Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de Gastroenterología del Perú
Print version ISSN 1022-5129
Rev. gastroenterol. Perú vol.37 no.2 Lima Apr./Jun. 2017
ARTÍCULOS ORIGINALES
Endoscopic prediction of tumor invasion depth in earlygastric neoplasia: a prospective study in Peru
Predicciónendoscópica de invasión tumoral en neoplasia gástrica temprana. Estudioprospectivo en Perú
Fernando Palacios Salas1,2,Estefanía LizaBaca1
1 Servicio deGastroenterología, Hospital Nacional Edgardo RebagliatiMar tins. Lima, Perú.
2 ClínicaDelgado. Lima, Perú.
ABSTRACT
Introduction: Endoscopic resection is the first option treatment of early gastric cancer with invasion to mucosa or superficial submucosa,because the risk of nodal metastasis is negligible. Then the prediction oftumor invasion is cardinal. Objectives: Determine the accuracy of endoscopicprediction for tumor invasion depth in early gastric neoplasia and defineendoscopic characteristics associated with massive submucosal invasion.Materials and methods: Prospective sudy of diagnostictest validation. We included patients with early gastric neoplasiasthat were endoscopically or surgically resected from January 2012 to May 2016.Every lesion was looked for the presence of these features: margin elevation,central elevation, irregular surface, enlarged folds,size > 30mm and rigidity. The invasion prediction was categorized in: M-Sm1when none feature was present, Sm2 when 2 or more features were present, and indeterminated when only one feature was present. Wecompared endoscopic prediction to pathological staging and determineddiagnostic accuracy. Results: The global accuracy for endoscopic prediction was98.2%. Sensitivity, specificity, positive and negative predictive values forM-Sm1 prediction were 97.6, 100, 100 y 92.8%, and for Sm2 prediction were 100,97.6, 92.8 y 97.6%, respectively. Rigidity, irregular Surface, margin elevationand enlarged folds were associated with Sm2 invasion. Conclusions: Endoscopicprediction of tumor invasion depth in early gastric neoplasia is very accurate.The main endoscopic feature associated with Sm2 invasion is rigidity.
Keywords: Stomach neoplasms; Endoscopy; Forecasting (source: MeSHNLM)
RESUMEN
Introducción: La resecciónendoscópica constituye el tratamiento de elección del cáncer gástrico tempranocon invasión a la mucosa o submucosa superficial, pues tiene riesgo casi nulode metástasis ganglionar. Por tanto, la predicción de invasión tumoralescardinal. Objetivos: Determinar la precisión de la predicción de invasióntumoral de neoplasia gástrica temprana por endoscopía convencional y definircaracterísticas endoscópicas asociadas a invasión submucosa masiva. Material ymétodos: Estudio prospectivo de validación de una prueba diagnóstica. Seincluyeron todos los pacientes con neoplasias gástricas tempranas que fueronresecadas endoscópica o quirúrgicamente de enero 2012 a mayo 2016. En cadalesión se definió la presencia de las siguientes características: Elevación demárgenes, elevación central, irregularidad de la superficie, engrosamiento depliegues, tamaño >30 mm y rigidez. La predicción de invasión se categorizóen: M-Sm1 cuando no tenía ninguna característica, Sm2 cuando tenía 2 o máscaracterísticas, e indeterminada cuando sólo tenía una característica. Secomparó la predicción endoscópica con el estadiajepatológico de los especímenes y se determinó su precisión diagnóstica.Resultados: La precisión global de la predicción endoscópica fue de 98,2%. Lasensibilidad, especificidad, VPP y VPN para la predicción M-Sm1 fue 97,6, 100,100 y 92,8%, y para la predicción Sm2 fue 100, 97,6, 92,8 y 97,6%,respectivamente. La rigidez, irregularidad en la superficie, elevación de losmárgenes y engrosamiento de pliegues, se asociaron significativamente coninvasión Sm2. Conclusiones: La predicción endoscópica de invasión tumoral enneoplasia gástrica temprana es muy precisa. La principal característicaendoscópica asociada a invasión Sm2 es la rigidez.
Palabrasclave : Cáncer gástrico; Endoscopía;Predicción (fuente: DeCS BIREME).
INTRODUCTION
In many western countries, the incidence of gastricadenomas. gastric cancer is declining; however, inPeru is still frequent, and it is the leading cause of cancer death, Endoscopicresection of early gastric cancer with almost unfortunately, because most casesare diagnosed in no risk of nodal metastasis is the first option treatment inadvanced stages (1,2). The early gastric cancer (EGC) is Japan andSouth Korea, as it has shown excellent short defined as the one thatcompromises the mucosa and/ or submucosa, regardless of nodal involvement (3),while early gastric neoplasia involves both EGC and gastric adenomas.
Endoscopic resection of early gastric cancer with almostno risk of nodal metastasis is the first option treatment in Japan and SouthKorea, as it has shown excellent short and long term oncological results (cureand survival rates), being, unlike surgery, a minimally invasive therapy (4-8).The enthusiasm generated by these good results has made the techniques ofendoscopic resection increasingly used in occidental countries, and soguidelines of the European Society of Gastrointestinal Endoscopy (ESGE) and theAmerican Society of Gastrointestinal Endoscopy (ASGE) recommend its use (9,10).
The Japan Gastroenterological Endoscopy Society (JGES)and the Japanese Gastric Cancer Association (JGCA), published a recentguideline defining the indications for endoscopic resection of early gastriccancer as absolute and expanded (11). The absolute indication is thedifferentiated adenocarcinoma, intramucosal, lessthan 20 mm. The expanded indications are based on the works of Gotoda and Hirasawa; and theyare: (1) Differentiated intramucosal adenocarcinoma,without ulcer, any size; (2) Differentiated intramucosaladenocarcinoma, with ulcer, less than 30 mm; (3) Differentiated adenocarcinomawith superficial submucosa invasion (Sm1, <500 um depth), without ulcer,less than 30 mm; (4) Undifferentiated intramucosaladenocarcinoma, without ulcer, less than 20 mm (12,13).The resected specimen should show lateral and vertical margins free ofneoplasia, and absence of lymphovascular involvementto consider curative resection as well.
There are two techniques of endoscopic EGC resection;mucosectomy (EMR: endoscopic mucosal resection) andendoscopic submucosal dissection (ESD). The last mentioned allowsen bloc resection of very large lesions, even with ulcer orscar, being able to satisfy the expanded indications proposed by Gotoda. Three recent meta-analysis have shown that ESD hassignificantly greater rates of en bloc and completeresections than EMR, even in small lesions with an absolute indication ofendoscopic resection (14-16).
To define the indication for endoscopic resection ofthe early neoplasic lesions detected, thecharacterization of the lesions is very important. The main point of thecharacterization is the prediction of depth of cancer invasion or "T"stage, and the distinction between intramucosaladenocarcinoma or with minimal invasion of the submucosa (M-Sm1), withnegligible risk of nodal metastasis and, therefore, candidates for endoscopicresection, and adenocarcinoma with massive invasion of the submucosa (Sm2, >500 um), with a risk of around 20% of nodal metastasis and, therefore,candidates for surgical treatment. The accuracy of the prediction is veryimportant, on one side, to avoid unnecessary surgery in potentially curablepatients with endoscopic resection; and on the other side, to minimize retreatmentafter non-curative ESD.
The prediction of tumor invasion can be made by echoendoscopy with an accuracy between 41.4 and 86% (17).The limitations of echoendoscopy are the low abailability, the ideal requirement of high-frequency miniprobesto obtain more precise results and, mainly, overstaging, being present in up to 42% of lesionscategorized as Sm2, especially in those with ulcer (18). Inaddition, some studies show that its precision in predicting tumor invasion is notgreater than that of conventional endoscopy (19-21). A recentmeta-analysis shows a relatively low precision of the echoendoscopyin the prediction of tumor invasion in EGC, not considering it an indispensablestudy (18).
The easiest and most practical way to perform theprediction of tumor invasion is with conventional endoscopy and so do manyexperienced centers in Japan and South Korea, with a diagnostic accuracy of73-83% (22-24). The problem is that this prediction is based onexperience and is usually done empirically without having standardized orobjectively defined criteria. The endoscopic features associated with massivesubmucosal invasion are based on only a few retrospective studies that testtheir accuracy without having replicated their experience in occidentalcountries. Yao and col. consider that two important characteristics associatedwith a massive submucosal invasion are the marked elevation of the margins andthe central elevation with submucosal aspect (25,26).An attempt to objectify the prediction of invasion is given by Abe's study,after defining 4 characteristics associated with Sm2 invasion, assigns a scoreof 2 for the elevation of margins and size greater than 3 cm, and a score of 1for marked redness and irregular surface, establishing, that a score of 3 ormore is associated with massive submucosal invasion (Sm2), with sensitivityfrom 29.7 to 45.9%, specificity from 93.1 to 93.7% and accuracy from 82.5 to84.8% (27).
The main objective of the present research is todetermine the accuracy of prediction of tumor invasion depth in early gastricneoplasia by conventional endoscopy, in relation to the pathological analysisof endoscopically or surgically resected specimens. The secondary objectivesare to define which endoscopic features are associated with massive submucosalinvasion and what clinical-pathological features affect the accuracy ofprediction of tumor invasion depth.
MATERIALS AND METHODS
Patients andtreatment
Prospective study to validate a diagnostic test at theEdgardo Rebagliati Martins National Hospital (Lima-Peru) between January 2012 and May 2016. Patients with endoscopic andhistological diagnosis of early neoplasic stomachlesions (gastric adenoma and early gastric cancer) in who were resectedendoscopically or surgically were included. Patients with lesions with a clearendoscopic appearance of advanced gastric cancer, lesions greater than 20 mmwhose histology was undifferentiated adenocarcinoma, and lesions resected morethan 3 months after endoscopy for staging were excluded. All patients withlesions that fulfilled the absolute and expanded indications of endoscopicresection were treated with endoscopic submucosal dissection by one operator.If the specimen showed lateral and vertical margins free of neoplasia, as wellas absence of lymphovascular involvement, it wasconsidered curative resection. In any other case, the resection was considerednon-curative, and the patient was oriented to surgical treatment.
Patients with early gastric neoplasia whith either difficult localization or had not absolut or expanded criteria for endoscopic resection(mainly prediction of Sm2 invasion) or who had incomplete or non-curativeendoscopic resections, were treated surgically.
All patients signed informed consent prior to stagingendoscopy and resection procedure.
Endoscopic assessment of depth of invasion
The equipment used was FUJINON EG-590WRgastroscopes, EPX-4400 video processor or EPX-4450 HD(FUJINON Co. Ltd., Tokyo - Japan). The examination was performed by oneendoscopist with more than 10 years of experience andtrained at the Keio University Hospital and at the National Cancer Center,Tokyo - Japan. The endoscopic evaluation was performed with white light and insome cases with FICE to highlight the characteristics of the surface;chromoendoscopy with contrast stain was not routinely usedfor not having indigo carmine. The macroscopic type of the neoplasiaswas defined according to the Japanese and Paris classification, as Type I(protruded), Type IIa (slightly elevated), IIb (flat), IIc (slightlydepressed), III (excavated) and combinations (3,28).Types I, IIa, and IIa + Iwere classified as elevated; types IIc, III, and IIc + III were classified as depressed; type IIb as flat; and types IIa + IIc or IIc + IIaas mixed. The location of the lesions was defined in upper thirds (fundus andupper body), middle (remaining body and angle) and lower (antrum). The scar oractive ulcer was considered as a positive ulcer finding (UL+) (3).
In gastric lesions, the following features wereevaluated as potential indicators of submucosal invasion:
-Size greater than 30 mm: This feature was onlyevaluated in protruding lesions.
-Marked margin elevation (with submucosal aspect): Thediscrete margin elevation, which almost flattened with maximum insufflation,was not considered.
-Central elevation (with submucosal aspect):
Manifested with maximum insufflation. -Pronouncedirregular surface. -Enlarged folds (drumstick, fused): Each distorted
fold feature wasevaluated separately. We did not consider the folds that converge to the lesionand gradually decrease in thickness, as well as those that are amputatedwithout being thickened.
-Rigidity: This feature is dynamic and it was evaluatedwith changes of insufflation and with peristaltism.
The prediction of the depth of invasion in lesionsconsidered as early gastric neoplasias was performedas follows:
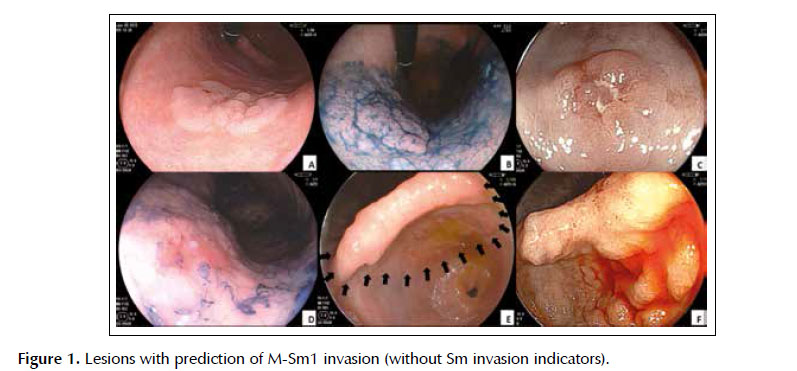
-Intramucosal lesions orwith superficial invasion of the submucosa (M-Sm1): Absence of submucosalinvasion indicators ( Figure 1 ).
A and B) Type 0-IIa lesions with 25 mm and 20 mm indiameter, in body, with very discrete surface irregularity. C) Type 0-IIc+IIalesion with 9 mm in diameter, in antrum, with slightly elevated margins. D)Type 0-IIc lesión with 16 mm in diameter, in body,with regular surface. E and F) Type 0-IIa with 55 mm in diameter, in angle andantrum, with regular granular surface.
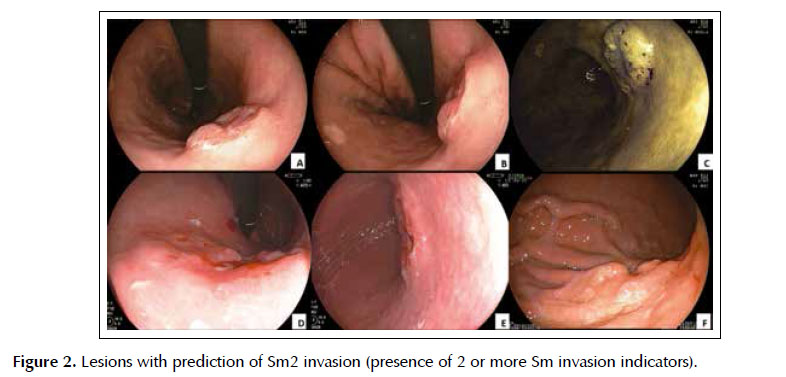
A and B) Type 0-IIc lesion with 22 mm in diameter, inbody, with irregular surface, marked central elevation and margins elevationswith submucosal appearence. C) Type 0-Is lesion withdepression with 25 mm in diameter, in body, with irregular surface and rigidity.D and E) Type 0-IIc lesion with 32 mm in diameter, in body, with very irregularsurface, margin elevation with submucosal appearenceand rigidity. F) Type 0-IIc lesion with 30 mm in diameter, in body, with
-Lesions with massive invasionofthe submucosa (Sm2): Presence of 2 or more indicators of submucosal invasion( Figure 2 )
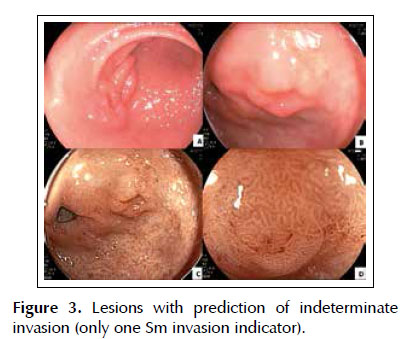
-Lesions with indeterminate invasion prediction:Presence of only 1 indicator of submucosal invasion ( Figure 3 ).
Histopathological analysis
Endoscopic or surgical specimens were fixed withformalin and sectioned every 2 or 5 mm, respectively, and stained with hematoxylin- eosin.
A, B and C) Type 0-IIc+IIa lesions with 26 mm and 10mm in diameter, in antrum, with only margin elevation. D) Previous lesion withmagnification and FICE, clear demarcation line, slight irregularity ofmicrostructure and microvasculature (this lesion was recategorizedas M-Sm1 due to these findings and resected by ESD).
The following features were determined: -Tumor size:<20 mm; 20 - 30 mm, >30 mm. -Degree of differentiation: Differentiated(tubular well or moderately differentiated and papillary adenocarcinoma) andundifferentiated (poorly differentiated adenocarcinoma, signet-ring or mucoidcell). Adenomas were categorized as differentiated. -Invasion depth: Mucosa(M), superficial submucosa (Sm1 <500 um), deep or massive submucosa (Sm2>500 um) and muscularis propia(Mp). Then, two categories were regrouped for thecorrelation with endoscopic prediction; M-Sm1 and Sm2-Mp. -Lymphovascularinvasion: Present or absent.
Statistic analysis
The accuracy of the endoscopic prediction of tumorinvasion depth was determined by comparing it with histopathology findings. Thesensitivity, specificity, PPV and NPV of both the endoscopic prediction ofM-Sm1 lesions and the endoscopic prediction of Sm2 invasion were alsodetermined. For this analysis only these two categories were used, and thegroup of patients with undetermined prediction was not included. Bivariateanalysis (chi-square test) was then performed to determine which endoscopicfeatures were significantly associated with Sm-2 invasion. Finally, we determinedwhich clinical and pathological features affected the precision of theendoscopic prediction of tumor invasion depth. All analysis were performedusing the statistical package (SPSS) version 23.0, considering as level ofstatistical significance any value of P < 0.05.
RESULTS Clinical -pathological features
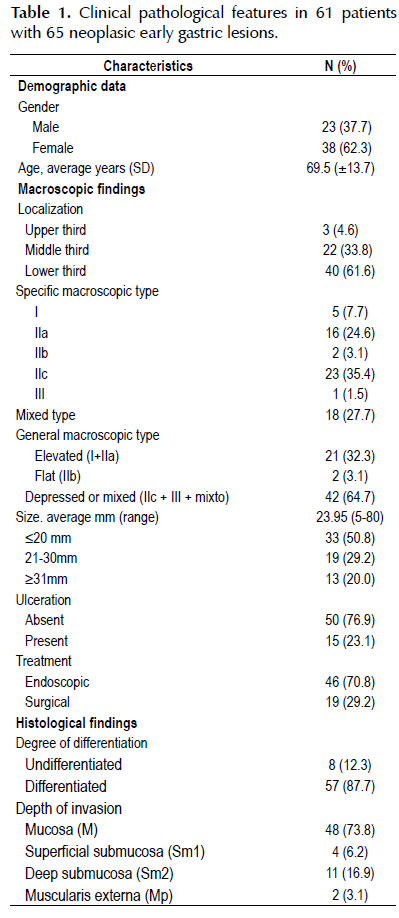
Clinical-pathological features of patients and lesionsare shown in Table 1 . A total of 61 patients, female most of them (61.5%) with an averageage of 69.5 years, in which 65 lesions compatible with early gastric neoplasiawere identified. Regarding the macroscopic type of lesions, 42 (62.7%) had adepressed component, and 15 (23.1%) had an ulcerative finding (ulcer or scar).The average lesion size was 23.95 mm, range 5-80 mm. Endoscopic resection of 46lesions (70.8%) and surgical resection of 19 (29.2%) were performed. From theresected lesions 17 were adenomas with high grade dysplasia and 48 were EGC.About the histological type, 87.7% of lesions were differentiated and 12.3%were undifferentiated. In terms of tumor invasion depth, 48 (73.8%) lesionswere intramucosal, 4 invaded Sm1, 11 invaded Sm2 and2 infiltrated the Mp.
Evaluation of the prediction of tumor invasion byconventional endoscopy
Table 2 presents data from evaluation of the prediction of tumor invasion depthby conventional endoscopy in relation to histopathological evaluation. From 65lesions, the prediction of tumor invasion depth by conventional endoscopy was:M-Sm1 in 42 (64.62%), Sm-2 in 14 (21.54%) and indeterminate in 9 (13.85%). The9 lesions with prediction of indeterminate invasion belong to superficiallesions, which in the histopathological evaluation showed M invasion in 5 casesand Sm1 invasion in 4. The overall accuracy of the prediction of tumor invasión depth by conventional endoscopy was 98.2% withoverestimation in one case (1.8%) and underestimation in two cases (3.6%). Thesensitivity, specificity, PPV, and NPV for the prediction of M-Sm1 invasionwere 97.6%, 100%, 100% and 92.8%, respectively; while the prediction of Sm2invasion were 100%, 97.6%, 92.8%, and 97.6%, respectively.
Endoscopic features associated with massive submucosalinvasion
Table 3 shows the endoscopic features of lesions associated with massivesubmucosal invasion in histopathology. Bivariate analysis showed that rigidity,irregular surface, margin elevation, central elevation, and enlarged folds wereassociated with significant submucosal invasion; the association was greaterwith rigidity.
Clinical pathological features affecting the accuracyof endoscopic staging
Table 4 shows the accuracy of the endoscopic staging in relation to themacroscopic and histological features of the lesions. The accuracy ofendoscopic staging is lower in lesions located in the upper third of thestomach, with protruded morphology and size more than 20 mm. The histologicaltype of the lesions, differentiated or undifferentiated, was not associatedwith an impairment of the accuracy of endoscopic staging. The lesions thatinvade Sm1 are very difficult to define as such by endoscopy, which is why theyare categorized together with intramucosal lesions.
DISCUSSION
Endoscopic resection of early gastric cancer is thefirst option treatment in Japan and South Korea, and its use has been spreadingthroughout the world (9-11). Only lesions that have negligible oralmost negligible chance of nodal metastasis are the chosen for endoscopicresection, and the most important feature associated with that is the depth ofthe tumor invasion, since superficial lesions, wichcompromise only the mucosa or superficial submucosa, that meet other criteriaproposed by Gotoda etal., have low risk of nodal metastasis; however, when the lesionsinvade the deep submucosa (Sm2), the risk increases significantly.12For this reason, the prediction of tumor invasion or T staging of the EGC iscrucial, and in Japan and South Korea, it is usually done by endoscopy, butwithout standardized criteria or prospective research that support its use.
In our research, we showed that the endoscopicprediction of tumor invasion has a high accuracy, estimated at 98.2%, which ishigher than that reported in previous studies, in which it fluctuates between73 and 83% (19-24). This favorable difference is due to severalfactors: (1) The design of our research, in whichendoscopic prediction of tumor invasion categorizes the lesions in 3 groups:M-Sm1, Sm2 and indeterminate; this last group comprised only 9 lesions(13.85%), was not considered in the statistical analysis. A sub analysis of ourdata, classifying the lesions in only 2 groups, M-Sm1 and Sm2, would reduce ouraccuracy to 84.6%, a rate similar to that reported in previous studies incenters of great experience. (2) Being a prospective research, all lesions wereexamined in vivo, not in photos, allowing better discrimination of endoscopicfeatures, including rigidity, which we consider to be very important in theprediction of submucosal invasion. A recent study determined that the"non-extension sign" was associated with Sm2 invasion, with anaccuracy of 96.9% (29). (3) Endoscopistthat performed a training at the Keio University Hospital and at the NationalCancer Center, Tokyo-Japan, which gained experience by observing a large numberof gastric lesions and experts making an evaluation of them.
The M-Sm1 tumor invasion prediction showed highsensitivity, specificity, PPV and NPV (97.6%, 100%, 100% and 92.8%,respectively). Therefore, based on our research, this category in clinicalpractice would have clear indication of endoscopic resection. The prediction ofSm2 tumor invasion also showed high sensitivity, specificity, PPV and NPV(100%, 97.6%, 92.8% and 97.6%, respectively). Therefore, this group of lesionswould have justified indication of surgical resection in clinical practice.
The endoscopic features we are looking for to make theprediction of tumor invasion are simple and identifiable with conventionalendoscopy: size greater than 30 mm, irregular surface, marked margin elevation,central elevation of lesion with submucosal appearence, enlarged folds and rigidity. In the bivariateanalysis, all of them, except size, showed a significant association with Sm2invasion. The different Asian studies have also shown similar results (21-24,27,29). However, since our research is prospective,it allowed to assess the rigidity, not evaluated in previous studies, and thisis the characteristic that showed greater association with Sm2 invasion (OR6.4; 95% CI: 3.5-9.2).
In our research, prediction of tumor invasion wasperformed as follows: Mucosa (M-Sm1) when they had none of the above features;massive submucosa (Sm2) when it had two or more of these features;indeterminate when only had one endoscopic feature. Inmost Asian studies, only two categories, M and Sm2, are considered, and onefeature present is enough to consider the lesion as Sm2. However, we wanted toimprove the accuracy of the prediction, particularly the specificity of thegroup that is categorized as Sm2, with the main objective of not referringpatients to whom the surgical specimen finally reveals only M-Sm1 invasion,since they can benefit from endoscopic resection, which provides equal curerates, but with less invasiveness and a better quality of life. There is onlyone antecedent of a similar approach, in which Abe et al. perform a logistic regression analysisand mainly determined that tumor size greater than 3 cm, margin elevation,irregular surface, and marked redness are associated with Sm2 invasion. Then,they establish a diagnostic model in which they assign 2 points to the presenceof any of the first two features and one point for either of the last two;thus, lesions with 3 or more points evaluated by 3 different observers,categorize them as Sm2, with an accuracy of 82,5 to84.8%, specificity from 93.1 to 93.7%, but with a low sensitivity of 29.7 to45.9% (27). We believe that in our research the prediction of Sm2invasion is simpler, achieving a high specificity of 97.6%, and also a highsensitivity of 100%.
Like other studies, we found that some macroscopicfactors, such as the location of lesions in the upper third, the protruded ordepressed shape, size larger than 20 mm may affect the accuracy of theprediction (23,30). Regarding thehistological characteristics, we found that the accuracy was lower in Sm1lesions, which are very difficult to differentiate endoscopically, that's whythey are usually categorized together with intramucosallesions, since in addition their risk of nodal metastasis is negligible and thesuggested treatment in both cases is usually the same, endoscopic resection.However, we did not find that the accuracy was lower in the undifferentiatedtype, as other studies suggest (23,30).
The importance of our research is that, except for theAsian studies, it is the first one that evaluates the T staging endoscopicaccuracy through simple endoscopic features, and is also performed withconventional endoscopy (although with high resolution and definition). Inaddition, we sought a greater accuracy of the prediction of tumor invasion Sm2,which was a weak point in previous studies, not to send to surgery patients whocould benefit from ESD. We thus created a small sub group of indeterminateprediction, 9 cases that eventually all turned out to be M-Sm1 lesions. Webelieve this sub group deserves a special strategy and management in centers ofreference or greater complexity through: (1) Echoendoscopywith miniprobes: although most studies do notconclude on the benefits or advantages from the echoendoscopyover the conventional endoscopy in defining the T stage of the EGC, somestudies show that both techniques can be complemented, and even echoendoscopy can redefine the staging of the lesionscategorized as Sm2 by conventional endoscopy, so some algorithms suggest itsuse in this sub group of patients (19-21,24,29-31).
(2) Magnifying endoscopy: Kikuchi et al. showed that thepresence of dilated vessels within the lesions was associated with submucosalinvasion in 6/18 (33.3%), with diagnostic accuracy, sensitivity and specificityof 81.5%, 37.5%, and 88.3%, respectively (32). Other studies showthat features such as loss of glandular microstructure, scattered and scarcevessels, and multicaliber vessels were associatedwith Sm2 invasion (33-34). Then, if these indicators are present,the patient must be referred for surgery, and if they are not present, forendoscopic resection. (3) Diagnostic ESD: especially if the lessionis not very large, its location is affordable and there is a small risk ofcomplications.
The limitations of this research are to have beingperformed in a single center, by a single endoscopistwith training in Japan, and a not so large number of neoplastic gastriclesions. We suggest training in the evaluation of the endoscopic features ofgastric lesions directed to non-expert endoscopistsin different medical centers, using photos and videos, and then seek toreplicate the research, at a multicentric level andwith a large number of lesions, that also allow to perform a multi variateanalysis to define individual endoscopic features as predictors of Sm2invasion.
In conclusion, the endoscopic prediction of tumor invasión depth in early gastric neoplasia is highlyaccurate. In addition, marked margin elevation, central elevation, irregularsurface, enlarged folds, and rigidity are associated with massive submucosalinvasion.
Thanks: To Professor Naohisa Yahagi and Dr. Toshio Uraokafrom University Hospital of Keio
Conflict of interests: the authors have declared that no competing interests exist.
Funding: none.
BIBLIOGRAPHIC REFERENCES
1. Ministerio de Saluddel Perú. Análisis de la situación del cáncer en el Perú. Lima: MINSA; 2013. [ Links ]
2. Espejo H, NavarreteJ. Cáncer gástrico temprano: estudio de 371 lesiones en 340 pacientes en elhospital E. Rebagliati, Lima - Perú. Rev GastroenterolPeru. 2005;25(1):48-75. [ Links ]
3. Japanese Gastric Cancer Association. Japanese classification of gastriccarcinoma: 3rd English edition. Gastric Cancer. 2011;14(2):101-12. [ Links ]
4. Oda I, Saito D, Tada M, Iishi H, Tanabe S, Oyama T, et al. A multicenter retrospective study ofendoscopic resection for early gastric cancer. Gastric Cancer. 2006;9(4):262-70. [ Links ]
5. Isomoto H, Shikuwa S,Yamaguchi N, Fukuda E, Ikeda K, Nishiyama H, et al.Endoscopic submucosal dissection for early gastric cancer :a large-scale feasibility study. Gut. 2009;58(3):331-6. [ Links ]
6. Chung IK, Lee JH, Lee SH, Kim SJ, Cho JY, Cho WY, et al. Therapeutic outcomesin 1000 cases of endoscopic submucosal dissection for early gastric neoplasms:Korean ESD Study Group multicenter study. GastrointestEndosc. 2009;69(7):1228-35. [ Links ]
7. Min B-H, Kim ER, Kim KM, Park CK, Lee JH, Rhee PL, et al. Surveillance strategybased on the incidence and patterns of recurrence after curative endoscopicresection for early gastric cancer. Endoscopy. 2015;47(9):784-93. [ Links ]
8. Suzuki H, Oda I, Abe S, Sekiguchi M, Mori G, NonakaS, et al. High rate of 5-year survival among patients with early gastric cancerundergoing curative endoscopic submucosal dissection. Gastric Cancer. 2016;19(1):198-205. [ Links ]
9. Pimentel-Nunes P, Dinis-Ribeiro M, Ponchon T, Repici A, Vieth M, De Ceglie A, et al. Endoscopic submucosal dissection:European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy.2015;47(9):829-54. [ Links ]
10. ASGE Technology Committee; Maple JT, Abu Dayyeh BK,Chauhan SS, Hwang JH, Komanduri S, et al. Endoscopicsubmucosal dissection. GastrointestEndosc. 2015;81(6):1311-25. [ Links ]
11. Ono H, Yao K, FujishiroM, Oda I, Nimura S, YahagiN, et al. Guidelines for endoscopic submucosal dissection andendoscopic mucosal resection for early gastric cancer. Dig Endosc.2016;28(1):3-15. [ Links ]
12. Gotoda T, Yanagisawa A, Sasako M, Ono H, Nakanishi Y, Shimoda T, et al. Incidence of lymph node metastasisfrom early gastric cancer : estimation with a large number of cases at twolarge centers. Gastric Cancer. 2000;3(4):219-25. [ Links ]
13. Hirasawa T, Gotoda T, Miyata S,Kato Y, Shimoda T, Taniguchi H, et al. Incidence oflymph node metastasis and the feasibility of endoscopic resection forundifferentiated-type early gastric cancer. Gastric Cancer. 2009;12(3):148-52. [ Links ]
14. Park YM, Cho E, Kang HY, Kim JM. The effectiveness and safety of endoscopicsubmucosal dissection compared with endoscopic mucosal resection for earlygastric cancer: a systematic review and metaanalysis.Surg Endosc. 2011;25(8):2666-77. [ Links ]
15. Lian J, Chen S, Zhang Y, Qiu F. Ameta-analysis of endoscopic submucosal dissection and EMR for early gastriccancer. Gastrointest Endosc.2012;76(4):763-70. [ Links ]
16. Facciorusso A, Antonino M, Maso MDi, Muscatiello N. Endoscopic submucosal dissectionvs endoscopic mucosal resection for early gastric cancer: a meta-analysis.World J Gastrointest Endosc.2014;6(11):555-63. [ Links ]
17. ZhouY, Li XB. Endoscopic prediction of tumor margin and invasive depth in earlygastric cancer. J Dig Dis. 2015;16(6):303-10. [ Links ]
18. Pei Q, Wang L, Pan J, Ling T, Lv Y, Zou X. Endoscopicultrasonography for staging depth of invasion in early gastric cancer: ameta-analysis. J Gastroenterol Hepatol.2015;30(11):1566-73. [ Links ]
19. Yanai H, Matsumoto Y, Harada T, Nishiaki M, Tokiyama H, Shigemitsu T, et al. Endoscopic ultrasonography andendoscopy for staging depth of invasion in early gastric cancer: a pilot study.Gastrointest Endosc. 1997;46(3):212-6. [ Links ]
20. Yanai H, Noguchi T, Mizumachi S, Tokiyama H, Nakamura H, Tada M, et al. A blind comparisonof the effectiveness of endoscopic ultrasonography and endoscopy in stagingearly gastric cancer. Gut. 1999;44(3):361-5. [ Links ]
21. Lee JY, Choi IJ, Kim CG, Cho SJ, Kook MC, Ryu KW, etal. Therapeutic decision-making using endoscopic ultrasonography in endoscopictreatment of early gastric cancer. Gut Liver. 2016;10(1):42-50. [ Links ]
22. Sano T, Okuyama Y, Kobori O, Shimizu T, Morioka Y. Early gastric cancer.Endoscopicdiagnosis of depth of invasion. Dig Dis Sci. 1990;35(11):1340-4. [ Links ]
23. Choi J, Kim SG, Im JP, Kim JS, Jung HC, Song IS.Endoscopic prediction of tumor invasion depth in early gastric cancer. Gastrointest Endosc. 2011;73(5):917-27. [ Links ]
24. Tsujii Y, Kato M, Inoue T, Yoshii S, Nagai K, Fujinaga T, et al. Integrated diagnostic strategy for theinvasion depth of early gastric cancer by conventional endoscopy and EUS. Gastrointest Endosc. 2015;82(3):452-9. [ Links ]
25. Yao K. The endoscopic diagnosis of early gastric cancer. Ann Gastroenterol. 2013;26(1):11-22. [ Links ]
26. Yao K, Nagahama T, Matsui T, Iwashita A. Detectionand characterization of early gastric cancer for curative endoscopic submucosaldissection. Dig Endosc. 2013;25Suppl 1:44-54. [ Links ]
27. Abe S, Oda I, Shimazu T, Kinjo T, Tada K, Sakamoto T,et al. Depth-predicting score for differentiated early gastric cancer. GastricCancer. 2011;14(1):35-40. [ Links ]
28. TheParis endoscopic classification of superficial neoplastic lesions: esophagus,stomach, and colon. Gastrointest Endosc.2003;58(6 Suppl):S3-43. [ Links ]
29. Nagahama T, Yao K, Imamura K, Kojima T, OhtsuK, Chuman K, et al. Diagnostic performance ofconventional endoscopy in the identification of submucosal invasion by earlygastric cancer: the "non-extensionsign" as a simple diagnostic marker. GastricCancer. 2017;20(2):304-13.
30. Hizawa K, Iwai K, Esaki M, Matsumoto T, SuekaneH, Iida M. Is endoscopic ultrasonography indispensable in assessing theappropriateness of endoscopic resection for gastric cancer? Endoscopy. 2002;34(12):973-8. [ Links ]
31. Park CH, Lee SK. Understanding the role of endoscopic ultrasonography in earlygastric cancer. Gut Liver. 2016;10(1):3-5. [ Links ]
32. Kikuchi D, Iizuka T, HoteyaS, Yamada A, Furuhata T, Yamashita S, et al. Usefulness of magnifying endoscopywith narrowband imaging for determining tumor invasion depth in early gastriccancer. Gastroenterol Res Pract.2013;2013:217695. [ Links ]
33. Kobara H, Mori H, Fujihara S, KobayashiM, Nishiyama N, Nomura T, et al. Prediction ofinvasion depth for submucosal differentiated gastric cancer by magnifyingendoscopy with narrow-band imaging. Oncol Rep. 2012;28(3):841-7. [ Links ]
34. Yoshida T, Kawachi H, SasajimaK, Shiokawa A, Kudo S. The clinical meaning of anonstructural pattern in early gastric cancer on magnifying endoscopy. Gastrointest Endosc. 2005;62(1):48-54. [ Links ]
Correspondencia: Fernando PalaciosSalas Hospital Edgardo RebagliatiMartins
E-mail: vipasal.fp@gmail.com
Recibido: 1-11-2016
Aprobado: 4-1-2017