y la EAR con sonda rígida se comparo com el patrón oro de los hallazgos patológicos em las muestras de resección. Cinco estudios cumplieronloscriterios de inclusión y fueron incluídos en este meta-análisis. La precisión fue 91,0% a 86,8% y EAR a la RNM (p=0,27). El resultado no tiene significación estadística pero com heterogeneidad marcada entre los ensayos incluidos, así como otras revisiones publicadas. Podemos concluir que existe una clara necesidad de buena calidad, a mayor escala y estudios prospectivos.
Palabras clave: Cáncer de recto; Imagen por resonancia magnética; Ultrasonografía (fuente: DeCS BIREME).
INTRODUCTION
Rectal cancer is a common cancer and a major cause of mortality in Western countries. The American Cancer Society (ACS) estimates about 39,600 new cases of rectal cancer will occur in 2015 (1).Treatment for rectal cancer has advanced greatly over the past decade. Our knowledge of total mesorectal excision has led to improved oncologic outcomes. The concept of extralevatorabdomino-perineal resection shows promise for reducing recurrence rates in tumors of the low rectum and anal canal. Minimally invasive techniques of laparoscopic and robotic surgery are technically feasible, and promise decreased complications in the future. New colonoscopic and endoscopic techniques offer alternatives for those unfit for surgery. Finally the adjuvant therapy with chemotherapy and radiation therapy in addition to surgery improves outcome for patients with carcinomas of the rectum.
The correct staging of rectal cancer plays a central role in prognosis as well as determining the appropriate treatment. The diagnosis is usually established by means of clinical examination (rectal digital examination), endoscopy (sigmoidoscopy and colonoscopy) and histologic confirmation. All of these techniques are poor indicators of the depth of invasion and lymph node involvement, which are both important features for treatment (2).
Advances in the management of rectal cancer over the last decade have resulted in an increased application of multimodal therapy with the aim of tailoring neoadjuvant, surgical and adjuvant therapy. The goal of the treatment is to preserve sphincter function while improving tumor resectability. Therefore, a more reliable imaging method would facilitate the design of more effective pre and post-operative treatment strategies (3).
Currently, several imaging modalities, including Endorectal Ultrasound (ERUS), Computed Tomography (CT), Magnetic Resonance Imaging (MRI), and Positron Emission Tomography (PET) or PET-CT, are used to assess preoperative staging. MRI and ERUS are the two standard local anatomical imaging techniques used for the primary staging of rectal cancer (4). The aim of this review was to assess the current evidence for both methods in the local staging of rectal cancer.
METHOD
This was a systematic review of diagnostic test accuracy studies, with meta-analysis and was approved by the local Research Ethics Committee.
The aim of this review was to establish the actual role of Endorectal Ultrasonography (ERUS) with rigid probe and Magnetic Resonance Imaging (MRI) in the local staging of rectal cancer.The target condition is rectal cancer. Patients with the diagnosis of primary rectal cancer were included. The exclusion criteria were obstructive tumor, neoadjuvant therapy previous to staging evaluation and patients undergoing emergency surgery or palliative treatment. The reference standard was the pathological examination of the surgical specimen, staged (TNM) according to the guidelines of the American Joint Committee on Cancer and International Union Against Cancer (UICC).
A systematic review of the literature was performed by searching the databases CINAHL, EMBASE, LILACS, MEDLINE, SCISEARCH and The Cochrane Library between 2004and 2015. Relevant studies on the diagnostic performance comparing endoluminal US (not endoscopic), MR imaging and histopathological analysis in the staging of rectal cancer were identified. The MEDLINE database was searched with the following keywords: (a) "rectal neoplasms" (medical subject heading, or MeSH) and (b) "magnetic resonance imaging" (MeSH) or "ultrasonography" (MeSH) and (c) "specificity" or "false negative" or "accuracy" as text words. The EMBASE, Cochrane, and CANCERLIT databases were also checked for relevant articles by using (a) "rectal cancer" and (b) "magnetic resonance imaging" or "ultrasonography" and (c) "specificity" or "false negative" or "accuracy" as text words. A hand-search of references from selected articles was also performed to identify additional relevant articles. Data were independently extracted by the reviewers and cross-checked; articles that did not present raw data were excluded.
Studies eligible for inclusion meta-analysis were required to compare, stage for stage with histopathology, MRI and ERUS assessment of T stage of rectal cancer in larger goal.
The methodological quality of the selected studies was critically assessed using the QUADAS-2 tool, a checklist to assess the quality of the diagnostic accuracy of studies (5).
Statistical analysis
The summary thereof was obtained by the weighted sensitivity and specificity and presented graphically by Forest Plot. The likelihood ratios were calculated. The accuracy of the methods was obtained by the SROC curve and was compared using the test differences between proportions.
Meta-analysis of the results was performed using software package Meta-Disc 1.4 (freeware developed by the Unit of Clinical Biostatistics, Ramón y Cajal Hospital, Madrid, Spain) (6).
RESULTS
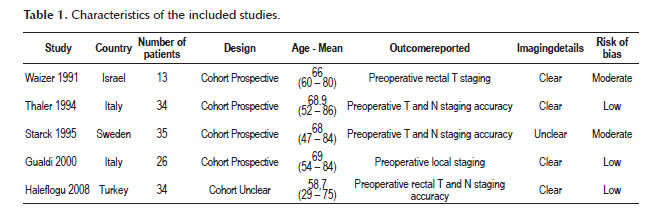
With the search strategy the authors chose 104 citations. After careful reading of the titles and abstracts, 26 articles were selected for full-text analysis and critical appraisal. Five were able to meet the inclusion criteria and were included in this meta-analysis (Table 1).
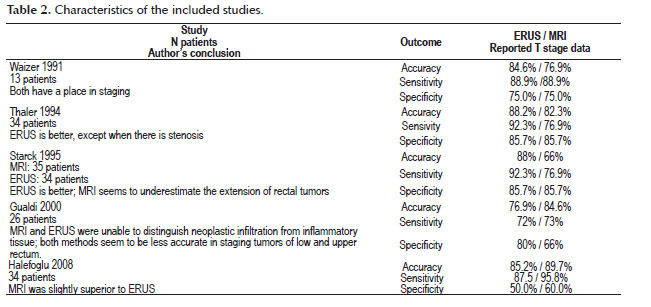
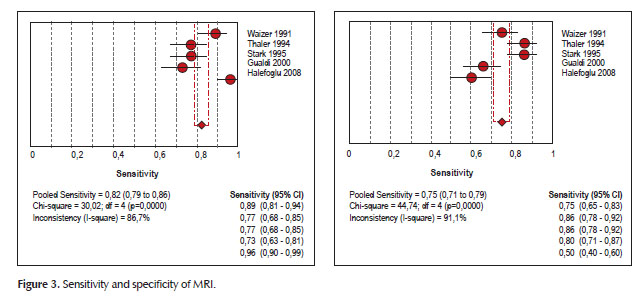
In Table 2 are described the results of each included study and the author’s conclusion.
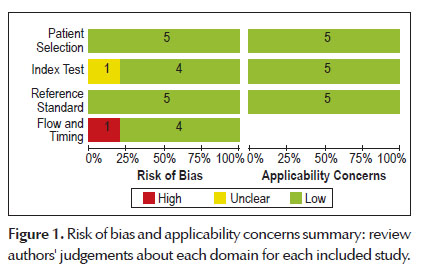
The methodological quality graph (QUADAS-2) presents the percentage of included studies for which the item was rated "low", "high" or "unclear", for each quality assessment domain. The graph shows that the potential area of concern was the description of the patient flow and the index test. Figure 1 summarizes the risk of bias and applicability judgments on the five studies included.
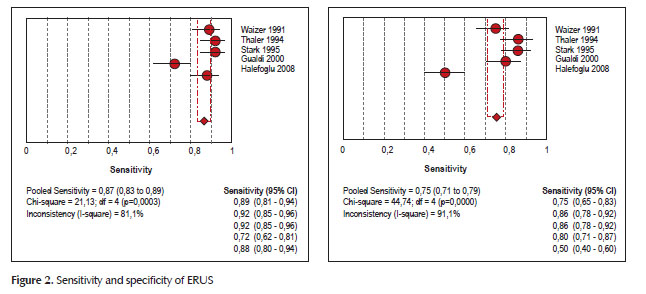
The sensitivity and specificity values of ERUS were equal to 86.6% (IC a 95%: 83.3%; 89.5%) and 75.4% (IC a 95%: 71.4%; 79.1%). respectively.
For MRI the values of sensitivity and specificity were 82.4% (IC a 95%: 78.8%; 85.6%) and 74.6% (IC a 95%: 70.5%; 78.4%). respectively.
The detailed sensitivity and specificity with 95 % of CI for each individual study were seen in Forest plot (Figures 2 and 3).
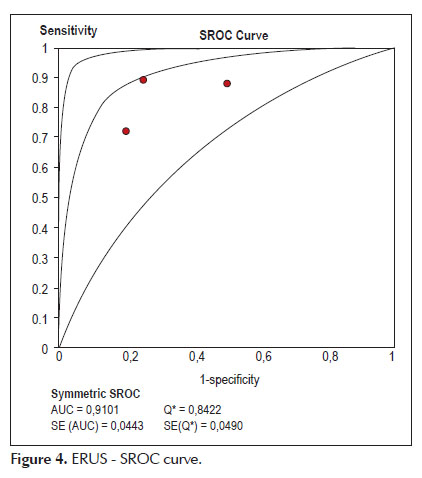
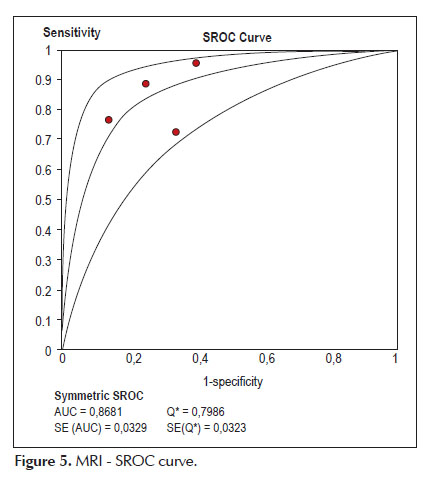
Using the fitted summary SROC curve we observed that the area under curve (Accuracy) is 91.0% to ERUS compared to 86.8% to MRI with p=0.27 (non-significant). showed in Figures 4 and 5.
DISCUSSION
Pre-operative staging of rectal cancer with accuracy is mandatory in planning the surgical treatment and is the strongest predictor for recurrence (7, 8).
This review searched the studies comparing Endorectal Ultrasound (ERUS) and Magnetic Resonance Imaging (MRI) with the surgical histopathological analysis for local staging of rectal cancer.
Studies analyzing Endoscopic Ultrasound were not included; despite Colaiácovo et al. in 2014 (9) declared that the findingsof there study may indicate equivalence in the diagnostic value of both flexibleand rigid devices, the reviewers believe there are differences between the methods.
Regarding to the staging the authors understanding that the N stage is another review with proper comments and decided directed the analysis and the results to the T stage - wall invasion.
The results haveno statistical significance (p=0.27) and with pronounced heterogeneity between the included trials. The major weakness of this review must be the relatively small number of identified studies, containing small numbers of patients. The significant heterogeneity is an additional weakness. The statistical heterogeneity depends on the clinical and methodological differences within the trials. Inevitably, studies brought together in a systematic review will differ. Further area of concern is the inclusion of many older studies that may have used technology that is now obsolete. Another question mark is the time interval between performance of diagnostic tests and the reference test that should be short. A longer period will lead to a greater change in the disease status and decrease in the discriminatory power of the diagnostic test. In all of the studies, this time period was not described; however a large interval is not likely, given the disease under consideration.
To address the question data were abstracted by the included studies and supplemented this analysis with information from narrative reviews and other sources (e.g., FDA alerts, National Guidelines).
A large number of features are involved in the staging of rectal cancer and also important for diagnostic accuracy:
-
Patient characteristics: disease stage, age, or sex distribution. Anatomic landmarks for identification of the most inferior portion of the peritoneal membrane were tip of seminal vesicles in men and the utero- cervical angle in women, but some limitations can occur as postoperative status or retroversion of the uterus for example. It is difficult to position the ultrasound probe and visualize high with stenosing tumors, resulting in inconclusive results in >10% of patients (10).
-
The importance of rectal cancer imaging protocols on interpretation accuracy has been reported (11).In a review for U.S. Department of Health and Human Services, Bruening et al. (12) consulted a Technical Expert Panel who determined, after discussion and consensus, that transabdominal ultrasound and MRI using endorectal coils are obsolete for staging rectal cancer. New protocols includes a pelvic phased-array multichannel coils and larger-FOV imaging for MRI (13) or 2D or 3D with 360°rotating anorectal transducers featuring high frequency and between 6-16 MHz, focal distance between2.8 and 6.2-cm, and automatic image acquisition without manual movement of the transducer for anorectal ultrasonography (14). The accuracy of three-dimensional ERUS in the assessment of the infiltration depth of rectal cancer is comparable to conventional ERUS (15). But the 3D volume can be freelyrotated, rendered, tilted and sliced to allow the operator to infinitely vary the different section parameters and visualize the lesion at different angles and in different planes(coronal, frontal, axial) to get the most information from the data and may be helpful for the planning of surgery in the future (16).
-
A drawback of ERUS compared to cross-sectional imaging techniques is that it is high lyoperator- dependent and requires a learning curve before optimal diagnostic performance can be obtained (10,17).
-
Some authors questioned the cost-effectiveness for MRI. Brown et al. (18) conducted a study cohort and showed that MRI results in significant treatment cost benefits that are very likely to offset the costs of the procedure itself.
Bipat et al. in 2004 (19) conducted a review and metaanalysis comparing Computed Tomography (CT) with ERUS and MRI and declared that endoluminal US seems to be a better diagnostic imaging test for local staging than are CT and MRI imaging but they cited that ERUS has several limitations: operator dependency; limitation to tumors located 8–10 cm from the anal verge when a rigid probe is used; and no assessment of stenotic tumors.
The authors of one exclusion study (20) concluded that both methods are still complementary techniques but their use is influenced by the operator experience, the availability of instruments and methods in the diagnosis centers. Unfortunately it was not possible to include this article since we could not clarify the data.
Arezzo et al. (21) in an Italian guideline propounded that ERUS can distinguish layers within the rectal wall. It provides the most accurate assessment of the depth of tumour penetration for early rectal cancer, and perirectal spread. It is recommended as the best modality for the staging. The use of 3D ERUS could potentially further increase the accuracy. MRI is recommended as the technique of first choice for the overall primary staging of rectal cancer. Differentiation between T1 and T2 tumours is not possible with MRI. Endorectal ultrasound remains the imaging method of first choice to differentiate between T1 and T2 tumours if local resection is being considered.
Kolev et al. (22) declared that the results from there study and the literature review show that 3-D ERUS is the diagnostic modality of choice in staging of cancer penetration in the rectal wall. In accordance with them Kwok et al. (23) also found ERUS to be the most accurate modality in the assessment of wall penetration.
The main strength of MRI is the evaluation of large T3 tumours that penetrate the muscular rectal wall and T4 tumours invading adjacent organs. MRI, however, is known to have difficulties in differentiating between superficial T1 and T2 tumours. As opposed to EUS, with MRI it is not possible to separately appreciate all three layers of the rectal wall. The submucosal layer of the rectal wall is not visualized on phased-array MRI (except when there is oedema). Hence, differentiation between a T1 tumour limited to the submucosa and a T2 tumour penetrating the muscularis propria is not feasible (10). Rafaelsen et al. (24) evaluating the agreement between transrectal ultrasound (TRUS) and magnetic resonance imaging (MRI) in classification of ≥T3 rectal tumors concluded there was very good inter modality agreement between TRUS and MRI in classification of tumors as early and advanced ≥T3.
Recently, innumerable reviews, guidelines and articles discussing the role of the different imaging modalities for local staging of rectal cancer were conducted. The definitive results are very resembling (25-29).
Conclusion
We will ask permission to Nielsen and Wille- Jørgensen (30) and finalize our conclusion: "despite much knowledge, there is no international consensus for the staging of rectal cancer".
Implications for practice
For rectal T staging, ERUS and MRI appear to not be statistically significantly different in accuracy. Rectal wall layers were reliably demonstrated with both methods, but with some restrictive condition: (a) the difference between T1 and T2 tumours remaining a point of obstacle to MRI despite the news technologies; (b) ERUS has lack of capacity with obstructive tumours due to the probe is unable to reach the upper limit of the tumor.
Implications for research
There is a clear need for good quality, larger scale prospective studies analyzing MRI and ERUS (with rigid probe), compared with pathological surgery specimen as a reference standard. Collaborative, properly designed trials that involve a large, representative number of individuals, with explicit clinical inclusion and exclusion criteria, description of the interval between the anatomical imaging techniques and the reference test.
Conflict of Interest: No funding was used for the preparation of this manuscript. None of the authors have commercial associations that might pose a conflict of interest in connection with the submitted article.
REFERENCES
1. Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, et al. GLOBOCAN 2012 v1.1, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon: IARC; 2013 [cited 2014 May 19]. Available from: http://globocan.iarc.fr [ Links ]
2. Brown G, Radcliffe AG, Newcombe RG, Dallimore NS, Bourne MW, Williams GT. Preoperative assessment of prognostic factors in rectal cancer using high-resolution magnetic resonance imaging. Br J Surg. 2003;90(3):355-64. [ Links ]
3. Arya S, Das D, Engineer R, Saklani A. Imaging in rectal cancer with emphasis on local staging with MRI. Indian J Radiol Imaging. 2015;25(2):148-61. [ Links ]
4. Samee A, Selvasekar CR. Current trends in staging rectal cancer. World J Gastroenterol. 2011;17(7):828-34. [ Links ]
5. Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, et al. QUADAS-2: a revised toolfor the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155(8):529-36. [ Links ]
6. Abraira V, Zamora J, Muriel A, Cano A, Royuela A. Web del diagnostico [Internet]. Madrid: Hospital Universitario Ramón y Cajal; [cited 2015 Jul 9]. Available from: www.hrc.es/investigacion/diag/diagnostico.htm [ Links ]
7. Smith N, Brown G. Preoperative staging of rectal cancer. Acta Oncol. 2008;47(1):20-31. [ Links ]
8. Garcia-Aguilar J, Pollack J, Lee SH, Hernandez de Anda E, Mellgren A, Wong WD, et al. Accuracy of endorectal ultrasonography in preoperative staging of rectal tumors. Dis Colon Rectum. 2002;45(1):10-5. [ Links ]
9. Colaiácovo R, Assef MS, Ganc RL, Carbonari APC, Silva FAOB, Bin FC, et al. Rectal cancer staging: Correlation between the evaluation with radial echoendoscope and rigid linear probe. Endosc Ultrasound. 2014;3(3):161-6. [ Links ]
10. Lambregts DMJ, Beets-Tan RGH. Optimal imaging staging of rectal cancer. EJC Suppl. 2013;11(2):38-44. [ Links ]
11. Suzuki C, Torkzad MR, Tanaka S, Palmer G, Lindholm J, HolmT, et al. The importance of rectal cancer MRI protocols on interpretation accuracy. World J Surg Oncol. 2008;6:89. [ Links ]
12. Bruening W, Sullivan N, Carter Paulson E, Zafar H, Mitchell M, Treadwell J, et al. Imaging tests for the staging of colorectal cancer. Comparative Effectiveness Review No. 142 [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2014 [cited 2015 Jul 9]. Available from: www.effectivehealthcare.ahrq.gov/reports/final.cfm [ Links ]
13. Jhaveri KS, Hosseini-Nik H. MRI of rectal cancer: an overviewand update on recent advances. AJR Am J Roentgenol. 2015;205(1):W42-55. [ Links ]
14. Regadas FSP, Murad Regadas SM, Barreto RGL. Three-dimensional ultrasonography of pelvic floor and anorectal anatomy - Charter 2:9-15. In: Imaging Atlas of the Pelvic Floor and Anorectal Diseases. Milano: Springer-Verlag Italia; 2008.
15. Hünerbein M, Pegios W, Rau B, Vogl TJ, Felix R, Schlag PM. Prospective comparison of endorectal ultrasound, three-dimensional endorectal ultrasound, and endorectal MRI in the preoperative evaluation of rectal tumors. Surg Endosc. 2000;14(11):1005-9. [ Links ]
16. Miro AGF, Grobler S, Santoro GA. Preoperative staging of rectal cancer:role of endorectal ultrasound – Charter 4: 49-74. In: Rectal Cancer – A Multidisciplinary Approach to Management. Rijeka, Croatia: InTech; 2011.
17. Regadas SMM, Regadas FSP, Rodrigues LV, Silva FR, Lima DMR, Regadas-Filho FSP. Importance of the tridimensional ultrasound in the anorectal evaluation. Arq Gatroenterol. 2005;42(4):226-32. [ Links ]
18. Brown G, Davies S, Williams GT, Bourne MW, Newcombe RG, Radcliffe AG, et al. Effectiveness of preoperative staging in rectal cancer: digital rectal examination, endoluminal ultrasound or magnetic resonance imaging? Br J Cancer. 2004;91(1):23-9. [ Links ]
19. Bipat S, Glas AS, Slors FJ, Zwinderman AH, Bossuyt PM, Stoker J. Rectal cancer: local staging and assessment of lymph node involvement with endoluminal US, CT, and MRimaging—a metaanalysis. Radiology 2004;232(3):773-83. [ Links ]
20. Balena V, Martino D, Lorusso F, Martino T, Valerio P. Endorectal ultrasound and magnetic resonance imaging (MRI) scan in rectal cancer: a comparative study. Arch Ital Urol Androl. 2010;82(4):259-61. [ Links ]
21. Arezzo A, Bianco F, Agresta F, Coco C, Faletti R, Krivocapic Z, et al. Practice parameters for early rectal cancer management: Italian Society of Colorectal Surgery (Societa Italiana di Chirurgia Colo- Rettale; SICCR) guidelines. Tech Coloproctol. 2015;19(10):587-93. [ Links ]
22. Kolev NY, Tonev AY, Ignatov VL, Zlatarov AK, Bojkov VM, Kirilova TD, et al. The role of 3-d endorectal ultrasound in rectal cancer: our experience. Int Surg. 2014;99(2):106-11. [ Links ]
23. Kwok H, Bissett IP, Hill GL. Preoperative staging of rectal cancer. Int J Colorectal Dis. 2000;15(1):9-20. [ Links ]
24. Rafaelsen SR, Vagn-Hansen C, Sørensen T, Pløen J, Jakobsen A. Transrectal ultrasound and magnetic resonance imaging measurement of extramural tumor spread in rectal cancer. World J Gastroenterol. 2012;18(36):5021-6. [ Links ]
25. Burdan F, Sudol-Szopinska I, Staroslawska E, Kolodziejczak M, Klepacz R, Mocarska A, et al. Magnetic resonance imaging and endorectal ultrasound for diagnosis of rectal lesions. Eur J Med Res. 2015;20(1):4. [ Links ]
26. Heo SH, Kim JW, Shin SS, Jeong YY, Kang HK. Multimodal imaging evaluation in staging of rectal cancer. World J Gastroenterol. 2014;20(15):4244-55. [ Links ]
27. Hoeffel C, Mulé S, Laurent V, Bouché O, Volet J, Soyer P. Primary rectal cancer local staging. Diagn Interv Imaging. 2014;95(5):485-94. [ Links ]
28. Kennedy E, Vella ET, Blair Macdonald D, Wong CS, McLeod R; Cancer Care Ontario Preoperative Assessment for Rectal Cancer Guideline Development Group. Optimisation of preoperative assessment in patients diagnosed with rectal cancer. Clin Oncol (R Coll Radiol). 2015;27(4):225-45. [ Links ]
29. Zhou Y, Shao W, Lu W. Diagnostic value of endorectalultrasonography for rectal carcinoma: A meta-analysis. J Can Res Ther. 2014;10 Suppl:319-22.
30. Nielsen LBJ, Wille-Jørgensen P. National and international guidelines for rectal cancer. Colorectal Dis. 2014;16(11):854-65. [ Links ]
Correspondence: Katia F. Guenaga.
Rua Ministro João Mendes, 60/31 – Santos. São Paulo, Brasil. CEP 11040-260
E-mail: kfg012@terra.com.br
Recibido: 23-12-2015
Aprobado: 28-01-2016