INTRODUCTION
Metabolic associated fatty liver disease (MAFLD), formerly known as non-alcoholic fatty liver disease (NAFLD), has emerged as the most frequent cause of chronic liver disease worldwide [1]. This disease is characterized by fat accumulation in the liver, in association with metabolic disease, obesity and diabetes mellitus 1,2. The severity of MAFLD can range from a simple hepatic steatosis to a metabolic associated steatohepatitis (MASH) that can eventually3,4) lead to cirrhosis and hepatocellular carcinoma . Histological changes in MASH are characterized by steatosis in more than 5% of hepatoctytes, ballooning degeneration, and intralobular inflammation 5.
Liver transaminases, mainly alanine aminotransferase (ALT) and aspartate aminotransferase (AST), are enzymes aggregated in the cytosol of hepatocytes [6]. These enzymes are normally detectable in the serum at low concentrations, typically at values <30 IU/L; however, any process that leads to the loss of hepatocyte membrane integrity or necrosis results in the release of ALT and AST in higher concentrations in the serum 6,7. Several physiological and risk factors may contribute to the serum levels of these enzymes including age, sex, body mass index, elevated levels of triglycerides, insulin resistance, and blood glucose level 8,9. In this way, serum levels of transaminases, particularly ALT, are important markers used to reflect hepatic inflammation and liver injury in patients with various liver diseases 6,7,10.
Studies on the dynamics in transaminase levels during the course of MAFLD and MASH are heterogeneous. For example, some studies have shown that higher ALT values are associated with a higher risk to develop MAFLD and eventually MASH 11,12. On the other hand, some studies have shown that ALT values do not correlate with the severity of histopathological changes in MAFLD 9. Patients with MASH with normal transaminase levels have also been described in variable proportions 13-15. A recent meta-analysis showed that up to 25% MAFLD patients and 19% MASH patients had normal ALT values in the overall patients evaluated 16. Close monitoring of this group of patients is important, given that over time more than 20% of patients with MASH, may also be at risk of progressive and severe liver disease, regardless of serum transaminases values 17.
Patients with MAFLD or MASH with normal serum transaminases values are often neglected given that physicians tend to evaluate hepatic risk of MAFLD based on changes in transaminases values, therefore, we aimed to identify the frequency of normal transaminases in obese patients with MASH, as well as to describe the clinical, biochemical and histological characteristics in this specific group of patients.
MATERIALS AND METHODS
Study design and patients
We performed an observational, descriptive, crosssectional, retrospective study. The current study was conducted in the bariatric surgery department of the Avendaño Clinic, a private clinic in Lima, Peru. The study was conducted in patients who were programmed for gastric sleeve surgery in the period between January 2017 through January 2019. Patients were included if they underwent gastric sleeve surgery, were older than 18 years with a BMI ≥ 30 kg/m2, and had associated at least 2 co-morbidities (diabetes mellitus type 2, insulin resistance, hypertriglyceridemia, hypertension, obstructive sleep apnea) with serious previous attempts for loss of weight, on at least two occasions. Exclusion criteria were patients with significant alcohol intake (> 20g/d), evidence of acute or chronic viral hepatitis, drug-induced hepatitis, autoimmune hepatitis, metabolic diseases of the liver or other chronic liver diseases.
Clinical, biochemical and anthropometric parameters
The clinical, biochemical and anthropometric parameters were evaluated for each patient. Blood sample collection was performed for each patient, the blood samples were obtained under fasting conditions and the following tests were carried out: total cholesterol, HDL cholesterol, LDL cholesterol, triglycerides, gamma-glutamyltransferase (GGT), hemoglobin, glucose and insulin. All laboratory procedures were performed under standardized conditions within the same laboratory. Insulin resistance was calculated using the homeostasis evaluation model (HOMA-IR), in the present study a cut-off point of 2.7 was used. The cutoff point for the serum ALT and AST values were 40 IU/L and 37 IU/L, respectively. Diabetes mellitus was diagnosed by the criteria of the American Diabetes Association (ADA) 18 and hypertension was diagnosed by the criteria of the Joint National Committee (JCN) 19.
Body mass index (BMI) was calculated based on body weight (kg) divided by height squared (m2). A BMI ≥ 30 kg/m2 was considered type I obesity; BMI ≥35 obesity type II, BMI ≥40 obesity type III.
All patients underwent liver biopsies prior to performing the surgery. All liver biopsies were taken from the right hepatic lobe, in a wedge, according to the clinic protocol. The patients signed an informed consent in all cases for the biopsy and for the surgery. The liver biopsy was read by two independent pathologists trained in hepatic histology, the diagnosis of MASH was based on the Brunt criteria 20.
Statistical analysis
For the analysis, a total of 159 patients with MASH were considered. Summary measures were calculated for quantitative and categorical variables. For the quantitative variables with the mean, standard deviation, median and interquartile ranges were calculated. For categorical variables, the frequency and percentage were calculated.
The analysis of the variables distribution and associated factors was performed in patients with high and normal transaminases. Also, a sub-group of 42 patients with significant fibrosis (F2 or greater) were analyzed. The following variables were analyzed in these groups: age, gender, BMI, blood pressure, Low HDL by gender, hypertriglyceridemia, glucose, diabetes and HOMA IR.
For the analysis of differences in the case of quantitative variables, Student’s t-test was performed if the variables followed a normal distribution, otherwise, the Mann-Whitney U-test was applied. For the categorical variables, the Chi Square test was used and for variables that had statistical significance, the odds ratio was also calculated. A p-value ≤0.05 was considered statistically significant. All statistical analysis were performed using the SPSS version 23.
RESULTS
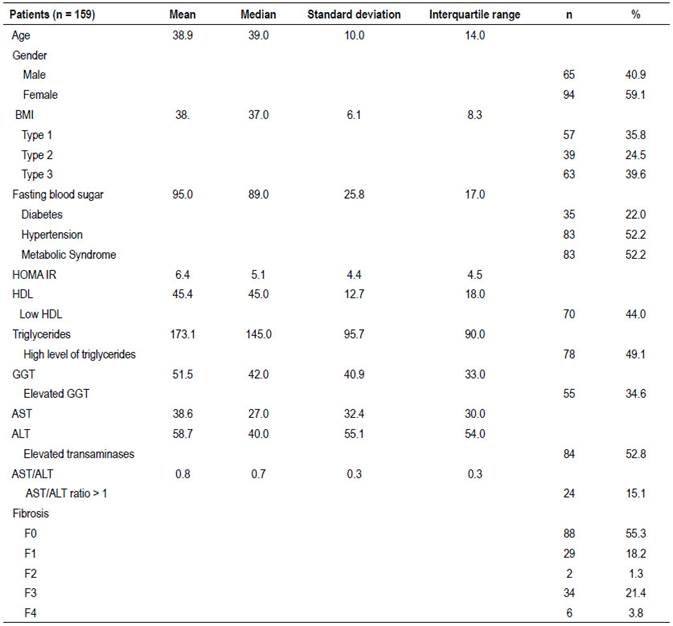
A total of 159 obese patients with MASH were included in the study. The descriptive statistics for the demographic, clinical and biochemical characteristics are shown in Table 1. The average age of presentation was 38.9 ± 10 years, 59.1% were female while 41.9% were male.
75 patients (47.2%) had normal transaminases and 84 patients (52.8%) elevated transaminases. 22% of the patients had diabetes mellitus type 2; and 52.2% met criteria for metabolic syndrome. 42 (26.5%) of the patients had significant fibrosis (F2 or greater).
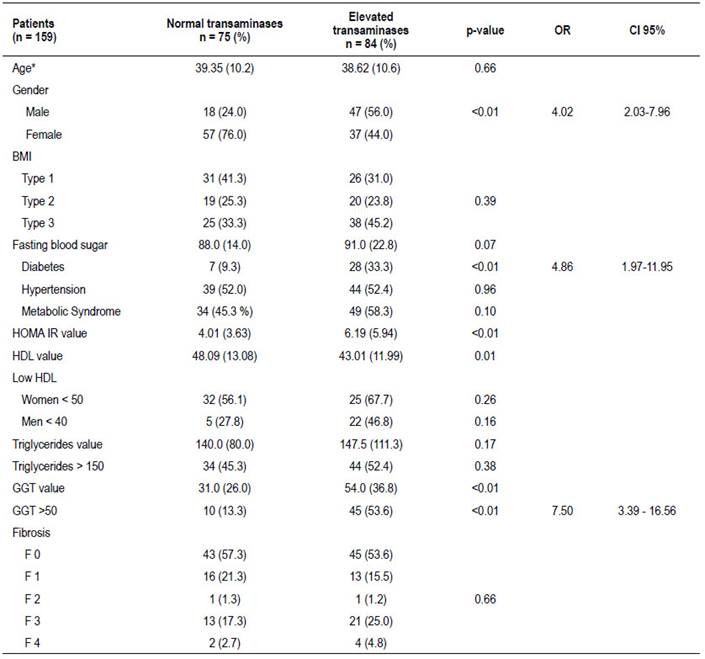
Table 2 shows the different characteristics in patients with normal and with elevated transminases. We could observe that patients with elevated transminases were predominantly male OR=4.04 (95% CI: 2.03-7.96; p<0.01). Also other factor associated with elevated transaminases were: patients with diabetes mellitus OR=4.86 (95% CI: 1.97-11.95; p<0.01) and values of GGT above 50 U/L OR=7.50 (95% CI: 3.39 - 16.56; p<0.01). In the group of elevated transaminases the values of HOMA IR and GGT were higher than the group of normal transminases, both statically significant with p-values <0.01. It should be noted that there were no statistically significant differences in fibrosis between the groups of patients with normal and elevated transaminases.
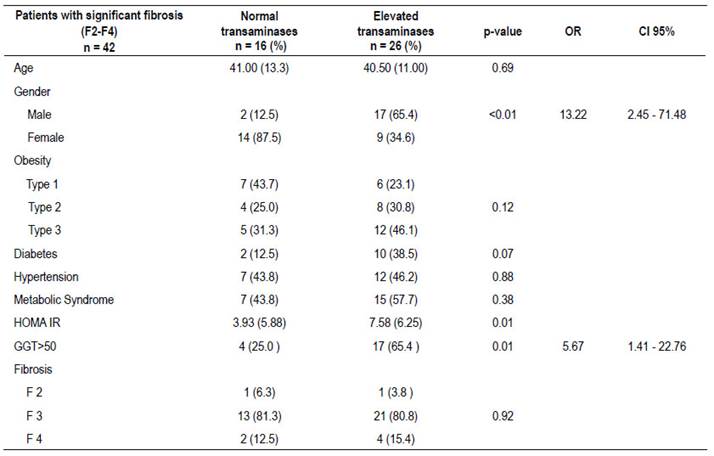
We found that 42 patients with MASH had significant fibrosis (F2 or greater), which were also analyzed according to transaminases levels. A total of 16/42 (38%) had normal transaminases and 26/42 (62%) had elevated transaminases as shown in Table 3. In these groups no differences were found in relation to the level of BMI, presence of diabetes mellitus or metabolic syndrome. It was found that most patients in the group with elevated transaminases were male OR=13.22 (95% CI: 2.45-71.48; p<0.01). Also, a higher proportion of patients with GGT values above 50 U/L was observed in the elevated transaminases group OR=5.67 (95% CI: 1.41-22.76; p<0.01) Finally, levels of HOMA IR were higher in the elevated transaminases group (p<0.01).
DISCUSSION
In our study we found that approximately half of the obese patients with MASH (47.2%) had normal levels of transaminases. Biochemical assessment of transaminases is usually performed by physicians as markers for liver injury, however, we found a surprisingly high proportion of patients with normal levels enzymes. Other studies have also evaluated the frequency of normal transaminases among patients with MASH. For example, Uslusoy et al.13 evaluated 23 patients with biopsy confirmed MASH, of which 7 (30.4%) had normal transaminases, somewhat lower than what we have found, it should be noted that the characteristics of their study population were similar to ours. A study performed by Francanzani et al.14 reported the frequency of MASH among patients with normal and elevated transaminases. They found that 332 patients had MASH diagnosis, of which 37 cases had normal ALT and 295 elevated ALT. The study by Verma et al. 15, found that among 54 patients with MASH a total of 6 (11.1%) had normal transaminases levels, in this case patients were diagnosed upon hepatic biopsy. A recent meta-analysis 16 have showed that the pooled prevalence of normal ALT was 25% in patients with MAFLD and 19% in patients with MASH; however, studies showed great heterogeneity. They also concluded that ALT value alone is not a significant metabolic indicator and did not possess enough accuracy for the diagnosis of MAFLD or MASH.
We evaluated the factors associated with elevation of liver enzymes among patients with MASH, also in patients with significant fibrosis (F2 or greater). Among these factors we found that being male, having the diagnosis of diabetes mellitus and having levels of GGT above 50 U/L were associated with elevation in liver enzymes. Also, the values of HOMA-IR and GGT were greater in the elevated transaminases group compared to the normal transaminases group, being statistically significant. We did not find a relationship between metabolic syndrome and the level of transaminases, confirming the same findings of other authors 13,14,15. Nonetheless, other studies have also reported an association between type 2 diabetes mellitus and a higher incidence of abnormal liver enzymes compared to individuals without diabetes, particularly ALT 21,22. Mandal et al.23 found alterations in ALT in 41.4% of their diabetic subjects, in this study ALT levels in diabetic patients with MAFLD were higher compared to patients without MAFLD. Although elevated liver enzymes may be a marker of hepatic disease in patients with diabetes, the evidence does not show a significant positive correlation between hepatic histological changes and biochemical alterations. Moreover, the advanced degree of MASH and fibrosis may occur in diabetic patients without significant abnormalities in liver enzymes 24.
In relation to insulin resistance, we found that patients with MASH and elevated transaminases had higher levels of HOMA-IR compared to patients with normal transaminases. These findings are similar to those reported by Maximos et al.4 which evidenced that insulin resistance in adipose tissue as well as hepatic triglyceride accumulation plays an important role in elevated transaminases leves in patients with MAFLD. The elevation of transaminases in the context of insulin resistance may be a cause-effect association or an early marker of insulin resistance 25. Markers of insulin resistance would help us identify those patients at risk for progression of liver damage, especially in the group of non-diabetic patients with MAFLD, where insulin resistance plays an important role in pathogenesis 26.
We did not find differences in the histological grade between the group of patients with normal and elevated transaminases, these findings are similar to those reported by other authors 13,14,15. It has been proposed that histological progression may occur even in the absence of elevated liver enzymes. The study by Mofrad et al.27 found that the whole spectrum of MAFLD can be seen in patients regardless of transaminases levels, moreover the only interdependent factor associated with advanced fibrosis was diabetes mellitus.
With regard to the analysis of obese patients with significant fibrosis (fibrosis ≥ 2), factors associated with elevated transaminases were being male and levels of GGT above 50 U/L. Previous studies have reported that elevations in GGT may be associated with risk of MAFLD, MASH and hepatic fibrosis 28,29.
In conclusion we found that the frequency of normal transaminases was 47.2% in obese patients with MASH. Factors associated with elevation in liver enzymes were being male, diagnosis of diabetes mellitus and elevation in GGT levels. It should be noted that the differences in the severity of fibrosis were not found in relation to transaminases. These findings suggest that transaminases levels do not correlate with histological liver damage and should not be used as markers for disease progression.