INTRODUCTION
Autoimmune hepatitis (AIH) is a liver disease of children and adults all over the world 1. It affects patients of any age, sex, race or ethnicity, with more prevalence in females 2,3 and considered as one of the most common causes of chronic liver disease in teenagers and children 4. AIH is an auto-inflammatory disease of liver which occurs mostly in children with genetic susceptibility 1,3. There may be a positive history of other autoimmune diseases in families of children with AIH 4. Environmental factors such as drugs or viruses can trigger autoimmune response to liver autoantigens, which will cause hepatocyte damage in presence of immunoregulatory impairment 5 .
Although the exact incidence rate of autoimmune hepatitis in Iran is unknown 6 according to a study in our center it is the fourth cause of cirrhosis in children in southern Iran (11.3% of cases) 7. It can present with a wide spectrum presentations, from mild non- specific symptoms to acute, chronic or fulminant hepatitis, or isolated liver enzyme elevation without any symptoms 2,8-10. So early diagnosis and treatment needs high suspicion 11 . There is a diagnostic criteria for AIH in children published in 1993 12 that was revised and simplified in 2008 (13). It is the most accepted criteria of AIH worldwide, although there are some doubts about its accuracy especially in children 14. AIH can be well controlled with immunosuppressive therapy with prednisolone and azathioprine if started early at the beginning of the disease 15-17. Although, there are some patients presenting with fulminant hepatitis or progressing into uncompensated cirrhosis with poor response to treatment, who need liver transplantation 18,19.
The aim of this study was to evaluate presentations, laboratory data, treatment and prognosis of patients with AIH in our center, Pediatric Hepatology ward of Nemazee hospital, as the referral center of children liver diseases and liver transplantation in Iran.
MATERIALS AND METHODS
We evaluated 86 patients treated and followed with final diagnosis of AIH in Pediatric Hepatology ward of Nemazee Hospital, Shiraz University of Medical Sciences, Iran, as a tertiary center, admitted between years 2010 to 2018. They were between 1 to 18 years old, both males and females were included. They were not known case of any other chronic liver disease. Children with Wilson’s disease or other liver diseases are excluded. Patients with scleorsing cholangitis were excluded. The diagnosis was established according to the International Autoimmune Hepatitis Group Scoring system (IAIHGS) published in 1993 and Simplified Criteria for the Diagnosis of Autoimmune Hepatitis in 2008 (12, 13). Patients with other acute liver diseases such as viral hepatitis and other inflammatory diseases were excluded from our study group. Physical findings including jaundice, liver size, splenomegaly and encephalopathy were recorded according to physicians admission notes in medical files. The laboratory tests, including serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, globulin and auto antibody tests, including immunofluorescence for ANA and ASMA and enzyme immunoassay for anti LKM-1 antibody were extracted from the medical files. The reference values for ALT and AST were considered from 10 to 37 U/L and for serum Albumin 3.8-4.2 and serum globulin levels 1.0 to 1.5 g/dL. Anti LKM and ASMA titer ≥ 1:20 and ANA ≥1:40 were considered positive according to the manufacturer guideline.
Percutaneous needle liver biopsy was done in patients before starting medical therapy if the patients’ general condition and hemostatic stability, ascites and coagulopathy profiles permitted. The treatment regimen was started with prednisolone, given at 1 to 2 mg/kg/ day (maximum 60 mg/day) and azathioprine given at 1.5 mg/kg/day (maximum 100 mg/day). AST and ALT were checked weekly up to 4-6 weeks and if they were decreased, prednisolone was tapered 5 mg each week, up to the minimum maintenance dose keeping patient in remission according to symptoms and biochemical profile (5-7.5 mg per day). Then prednisolone was discontinued after 6 months and azathioprine was continued. Treatment response was assessed according to the criteria established by 1993 IAIHGS after 2.5-3 years. Then if the patient was asymptomatic and also liver enzymes were in normal range, liver biopsy was repeated. In those cases with mild inflammatory or normal liver histology, medical treatment was discontinued with close follow up. If the patient had disease flare-up then medical therapy was restarted. For non-responding patients to the above mentioned treatment and progressing to decompensated cirrhosis or patients presenting with fulminant hepatitis, liver transplantation was considered.
All the results were expressed as mean (±standard deviation). The data were statistically analyzed using Student’s t-test and Chi-Square test. In addition, p values less than 0.05 were considered as statistically significant. The statistical analysis was done using the SPSS statistical software (version 22).
This study was approved by ethical committee of the Shiraz University of Medical Sciences.
RESULTS
We included 86 subjects (male=29, female=57) with final diagnosis of AIH. The mean age was 9.10±4.36 years old (range 1 to 18 years old). Among 86 cases, 42 patients were known case of AIH referred to this center (including 20 patients directly referred for liver transplantation), and 44 patients were new cases.
We considered four main items in physical examination including jaundice, hepatomegaly, splenomegaly and encephalopathy. Twenty one of 86 patients had jaundice (75.6%), 40 of 86 patients had hepatomegaly (46.5%), 28 of 86 patients had splenomegaly (32.6%) and 15 of 86 patients had encephalopathy (17.4%). 42 out of 86 patients were known case of AIH referred to our center and 44 patients were new cases on admission.
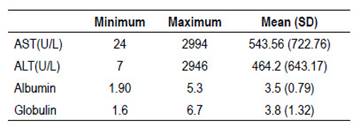
The paraclinical findings that we evaluated in this study are shown in Table 1.
Liver enzymes including AST and ALT were increased more than 3 times normal range in 53/86 (61.6%) and 48/86 (55.81%), respectively. AST was elevated more than 1000 U/L in 19/86 (22.1%) and ALT was more than 1000 U/L in 13/86 (15.1%).
Autoantibodies were available in 53/86 patients, including Anti LKM-1 antibody, ASMA and ANA which were positive in 4/53 (7.5%), 8/53 (15.1%) and 8/53 (15.1%) patients. 36/53 (67.9%) patients had seronegative AIH, 13/53 (24.5%) had AIH-I and 2/53 (3.8%) had AIH-II. Two patients had simultaneous Anti LKM and ANA positive, not included in these three categories.
Histopathologic evaluation of liver biopsies was mainly categorized in three groups: autoimmune hepatitis (AIH) in 33 of 70 (47.1%); chronic hepatitis in 19 of 70 (25.1%); and cirrhosis in 18 of 70 (25.7%).
Liver biopsy results were not available in 16 patients due to their unstable condition or bleeding tendency, which did not let the procedure to be done.
Treatment with prednisolone was started after confirmation of diagnosis, for 80 of 86 (93%) patients.
Azathioprine was added in 56 of 86 (65.11%) patients. 53 of 86 (61.6%) patients had initial remission after 4-6 weeks of starting immunosuppressive therapy, became symptom free with normal liver enzymes. Overall, 26 of 86 (30.2%) patients needed liver transplantation. Twenty patients were known cases of AIH on medical treatment (referred here from other centers specifically for being transplanted), and 6 patients needed transplantation due to fulminant hepatitis. Treatment was stopped in 11 of 86 (13.7%) patients after normalization of symptoms, liver enzymes and pathology.
Regarding physical findings, size of liver was significantly related to liver transplantation (p-value 0.02). 41.3% of patients with normal liver size led to liver transplantation, maybe due to their end stage liver disease causing liver shrinkage. While only 15.0% of patients who had hepatomegaly needed transplantation. Encephalopathy was also significantly related to final outcome (p-value= 0.03). Eight of 15 (53.3%) patients with encephalopathy didn’t have enough response to medical treatment, 4 (26.7%) leading to transplantation and 4 (26.7%) patients expired. While 46/71 (64.8%) patients without encephalopathy had acceptable response to treatment and 21/71 (29.6%) led to transplantation. Mortality in patients without encephalopathy was 4/71 (5.6%).
Among liver enzymes elevated ALT was significantly related to final outcome (p-value=0.02). 38/86 (44.2%) had ALT 10-150 U/L, 35/86 (40.7%) had ALT 150-1000 and 13/86 (15.1%) had ALT>1000. In ALT 10-150 patients 20/38 (50.2%) had response to treatment while 17/38 (44.73%) patients led to transplantation and only 1/38 (1.2%) patient expired. While in ALT 150-1000 U/L group, 23/35 (65.71%) patients had response to treatment, 7/35 (20%) needed transplantation and mortality rate was 5/35(14.28%). In patients with ALT>1000 U/L, 10/13 (76.92%) had response to medical treatment, 1/13 (7.7%) was transplanted and 2/13 (15.38%) expired, which seems to be the best prognosis between different ALT levels in patients.
AST, autoantibodies and AIH type were not significantly related to final outcome (p-value>0.05).
Totally 9 patients (10.5%) expired, 8 of them were due to severity of liver disease and hepatic encephalopathy and 1 due to post liver transplant complications.
DISCUSSION
AIH is a chronic inflammatory liver disease mostly involving patients in second and third decade of life with peak age around 10 years old in children 6,8. Mean age in our study group was 9.10±4.36 years which was compatible with other studies in children with AIH 8,20,21. AIH is known as a disease with female predominance 4,6,8,20,22, which was noticed in our study too, with 66.27% females and 33.72% males.
Symptoms and signs can present differently in severity in patients with AIH, varying from asymptomatic to fulminant hepatic failure 3,4,23. Although asymptomatic patients include a noticeable population (34-45% in Manns MP et al.) when the disease is diagnosed, they should be closely followed for symptoms presentations, as most of them (more than 70% in Manns MP et al.) will become symptomatic during disease course 18. Several studies reported that most of the patients present with viral hepatitis like illness 8,24.
We reported four major symptoms and signs which were reported with more frequency in other studies 3,8 including jaundice (75.6%), hepatomegaly (46.5%), splenomegaly (32.6%) and encephalopathy (17.4%).
In current study compared with the previous study in our center by Dehghani et al., splenomegaly had lower frequency, but jaundice and hepatomegaly were the most frequent physical findings, found in about half of patients of both studies8. In the study by Yassin et al., jaundice was the most frequent sign and was seen in 97% of the children with AIH 25.
In our subjects, among physical findings size of liver and encephalopathy were significantly related to final outcome. Size of liver had a significant relationship with need to transplantation, with p-value 0.02. 41.3% of patients without hepatomegaly needed liver transplantation, maybe due to their end stage liver disease causing liver shrinkage. While only 15.0% of patients who had hepatomegaly needed transplantation. Encephalopathy was also significantly related to final outcome.
Eight of 15 (53.3%) patients with encephalopathy didn’t have enough response to medical treatment, 4 (26.7%) leading to transplantation and 4 (26.7%) expired. While 46/71 (64.8%) patients without encephalopathy had acceptable response to treatment and 21/71 (29.6%) led to transplantation. Mortality in patients without encephalopathy was 4/71 (5.6%). The relationship between first physical findings and final outcome was not evaluated in other similar studies.
Liver enzymes including AST and ALT are suitable markers for showing liver damage in different stages of the disease, increasing more than 1000 U/L in patients presenting with acute fulminant liver failure, and decreasing to normal range in remission time or patients with cirrhosis, which shows the importance of attention to clinical and other para-clinical findings for differentiating activity or chronicity of the disease 26. In our study group AST and ALT were detected in a wide range due to their different stages of liver disease. Elevated AST and ALT more than three times the upper limit of normal was detected in 61.6% and 55.8% of patients, respectively. We noticed significant increase in AST and ALT, more than 1000 in 22.1% and 15.1% of patients, respectively. While this significant elevated transaminase levels is considered as a paraclinical sign of viral, drug or toxin induced hepatitis or liver hypoxemia and hypoperfusion (shock liver) 26. Among liver enzymes only elevated ALT was significantly related to final outcome (p-value=0.02). Most of the patients (44.2%) had ALT 10-150, 40% had ALT 150-1000, and only 15% had ALT>1000. The majority of the patients with ALT 150-1000 had response to medical treatment (65.71%), while in ALT 10-150 about 50% had enough response to medication.
As AIH is an autoimmune disease, autoantibodies are considered an important part of diagnostic criteria 12,13. There are different autoantibodies which can have increased titers in AIH such as anti-nuclear antibody (ANA), anti-smooth muscle antibody (ASMA), anti- liver kidney microsomal antibody type 1 (anti-LKM-1), antibody to soluble liver antigen (SLA), autoimmune polyendocrinopathy-candidias-ectodermal dystrophy antibody (APECED), antibody to asialoglycoprotein receptor (ASGPR), liver cytosol type 1 antibody (LC1), perinuclear anti-neutrophil cytoplasmic antibody (p-ANCA) and anti-mitochondrial antibody (AMA) 27. Diagnostic criteria and worldwide guidelines come to an agreement to use three of these autoantibodies, including ANA, ASMA and anti LKM-1, which are more helpful and sensitive for liver diseases, although not very specific (12,18,24). ANA can be positive in chronic viral hepatitis B and C, and ASMA can be observed in these viral hepatitis, non-autoimmune inflammatory diseases of liver and autoimmune diseases of other organs rather than liver 8,18. Anti LKM-1 can be detected in chronic hepatitis C 26. So it seems logical to make the diagnosis in combination with other parameters including clinical, biochemical and histological findings rather than autoimmune antibodies alone 8,12,18. According to detected autoantibodies AIH is classified into two types, and when there is no detectable autoantibody it is defined as seronegative AIH1,19,28. Autoantibodies were not available in 16/86 of our subjects, in rest of the patients frequency of AIH type 1 and 2 were 24.5% and 3.8% respectively, and 67.9% were seronegative; which seems not compatible with other studies. Regarding Dehghani SM et al. study in 2012 frequency of AIH types has changed during recent 8 years, as only 29.9% of their patients were seronegative (8). In another study on AIH in Iranian children by Rafeey M et al. in 2007 about 11% of patients were seronegative 21. In North America and European countries 60-70% of patients have AIH-1 and 10-15% have AIH-2 28-30.
All of our patients had fulfilled 2008 simplified diagnostic criteria for AIH with score ≥6. Liver biopsy is a determinative factor in diagnosis according to the criteria, with 1 to 2 points in scoring 13. Our patients had histopathologic findings typically in favor of autoimmune hepatitis, or chronic hepatitis and cirrhosis. Interface hepatitis is the hall mark in histology, in favor of hepatitis, but fibrosis and cirrhosis are not uncommon in first presentation of the disease 18. Cirrhosis in first presentation of AIH is reported in a study in UK in 14% of patients by Deneau et al. 31, in 38% of Caucasian children with AIH by Oettinger et al. 32 , in about 50% of AIH patient in north west of Iran, by Rafeey et al. 21 and previously in our center in 25.3% of patients by Dehghani et al. 8. In our study group 25.7% of patients had cirrhosis in first liver biopsy which was compatible with previous findings in our center.
Treatment with prednisolone and azathioprine in patients with possible or definite AIH is the first choice, although there are different protocols 15,16,18,24, and good response to treatment was considered as a diagnostic criteria in 1993 IAIHGS 12. Goals of treatment are to make the patient symptom free with complete normalization of liver enzymes, autoantibodies and liver histology, which if it is not achieved can lead to cirrhosis 4. In a study in Australia in 2001 by Saadah et al. 50% of patients had good response to medical therapy 33. Chai PF et al. reported a 68% initial response to treatment in Malaysia in 2010 19. Ngu et al. had 65% good response in 2013 34. Deneau M et al. reported 76.4% remission rate in Canada in 2014 31. Previous study in our center in 2012 showed 59.8% remission with treatment 8, which was compatible with our results, 61.6% remission in response to standard therapy. Overall it seems that response to medical treatment had improvement during recent years. Withdrawal of immunosuppressive therapy was tried for selected patients who achieved and maintained remission for 2 to 3 years 18,24 . Overall in our study treatment cessation was successful in 11 of 80 patients (13.7%), which was lower than previous study by Dehghani et al. with 38% successful medication withdrawal 8, and a study in Canada by Deneau et al. that could discontinue medication for 41.6% of their patients 31. Sheiko et al. also reported 18% successful immunosuppressive withdrawal 17.
Liver transplant is the treatment of choice if the patient didn’t respond to medical therapy, progressing to end stage liver disease, or in patients with fulminant hepatitis 35. Different centers reported different rates of liver transplantation: 18% in Chai PF et al. study 19 , 6.2% in Lee WS et al. study 22, 6% in Sheiko et al. study 17 and 27.6% in Dehghani SM et al. study 8. In another study from Canada, among 159 children with AIH, 9 children underwent Liver transplantion and response of treatment at one year was 90% 36. Our transplanted patients were (26 of 86) 30.2% of total, compatible with previous study, and higher than other studies worldwide, maybe because Nemazee hospital is the only referral center for children liver transplantation in Iran. Therefore, most of the patients of this group, 20 out of 26 patients, were referred here from other centers.
Totally 9 patients (10.5%) expired in current study, 8 of them due to severity of liver disease and hepatic encephalopathy and 1 due to post liver transplant complications, which is increased compared to previous study by Dehghani et al. with mortality rate of 4.6% 8. Mortality rate in other studies were more than our center: 21.8% in Lee et al. study 22, 20% in Ngu et al. study 34, which may be due to liver transplantation facilities available in our hospital.
It seems that chronicity of liver disease and liver shrinkage due to that will lead to poorer response to medication, which maybe is the reason for higher need to transplantation.
Higher mortality was seen in patients with extremely elevated ALT which may be due to their instability that does not permit optimum medical or surgical interventions to be done at early stages of presentation. This means that patients with AIH and very high levels of ALT (>1000) have high mortality rates, therefore they need urgent evaluation, diagnosis and management.
AIH types which is considered a prognostic factor in some centers seems not that useful in our subjects, since we had the highest rate of seronegative AIH.
Response to treatment was acceptable in our subjects, 61.6%, approximately the same as previous experiment in our center, but still lower than other centers.
In conclusion, mortality rate was lower in our patients regarding other centers (10.5%), on the other hand we had higher rates of transplantation (30.2%) which can be explained with availability of liver transplantation for children in our center and patients from all over Iran referred here for this reason.
Limitation: Single center study.