Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de Gastroenterología del Perú
Print version ISSN 1022-5129
Rev. gastroenterol. Perú vol.37 no.4 Lima Oct./Dec. 2017
ARTÍCULOS ORIGINALES
The effect of gluten-free diet among celiac patients aged 3-12 years old on BMI during 2006 to 2014 at Nemazee Teaching hospital
El efecto de la dieta libre de gluten en pacientes celiacos de 3 a 12 años en su índice de masa corporal desde el 2006 al 2014 en el hospital universitario de Nemazee
Seyed Mohsen Dehghani1, Sedighe Ostovar1, Maryam Ataollahi1, Hazhir Javaherizadeh2,3
1 Gastroenterohepatology Research Center, Shiraz University of Medical Sciences. Shiraz, Iran.
2 Nursing Care Research Center in Chronic Diseases, Ahvaz Jundishapur University of Medical Sciences. Ahvaz, Iran.
3 Research Center for Infectious Diseases of Digestive System, Ahvaz Jundishapur University of Medical Sciences. Ahvaz, Iran.
ABSTRACT
Introduction: Celiac disease (CD) is increasingly diagnosed and weight changes are common after adoption of a gluten- free diet (GFD) and there is concern that patients might gain further weight on a GFD. Objectives: This study examined to evaluate the impact of a GFD on the body mass index (BMI), whether favorable or unfavorable. Materials and methods: In this retrospective study, we reviewed electronic records of 44 patients with serologic study and intestinal biopsy confirmed CD who was visited in Nemazee hospital, Shiraz. All patients were put on GFD for 2 years and followed closely by pediatric gastroenterologist. BMIs were categories to four group underweight, normal weight, overweight and obese. Initial BMI and follow-up BMI was comparing together and also compared with general population. Result: At diagnosis, 27.27% of subjects were underweight, 63.64% normal and 9.09% were obese. On a GFD, 66.66% of underweight patients gained weight and became normal weight and 25% of normal weight and 75% of obese patients had increase weight; and the rest of the patients, BMI remained stable. The follow-up BMIs were statistically higher than initial BMIs (mean 17.17 vs. 15.62, p <0.0001). Conclusion: Individuals with celiac disease have lower BMI than the regional population at diagnosis. On the GFD, BMI is increased significantly in all categories.
Keyword: Celiac disease; Diet, gluten-free; Body mass index (source: MeSH NLM).
RESUMEN
Introducción: La enfermedad celiaca está siendo cada vez más diagnosticada y los cambios en el peso corporal son comunes después de ponerlos en una dieta libre de Gluten (DLG) y hay preocupación de que sigan ganando peso con esta dieta. Objetivos: Este estudio evalúa el impacto de la DLG en el índice de masa corporal (IMC) ya sea este favorable o desfavorable. Materiales y métodos: En este estudio retrospectivo se revisaron las historias de 44 pacientes con estudio serológico y biopsia intestinal confirmatorios de enfermedad celiaca que llegaron al hospital en Nemazee en Shiraz. A todos los pacientes se les puso en una DLG por 2 años y fueron seguidos por un gastroenterólogo pediatra. El IMC fue dividido en cuatro categorías, peso bajo, normal, sobrepeso y obesos. El IMC inicial y su seguimiento fueron comparados también con la población general. Resultados: Al momento del diagnóstico, el 27,27% estaban con bajo peso, el 63,64% con peso normal y el 9,09% eran obesos. En una DLG, el 66,66% de los pacientes con bajo peso llegaron a su peso normal y el 25% de los de peso normal y el 75% de los obesos también ganaron peso. Esta variación de IMC fue estadísticamente significativa (media 17,17 vs. 15,62, p <0,0001). El resto de los pacientes permaneció con su IMC estable. Conclusión: Las personas con enfermedad celiaca tienen el IMC más bajo que la población de la región al momento del diagnóstico. Puestas en la DLG, el IMC aumenta significativamente en todas las categorías.
Palabras clave: Enfermedad celíaca; Dieta sin gluten; Índice de masa corporal (fuente: DeCS BIREME).
INTRODUCTION
Celiac disease (CD) is an immune-mediated enteropathy caused by a chronic inflammatory response in small intestine to consumption of gluten in genetically susceptible patients (1). Gluten is a general term used to describe a mixture of storage proteins, including prolamins, hordeins and secalins found in wheat, barley and rye, respectively (2). Clinical presentations of the disease are widely various ranging from minor symptoms mimicking IBS or dyspepsia to classic symptoms of malabsorption; includes poor linear growth and poor nutritional status, abdominal pain and distension, diarrhea and iron deficiency anemia (3,4).
The availability of serological testing has seen a rise in individuals being diagnosed with atypical, minimal or no symptoms (5). Classical form of the disease is diagnosed using serologic and histopathologic tests. Anti-tissue transglutaminase antibody (anti-tTG) as the most sensitive and specific serologic study is used in suspected cases (6). A positive serologic study must be followed by endoscopy of small intestine that villous atrophy, crypt hyperplasia and increased intraepithelial lymphocytes (the threshold is >25 lymphocytes in 100 enterocytes) are the main histopathologic findings for confirmation of CD (7,8).
The prevalence of CD may be approximately 0.5-1% of the population, two thirds of whom remain undetected and is increasingly first diagnosed in adulthood (9-11). Diagnosis of CD can be delayed by low suspicion in patients with a normal or high body mass index (BMI) on initial presentation. Although, it is now well established that many CD patients have a high or normal BMI at diagnosis (11).
A lifelong strict gluten-free diet (GFD) is the only available treatment for CD (12). Adherence to GFD leads to regression of symptoms, normalization of histological and laboratory findings and reduces the risk of CD associated complications (8,13). Recently concern has increased for treated CD patients gaining weight on a GFD due to improved absorption of nutrients, as BMI above the normal range may increase the risk of morbidity i.e., metabolic syndrome, type II diabetes mellitus and a higher risk of vascular diseases and some forms of cancer (14,15). This concern over weight gain applies specially to screen-detected CD patients, and it would thus be essential to evaluate the consequences of gluten-free dietary treatment on the BMI of celiac patients today before any screening programs for the disease are instituted (15,16).
We performed this study to assess the distribution of BMI at diagnosis in CD patients aged 3-12 years old containing positive serologic study and small intestine biopsy and to evaluate the impact of two years on a GFD on the BMI of the patients. In addition, the BMI distributions at diagnosis and after treatment were compared with normal population curve in the same aged.
A systematic review and meta-analysis study was done in Iran to assess the prevalence of CD in March 2016. This study was conducted on seven publications with 9,720 subjects. Finally, they concluded CD prevalence among the Iranian population was approximately similar to that of the American and European populations (9).
Another study was done by Talaie to evaluate symptomatic and histopathologic response to GFD in patients with gluten-sensitive enteropathies in Iran in spring 2015. Thirty-five patients were put on GFD for 6 month and subjective symptomatic response, serology, endoscopy and histopathologic tests were repeated and compared with baselines and among groups that showed a significant increase in BMI as well as decrease all symptoms and histopathologic finding although these effects were more implications in CD than other gluten-sensitive enteropathies (8).
In the study by Kabbani et al. and colleagues to analyze changes in BMI in individuals after diagnosis of CD with attention to effects of GFD on weight change (15). A total of 679 patients BMI was recorded at initial and after 39.5 month and compared to regional population data. This study showed that CD patient has lower BMI than regional population at diagnose. On the GFD, main BMI increased and almost 38% of patient became overweight therefore weight maintenance counseling should be an integral part of celiac dietary education (15).
In a prospective study by Ukkola et al. and his group in Finland investigated weight and disease-related issues in 698 newly detected adults diagnosed due to classical or extra intestinal symptoms or by screening (17). They assessed the impact of a GFD on BMI at diagnosis and after one year on a GFD and compared with that in the general population. As a result, they found that BMI improved similarly in screen- and symptom-detected CD patients on a GFD and the celiac group had a more favorable BMI pattern than the general population. Favorable BMI changes were associated with subjects' self-rated expertise on GFD and young age at diagnosis, but not dietary counseling received (14).
Reilly et al described as the presentation of CD among children with a normal and an elevated BMI for age, and to study their BMI changes following a GFD in USA. One hundred forty-two patients were followed up for 35.6 months that after treatment significantly decreased BMI in overweight patients and increased BMI in normal and underweight patients (13% became overweight) were seen. Eventually they concluded both normal weight and overweight frequently occur in children with celiac disease and a GFD may have a beneficial effect upon the BMI of overweight and obese children with celiac disease (18).
In the study by Cheng et al. the effects of GFD on BMI were evaluated in 369 CD patients. Compared BMI at diagnosis and after 2.8 years showed that 66% of those who were underweight gained weight, whereas 54% of overweight and 47% of obese patients lost weight. So this dual effect of GFD adds to the impetus to diagnose celiac disease as well as expert dietary counseling was essential (19).
Another literature was done in October 2006 in United Kingdom to review BMI measurements and other clinical and pathological characteristics of 371 celiac patients presented with "classic" symptoms and compare BMI at diagnosis and after 2 years’ treatment in patients with serological support for dietary compliance, to assess response to gluten exclusion. They found a significant association between low BMI and female gender, history of diarrhea, reduced hemoglobin concentration, reduced bone mineral density (BMD), osteoporosis, and higher grades (subtotal/total) of villous atrophy while a greater percentage of patients had normal to obese BMI (95%) so failed or delayed diagnosis of CD may reflect lack of awareness of this large subgroup. Also this investigation showed that about 81% patients had gained weight after 2 years that according to high percentage of overweight patients this is a potential cause of morbidity, and the GFD as conventionally prescribed needs to be modified (20).
MATERIALS AND METHODS
The gastroenterology clinic of Nemazee Hospital of Shiraz University of medical sciences maintains a secure database of all patients with known or suspected CD, including demographic information, age and symptoms at diagnosis that was archived in Motahari and Imam Reza clinic. For shipping information, we referred to these places and after getting permission from staff studied the patient’s electronic records.
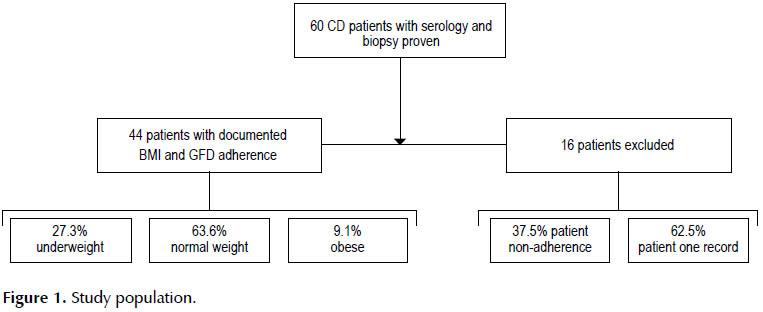
This study was carried out on 60 patients who referred to this center for evaluation of chronic significant complaints of low weight, greasy stool, diarrhea and excessive bloating from 2006 to 2014. The inclusion criteria included: age of 3 to 12 years old, GFD adherence, and available BMI measurements on at least two separate hospital visits. The sixteen patients were excluded from the study due to inaccessibility to patient records, being non-compliance and having only one record BMI.
Electronic records of 44 patients with serologic study and intestinal biopsy confirmed CD was reviewed retrospectively. All patients were followed closely by gastroenterologist, seen after diagnosis, at 3 and 6 months. Data regarding age of diagnosis, sex, height, weight, and BMI prior to the diagnosis of CD and after initiation of GFD in follow up was collected and patients was checked for GFD adherence.
BMI defined as body weight (kg)/height (m2) was recorded before and after gluten exclusion. BMI values were categorized according to the World Health Organization BMI for age percentile. BMIs for each percentile from 3 to 12 years old were excluded and mean BMIs were measured for the same percentile and were categorized as follows: <14 kg/m2 (<percentile 5th) as underweight, 14-18.2 kg/m2 (percentile 5th-85th) as normal weight, 18.2-20.14 kg/m2 (percentile 85th- 95th) as overweight and ≥20.4 kg/m2 (>percentile 95th) as obese. Changes in BMI were classified as favorable if underweight patients gained weight, those with normal weight remained normal and overweight and obese patients lost weight. Weight gain was rated unfavorable if overweight or obese patients gained weight and those with normal weight became overweight or obese.
Baseline BMIs were compared with followed up BMIs and also both were compared with general population in our retrospective study. On the other hand, we assessed the correlation between sex and BMIs and BMI changes.
Ethical consideration: This study was approved by ethical committee of the Shiraz University of Medical Sciences (EC-P-92-6229).
Statistical analysis:
Statistical analysis was performed using SPSS version 18. Study outcomes (height, weight, and BMI prior to the diagnosis of CD and after two years from initiation of GFD) were assessed using paired t-test. p <0.05 was considered significant.
Study population:
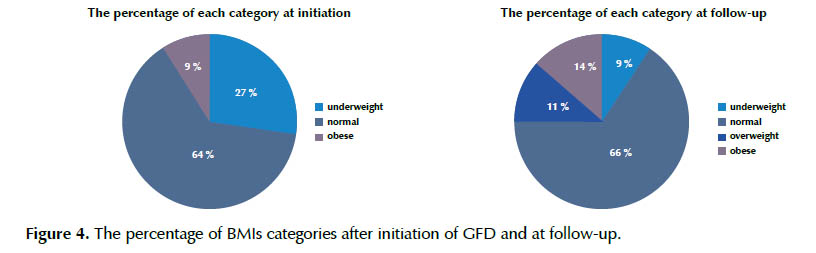
Of the 60 patients with serology and biopsy proven CD, 44 (73.3%) had documentation of BMI and GFD adherence from at least two separate visits. Ten (16.66%) patients had only one recorded BMI and 6 (10%) patients either had no records of BMI or had no assessment of their adherence with a GFD and were not included in analysis (Figure 1). Consistent with all the studies, females predominated (68.5%). The mean±SD BMI before GFD was 15.62±3.47; median 15.07 and range: 8.01 to 29.52. The mean duration of follow up was 2 years. The majority (63.6%) were normal weight, whereas 27.3% was underweight and 9.1% had obesity.
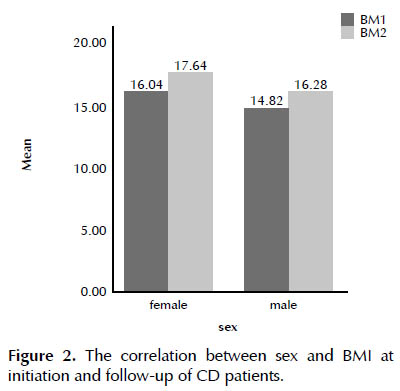
Among the CD patients, females were predominant. The correlation test was done for comparison the male and female BMI and BMI changes that showed that females had greater weight than males (Figure 2).
Comparison to general population
In comparing the CD patients BMI to general population, we saw mean our patients BMI was less than normal percentile (15.6 vs. 16.1).
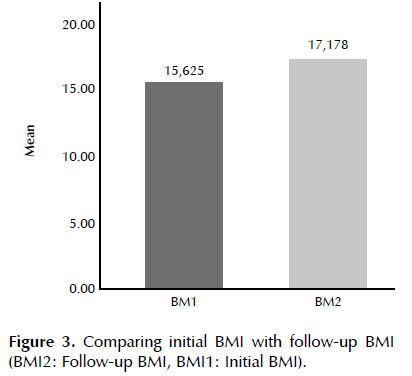
The mean time between first and last BMI was 24 months. First normality test was done for data that showed our data was normal (p-value <0.0001). Individual patients’ follow-up BMIs were statistically higher than initial BMIs (mean 17.17 vs. 15.62, p <0.0001) (Figure 3).
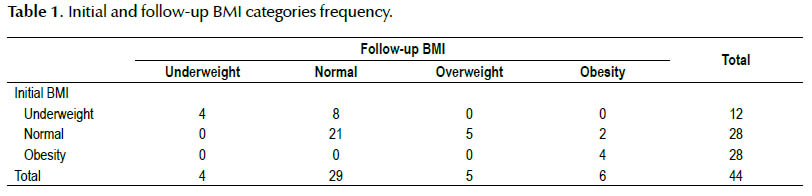
Changes at follow-up for the subjects by BMI category are shown in Table 1. This table shows the frequency of CD patients in each category. The most changes were seen in underweight category with 8 patients became normal weight whereas all obese patients stayed in the same category.
In patients who were underweight at diagnosis, the majority (66.66%) attained a normal weight at follow-up, whereas 33.33% remained underweight. None of patients became overweight or obese. Drop in the total percent of underweight patients from baseline to follow-up was 18.2%.
In the normal weight category, 75% remained in the same weight class. However, 17.8% became overweight and 7.14% also became obese. Overall 2.3% increasing was seen in this category.
Of those patients starting in the obese category, very few managed to leave this category, as 100% remained obese at follow-up. In this group, 75% of patients had increasing in BMI and only 25% lost their weight.
Overall all patients in each category trend were to increase weight that some patients have changed their categories although the majority stayed in the same categories.
We show the percentage of BMIs categories after initiation of GFD and at follow-up in Figure 4.
DISCUSSION
Our retrospective research surveyed better comparison between celiac patients and general population BMI and characterize the effect of CD on body weight in the celiac population, also showed the effect of GFD on celiac patients BMI over 2 years.
While it is well known that children with CD can have weights ranging from underweight to obese, less is known about the effect treatment has on weight status. An increase in the diagnosis of celiac disease in children with normal weight, overweight, and obesity may be reflective of both the shift in disease presentation and the effect of serological screening (18,19). The majority of patients in our study had a normal BMI at diagnosis and recent studies have shown that children with celiac disease commonly present with a normal body weight. The relative paucity of those with a low BMI (underweight category) emphasizes the fact that celiac disease should not be considered primarily as a malabsorption disorder in which patients may present underweight or malnourished although we know the CD as disease with decreased intestinal absorption and decreased food intake due to gastrointestinal symptoms including bloating, nausea and abdominal pain (15,21).
Although a GFD improves the weight and body composition of children with CD who are underweight at diagnosis (19,22), there are few data regarding growth outcomes of children with CD with a normal or elevated BMI (18,21). Recent studies of patients in the United States with CD have shown a beneficial effect of the GFD regardless of BMI at diagnosis: obese patients lost weight, whereas underweight patients gained weight (23). Overall, by comparing to general population, celiac population BMIs were lower than normal population. As might be expected, we found that the mean BMI of the celiac patients significantly increased after initiating GFD. The largest improvement in BMI (weight gain) was for the underweight group. Among the underweight patients in our study, 66.66% increased BMI that became normal weight and 33.33% stayed in the same category (91.66% had increase BMI) therefore we have demonstrated that most patients’ weight gain was common and increases with duration of follow-up and the BMI category was changed (20).
While weight gain on the GFD may be welcome for those who are underweight at diagnosis similar weight gain may be very unwelcome in those who begin at a normal weight or who are overweight or obese at diagnosis that our CD patients in each category had trend to increase body weight and BMI. BMI increases, whether desired or undesired, after treatment of childhood CD are probably multi-factorial. Improved absorption likely plays a significant role also lifestyle factors, particularly dietary choices, have been shown to play a role (15,24).
Although our investigation showed that CD patients BMIs improve after starting GFD but we also consider that this weight gain can be cause of becoming overweight or obese as we saw 15.91% increasing in these groups. Therefore, a better dietary counseling is needed for control and management of CD patients. Prospective studies to identify the factors responsible for these BMI alterations are warranted, with particular attention paid to macronutrient intake and activity (25).
Dietary management is the only therapy for CD (26). Expert dietary counseling is important, because not only do patients need to be aware of what they have to avoid but they need to be able to make wise food choices among the gluten-free foods (27). It is our practice to advise patients about a high quality GFD in which naturally gluten-free foods (e.g., fruit and vegetables) and use of alternate, non- gluten containing grains (e.g., quinoa and buckwheat) (28). There is unfortunately a paucity of dietitians available for the management of CD. The importance of expert dietary guidance is demonstrated in this study due to BMI changed reflecting to gluten avoidance (29,30).
To our knowledge, our study is retrospective to evaluate the effect of the GFD on BMI in patients with CD with statistical validation. Also we assessed the influence of GFD, as recorded by gastroenterology, on weight class and weight change, we had some limitations. First, adherence data from an expert celiac dietitian, currently regarded as the gold standard for adherence measurement, were not always available. Second, GFD adherence could only be assessed at the time of clinic visits, and variation in adherence overtime, which may be significant, was not evaluated. This highlights a general need, in patient management and in research, to develop more comprehensive and standardized methods to measure adherence with the GFD over time. Finally, regional differences in BMI are common and our results may not be fully applicable to other populations, owing to differences in dietary profiles, exercise, means of transportation and other variables.
Limitation: Single center study and low sample size are the main limitation. Retrospective method is another limitation of the current study.
In conclusion, normal and low BMI are frequent presenting growth profiles among our patients with CD. The GFD has a beneficial effect upon the BMI of underweight children with CD (66.66% became normal weight). It is unclear whether the diet is responsible for unwanted BMI increases among children with a normal and elevated BMI. Prospective controlled studies of the effects of the GFD upon the growth patterns and dietary choices of children with CD are warranted.
Acknowledgment:
This thesis is supported by deputy dean of school of medicine based on research project number 92-01-21- 6229 dated February 18, 2016 and sponsored by deputy chancellor of Shiraz University of Medical Sciences.
Conflict of interests: The authors have declared that no competing interests exist.
Funding: The article did not have any type of financing for its realization.
BIBLIOGRAPHIC REFERENCES
1. Niewinski MM. Advances in celiac disease and gluten-free diet. J Am Diet Assoc. 2008;108(4):661-72. [ Links ]
2. Rodrigues AF, Jenkins HR. Investigation and management of coeliac disease. Arc Dis Child. 2008;93(3):251-4. [ Links ]
3. Fasano A. Clinical presentation of celiac disease in the pediatric population. Gastroenterology. 2005;128(4):S68-S73. [ Links ]
4. Schuppan D, Dennis MD, Kelly CP. Celiac disease: epidemiology, pathogenesis, diagnosis, and nutritional management. Nutr Clin Care. 2005;8(2):54-69. [ Links ]
5. Fasano A, Catassi C. Current approaches to diagnosis and treatment of celiac disease: an evolving spectrum. Gastroenterology. 2001;120(3):636-51. [ Links ]
6. Petrarca L, Nenna R. Coeliac disease and obesity in children. J Pediatr Gastroenterol Nutr 2015;61(1):e4. [ Links ]
7. Pelkowski TD, Viera AJ. Celiac disease: diagnosis and management. Am Fam Physician. 2014;89(2):99-105. [ Links ]
8. Talaie R. Does gluten free diet have more implications than treatment of coeliac disease? Gastroenterol Hepatol Bed Bench. 2015;8(2):160-6. [ Links ]
9. Ahadi Z, Shafiee G, Razmandeh R, Keshtkar AA, Sani MN, Azemati B, et al. Prevalence of celiac disease among the Iranian population: A systematic review and meta-analysis of observational studies. Turk J Gastroenterol. 2016;27(2):122-8. [ Links ]
10. White LE, Merrick VM, Bannerman E, Russell RK, Basude D, Henderson P, et al. The rising incidence of celiac disease in Scotland. Pediatrics. 2013;132(4):e924-31. [ Links ]
11. El MouzanMI, Al Mofarreh MA, Saadah OI, Al-Hussaini AA, Al-Saleem KA, Al Mehaidib AI. Impact of pediatric inflammatory bowel disease on linear growth: Data from a national cohort study in Saudi Arabia. Saudi J Gastroenterol. 2016;22(2):106-8. [ Links ]
12. Bioletti L, Capuano M, Vietti F, Cesari L, Emma L, Leggio K, et al. Celiac disease and school food service in Piedmont Region: Evaluation of gluten-free meal. Ann Ig. 2016;28(2):145-57. [ Links ]
13. Kupper C. Dietary guidelines and implementation for celiac disease. Gastroenterology. 2005;128(4):S121-7. [ Links ]
14. Ukkola A, Mäki M, Kurppa K, Collin P, Huhtala H, Kekkonen L, et al. Changes in body mass index on a gluten-free diet in coeliac disease: a nationwide study. Eur J Intern Med. 2012;23(4):384-8. [ Links ]
15. Kabbani T, Goldberg A, Kelly C, Pallav K, Tariq S, Peer A, et al. Body mass index and the risk of obesity in coeliac disease treated with the gluten-free diet. Aliment Pharmacol Therap. 2012;35(6):723-9. [ Links ]
16. Mahadev S, Simpson S, Lebwohl B, Lewis SK, Tennyson CA, Green PH. Is dietitian use associated with celiac disease outcomes? Nutrients. 2013;5(5):1585-94. [ Links ]
17. Ukkola A, Maki M, Kurppa K, Collin P, Huhtala H, Kekkonen L, et al. Changes in body mass index on a gluten-free diet in coeliac disease: a nationwide study. Eur J Intern Med. 2012;23(4):384-8. [ Links ]
18. Reilly NR, Aguilar K, Hassid BG, Cheng J, DeFelice AR, Kazlow P, et al. Celiac disease in normal-weight and overweight children: clinical features and growth outcomes following a gluten-free diet. J Pediatr Gastroenterol Nutr. 2011;53(5):528-31. [ Links ]
19. Cheng J, Brar PS, Lee AR, Green PH. Body mass index in celiac disease: beneficial effect of a gluten-free diet. J Clin Gastroenterol. 2010;44(4):267-71. [ Links ]
20. Dickey W, Kearney N. Overweight in celiac disease: prevalence, clinical characteristics, and effect of a gluten-free diet. Am J Gastroenterol. 2006;101(10):2356-9. [ Links ]
21. Valletta E, Fornaro M, Cipolli M, Conte S, Bissolo F, Danchielli C. Celiac disease and obesity: need for nutritional follow-up after diagnosis. Eur J Clin Nutr. 2010;64(11):1371-2. [ Links ]
22. Szajewska H, Shamir R, Chmielewska A, Piecik-Lech M, Auricchio R, Ivarsson A, et al. Systematic review with meta- analysis: early infant feeding and coeliac disease–update 2015. Aliment Pharmacol Therap. 2015;41(11):1038-54. [ Links ]
23. Hörnell A. Effects of a gluten-free diet on gastrointestinal symptoms in celiac disease. The Am J Clin Nutr. 2005;81(6):1452-3. [ Links ]
24. Theethira TG, Dennis M. Celiac disease and the gluten-free diet: consequences and recommendations for improvement. Dig Dis. 2015;33(2):175-82. [ Links ]
25. Pietzak MM. Follow-up of patients with celiac disease: achieving compliance with treatment. Gastroenterology. 2005;128(4):S135-41. [ Links ]
26. Brambilla P, Picca M, Dilillo D, Meneghin F, Cravidi C, Tischer M, et al. Changes of body mass index in celiac children on a gluten- free diet. Nut Metab Cardiovasc Dis. 2013;23(3):177-82. [ Links ]
27. Mulder C, Wierdsma N, Berkenpas M, Jacobs M, Bouma G. Preventing complications in celiac disease: Our experience with managing adult celiac disease. Best Pract Res Clin Gastroenterol. 2015;29(3):459-68. [ Links ]
28. Newnham ED, Shepherd SJ, Strauss BJ, Hosking P, Gibson PR. Adherence to the gluten-free diet can achieve the therapeutic goals in almost all patients with coeliac disease: A 5-year longitudinal study from diagnosis. J Gastroenterol Hepatol. 2016;31(2):342-9. [ Links ]
29. Singh P, Sharma PK, Agnihotri A, Jyotsna VP, Das P, Gupta SD, et al. Coeliac disease in patients with short stature: A tertiary care centre experience. Natl Med J India. 2015;28(4):176-80. [ Links ]
30. Plesa A, Lepsa A, Crumpei F. [Adult celiac disease (gluten enteropathy]. Rev Med Chir Soc Med Nat Iasi. 2002;107(2):405-10. [ Links ]
Correspondence:
Hazhir Javaherizadeh
Dept. of Pediatric Gastroenterology, Abuzar Children’s Hospital, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
E-mail: Hazhirja@yahoo.com, Hazhirja@ajums.ac.ir
Recibido: 2-3-2017
Aprobado: 27-7-2017