Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Ecología Aplicada
versión impresa ISSN 1726-2216
Ecol. apl. v.4 n.1-2 Lima ene./dic. 2005
Senna reticulata (Willd.) H. S. Irwin & Barneby (Fabaceae) as Pasture Killer (Matapasto) pioneer tree in amazonian floodplains
Senna reticulata (Willd.) H.S.Irwin & Barneby (Fabaceae) "Matapasto", árbol pionero en las planicies inundables amazónicas
Pia Parolin1
1 Max Planck – Instiute for Limnology, Tropical Ecology, P.O. Box 165, 24302 Plön, Germany.
Abstract
Senna reticulata ( Willd. ) H. S. Irwin & Barneby ( Fabaceae ) is a pioneer tree which colonizes open areas in Amazonian floodplains. It is very abundant throughout Amazonia along the nutrientrich whitewater rivers, and can be considered to be one of the most efficient colonizers of open areas. It possesses a very high tolerance towards flooding of the roots and stem, and performs extremely high photosynthetic assimilation and fast growth despite prolonged waterlogging. Efficient seed dispersal and the ability of resprouting after a period of unfavourable conditions add to its effectiveness. These features led the local people to consider Senna reticulata a noxious woody weed called matapasto ( pasture killer ) although in the successional sequence it represents a short-lived woody stage which enhances the establishment of highly diverse forests.
Key words: Amazonia, floodplain forest, Senna reticulata, succession, várzea, waterlogging, woody weed.
Resumen
Senna reticulata ( Willd. ) H. S. Irwin & Barneby ( Fabaceae ) es un árbol pionero que coloniza áreas abiertas en las planicies inundables de la amazonía. Es muy abundante a lo largo de los ríos de aguas claras y nutritivas y puede considerarse uno de los más eficientes colonizadores de áreas abiertas. Las raíces y tallos poseen una alta tolerancia a la inundación y presenta una alta asimilación fotosintética y rápido crecimiento a pesar de la inundación prolongada; además la eficiente dispersión de la semilla y la habilidad para rebrotar después de un periodo de condiciones desfavorables, la hacen muy efectiva. Estas características llevan a la población local a considerar a Senna reticulata como una nociva maleza leñosa, llamada matapasto ( pasture killer ) aunque en la secuencia sucesional represente una etapa leñosa de corta duración, la cual permite el establecimiento de bosques altamente diversos.
Palabras clave: Amazonía, planicie inundable, Senna reticulata, sucesión, várzea, inundación, maleza leñosa.
Introduction
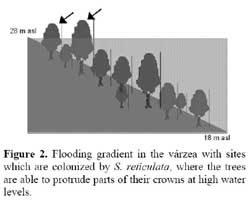
Senna reticulata ( Willd. ) H. S. Irwin & Barneby ( Fabaceae ) ( Figure 1 ) is a pioneer woody species which is common in nutrient-rich disturbed floodplain sites ( seasonal várzea, Prance, 1979 ) of the Amazon and its tributaries. It occurs mainly close to river channels and on abandoned pastures in the floodplains. It is restricted to the higher sites in the flooding gradient which get waterlogged for up to 7 months every year (Wittmann et al., 2002; Figure 2 ) . The duration of the high-water period is not crucial since this species is extremely flooding tolerant as long as it maintains some leaves above the water surface. It is fast-growing and outcompetes grasses and other woody pioneers by its fast growth. Since pastures which have been left without cattle for some months get overgrown by S. reticulata, the local farmers called this species pasture killer (matapasto ) .
The present paper aims at explaining why this species is interesting in the first place, why it deserves the local name matapasto (pasture killer) and how it can be so efficient to dominate over other fastgrowing woody and herbaceous species of the same ecosystem.
Senna reticulata ( Willd. ) H. S. Irwin & Barneby ( Fabaceae ) can reach a height of 12 m but trees exceeding 4 to 8 m are seldom found because they are cut by the local people for maintainance of their pastures. Sometimes S. reticulata occurs only in shrubby form of maximum 3 m height (Drury, 1873 ). Maximum recorded diameter at breast height ( dbh ) was 13 cm ( Parolin, 2001a ) , but the average dbh of adult, six-year old trees with a height of 5 m is 5.2 cm ( Parolin, 1998 ) .
Leaves of mature trees are compound with 8-14 pairs of leaflets which are obovate-oblong, obtuse, mucronate, glabrous on both sides, and thin. Flowers are large, yellow, fruits are long legumes, enlarged on each side with a broad crenulated wing.
Branches are many, spreading, irregularly angled, with bifurcation starting low on the stem. The canopy is very dense and wide. The trees of 4-8 m height have the densest canopies with diameters reaching 4-6 m. The species is rich in secondary compounds. In India, Senna reticulata was cultivated for medical uses: the juice of the leaves, mixed with lime juice, was used as a remedy for ringworm ( Drury, 1873 ) . The plant is said to be a cure in poisonous bites and cutaneous affections like fungal diseases ( Parolin, 2001a ) . The original geographical distribution is throughout Amazonian floodplains of whitewater rivers ( Ducke, 1949; De Menezes, 1978; Kalliola et al., 1991 ) but it also grows on wet sites on the terra firme, e.g. in the city of Manaus. It is typically found in sites which are wet and with high nutrient supply ( Parolin, 2001a ) .
Study area
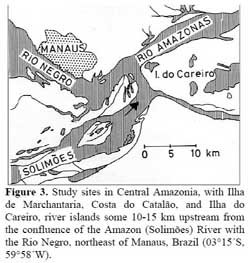
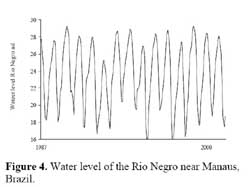
The studies were performed in Central Amazonia, mainly on the Ilha de Marchantaria, Costa do Catalão, and Ilha do Careiro, river islands some 10-15 km upstream from the confluence of the Amazon ( Solimões ) River with the Rio Negro, northeast of Manaus, Brazil ( 03°15´S, 59°58´W, Figure 3 ) . The climate is hot and humid. Mean monthly temperature is 26.6°C ( Ribeiro & Adis, 1984 ) and mean annual precipitation about 2105 mm, with 75 % falling in the rainy season from December to May. The seasonality of precipitation in the catchment area of the Amazon ( Solimões ) River results in an annual cycle of river discharge; water levels near Manaus change with a mean amplitude of 9.90 m per year ( Figure 4 ) . Germination experiments were performed in the city of Manaus, at the Instituto Nacional de Pesquisas da Amazônia ( INPA ) .
Parameters studied
Phenology and photosynthetic activity were measured directly in the field, on marked individuals which were maximum 20 m from the river margin when it is at its lowest level. Only adult trees were chosen, with heights of 4-6 m. At high water, the stems were flooded up to 5 m high. The tree crowns were not or only partially submerged. The phenological events were recorded at bimonthly intervals and included presence of leaves, leaf flush, flowering and fruiting. For measurements of photosynthetic CO2-uptake, fully exposed non-flooded sun leaves from five marked individuals were taken. Gas exchange was measured with an infra-red gas analyser ( IRGA, ADC LCA-2, Analytical Development Co. Ltd., Hoddesdon, Herts, UK) .
Germination and initial seedling growth, as well as adaptations against waterlogging were analysed under semi-natural conditions at the INPA, Manaus. Seeds were collected in the floodplains of the Amazon river near Manaus. During the rising period of the rivers up to 50 mature seeds were collected directly from five different trees. Seeds were placed in plastic bags, transported to the Amazon Research Institute ( INPA ) in Manaus and brought to an experiment site which was sunny in the morning and in the afternoon, but shady between 11.00 and 14.00 h. Seeds were placed in plastic bags containing 2 l of várzea soil for germination and seedling production.
Waterlogging experiments were performed in tanks at the experiment site using ground water from the INPA which was changed at weekly intervals. The vegetation analyses were performed in stands of different ages on the study islands. Species were identified in the field with the help of José F. Ramos from INPA ( Instituto Nacional de Pesquisas da Amazônia, Manaus ) and Leandro V. Ferreira ( INPA/Smithsonian Institution, Manaus ) . Identifications were checked in the herbarium of the INPA. All woody species ( including lianas ) with dbh > 1 cm or a height > 1 m were inventoried in seven plots of 25 x 25 m each.
Results
Phenology. Leaf shedding and replacement occurred continuously. The production of new leaves occurred during the whole year, but was reduced for 2-4 mo in the period of the highest water level. S. reticulata produces flowers and mature fruits starting at an age of 9-12 months, in the first period of flooding after establishment. It flowers in the high water period, from May to July. Fruits are mature in the high water phase, and seeds are released between June and August.
Photosynthetic activity. CO2-uptake ranged between a minimum of 9.8 μmol m-2 s-1 and a maximum of 24.5 μmol m-2 s-1. On average, CO2-uptake was 8% lower during the aquatic phase ( 18.4 ± 4.3 μmol m-2 s- 1 ) than in the non-flooded terrestrial phase ( 20.0 ± 4.0 μmol m-2 s-1 ) , although single measurements showed that waterlogged plants could have CO2-uptake values that exceeded those of the terrestrial phase.
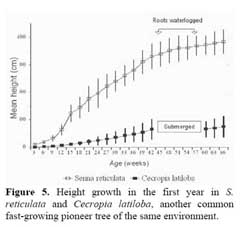
Germination and initial seedling growth. The seeds germinate within six days, but only if not submerged. Germination rate of non-flooded seeds was 85 % , compared to 0 % in submerged seeds. In the 8 months before the first flooding, the seedlings reached a height of 4 m in sunny places (Figure 5 ) .
Adaptations against waterlogging. S. reticulata forms high amounts of adventitious roots and lenticels in waterlogged seedlings and adult trees (Figure 6 ) . Tipped over trees have a very high capacity of resprouting ( Figure 7 ) . Waterlogged individuals produce significantly smaller leaves thus reducing the transpirational surface ( Borchert, 1994; Parolin, 2002a ) .
Vegetation analyses in stands with different ages. The stands of 2 years of age were almost monospecific with 86 % of all trees being individuals of S. reticulata. The crown in 4-5 m height was built exclusively by S. reticulata ( Figure 8 ) . With increasing age there was an increasing number of species: in the 6 year old stands only 28 % of all trees were individuals of S. reticulata. The crown in 7-8 m height was built by 12 species.
Discusión
Senna reticulata is one of the most fast-growing and productive trees of the Amazon floodplains. It is so efficient that justifiably it has the name matapasto - pasture killer. How can it be so efficient? This species has several adaptations against periodical waterlogging, as have many other common Amazonian floodplain trees ( Parolin et al., 2004 ) , e.g. the formation of adventitious roots and lenticels and leaf size adjustment ( Parolin, 2002a) . These adaptations, together with the extremely high photosynthetic capacity of this woody species, allow it to grow despite prolonged waterlogging with high water columns. In flooding experiments, seedlings survived periods of up to two years with consecutive waterlogging in standing water ( 2/3 of the plant submerged ) which still reached 80% of the photosynthetic rates. Waterlogging even may cause accelerated seedling growth in this species ( Parolin, 2001b ) and increased photosynthetic rates. The strategy that makes S. reticulata so efficient is a combination of this very high photosynthetic activity, as long as some cm of the plant are above the water surface, together with a high productivity of small seeds for several weeks, very fast germination ( Parolin, 2001c ) and extreme height growth in the first year ( Parolin, 2002b ) . Its high capacity of resprouting and intense lateral growth make it even more efficient in its subsequent years after establishment. The lateral growth leads to a very dense crown which is responsible for the outshading of competitors ( Figure 8 ) .
S. reticulata has a very efficient strategy of escape from submergence ( Parolin, 2002b ) characterized by high shoot elongation, leaf protrusion above the water surface and responses to waterlogging directed towards gaining oxygen. As contrast to this strategy, other species have the strategy of submergence tolerance, with slow seedling growth, dormancy during submergence, preservation of starch reserves by lower metabolism, leaf shedding or leaf maintenance with the postulated capacity to photosynthesize under water ( Schlüter & Furch, 1992; Schlüter et al., 1993 ) .
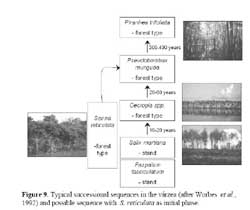
When compared to another evergreen fast-growing pioneer tree, Cecropia latiloba Miq., which also colonizes open areas very successfully and forms monospecific stands in the whitewater floodplains of the Amazon river ( Figure 5, 9 ) two distinct strategies may be found ( Parolin, 1999 ) . Senna reticulata has fast initital growth and intense lateral growth, and outshades plants competing for space and light, but does not tolerate total submersion. Cecropia latiloba grows more slowly but constantly, and its high flood tolerance allows vertical evasion from concurring species. The strategy of Cecropia latiloba is efficient in várzea environments with average to long periods of flooding. With shorter periods of flooding, and high nutrient availability ( e.g. on abandoned pastures ) , the strategy of Senna reticulata is more efficient and leads to its complete dominance.
So the final question is: How to get rid of `matapasto`? The local farmers have their efficient strategy to maintain their pastures clean from S. reticulata: first they cut the trees, then they burn the remaining stumps and finally they wait for the flood to cover the roots and the combination of these three measures kills the trees.
On the other hand, if the aim is not an open pasture but the return to a highly diverse forest, the easiest strategy is to wait. In fact, the vitality of S. reticulata decreases rapidly with increasing age ( Parolin, 1998 ) : the trees get higher but the crowns get lighter, with few leaves ( Figure 8 ) , and species diversity increases with species common to higher successional sequences dominating after few years. The typical successional sequences in the várzea ( Worbes et al., 1992 ) start – after an initial phase dominated by the grass Paspalum fasciculatum Willd. ex Flüggé – with monodominant stands of Salix martiana Leyb ( formerly called S. humboldtiana var. martiana ( Leyb. ) Anders. ) and Cecropia latiloba. S. reticulata may take the place of Salix martiana / Cecropia latiloba stands and lead to highly diverse forests as well ( Figure 9 ) .
Concluding, Senna reticulata is a `hated` woody weed in Amazonian floodplains, and therefore it is called matapasto. But its dominance is clearly limited in time: S. reticulata represents the initial phase of a successional sequence which leads to similarly structured and diverse forests as in areas which were not disturbed.
Acknowledgments
These studies were possible by financing from Deutscher Akademischer Austauschdienst ( DAAD ) , Capes, CNPq and the INPA/Max-Planck project. I want to thank the local fishermen and barqueiros of the INPA for help and advice received during my field work.
References cited
Borchert R. 1994. Water status and development of tropical trees during seasonal drought. Trees. 8: 115-125. Menezes E.M. de. 1978. Contribuição á morfologia comparativa de espécies daninhas do gênero Cassia L. ( Leguminosae - Caesalpinioideae ) . I: Estudo das plantas jovens. Rev. Brasil. Biol. 38 ( 3 ) : 537-548.
Drury H. 1873. The useful plants of India. Cassia alata. William H. Allen & Co., London. : 119-120.
Ducke A. 1949. Notas sobre a flora neotrópica II. As leguminosas da Amazônia brasileira. Instituto Agronomico do Norte, 2 ed., Belém Pará , Brasil. 18: 112-125.
Kalliola R., Salo J., Puhakka M. & Rajasilta M. 1991. New site formation and colonizing vegetation in primary succession on the Western Amazon floodplains. J. Ecol. 79: 877-901.
Parolin P. 1998. Floristic composition and structure of two stands of Senna reticulata differing in age. Amazoniana 15: 113-124.
________ . 1999. Growth strategies of Senna reticulata and Cecropia latiloba, two pioneer tree species of Central Amazonian floodplains. Bielefelder Ökologische Beiträge. 14: 272-277.
________ . 2001a. Senna reticulata, a pioneer tree from Amazonian várzea floodplains. The Botanical Review. 67( 2 ) : 239-254.
________ . 2001b. Morphological and physiological adjustments to waterlogging and drought in seedlings of Amazonian floodplain trees. Oecologia. 128: 326-335.
________ . 2001c. Seed germination and early establishment in 12 tree species from nutrient-rich and nutrient-poor Central Amazonian floodplains. Aquatic Botany. 70: 89-103.
________ . 2002a. Seasonal changes of specific leaf mass and leaf size in trees of Amazonian floodplains. Phyton. 42: 169-186.
________ . 2002b. Submergence tolerance vs. escape from submergence: two strategies of seedling establishment in Amazonian floodplains. Environmental and Experimental Botany. 48: 177-
Parolin P., De Simone O., Haase K., Waldhoff D., Rottenberger S., Kuhn U., Kesselmeier J., Schmidt W., Piedade M.T.F. & Junk W.J. 2004. Central Amazon floodplain forests: tree survival in a pulsing system. The Botanical Review. 70 (3) : 357-380.
Prance G.T. 1979. Notes on the vegetation of Amazonia. III. Terminology of Amazonian forest types subjected to inundation. Brittonia. 31 (1) : 26- 38.
Ribeiro M. de N.G. & Adis J. 1984. Local rainfall variability - a potential bias for bioecological studies in the Central Amazon. Acta Amazonica. 14(1/2) : 159-174.
Schlüter U.-B. & Furch B. 1992. Morphologische, anatomische und physiologische Untersuchungen zur Überflutungstoleranz des Baumes Macrolobium acaciaefolium, charakteristisch für die Weiß- und Schwarzwasserüberschwemmungswälder bei Manaus, Amazonas. Amazoniana. XII( 1 ) : 51-69.
Schlüter U.-B., Furch B. & Joly C.A. 1993. Physiological and anatomical adaptations by young Astrocaryum jauari Mart. ( Arecaceae ) in periodically inundated biotopes of Central Amazonia. Biotropica. 25( 4 ) : 384-396.
Wittmann F., Anhuf D. & Junk W.J. 2002. Tree species distribution and community structure of Central Amazonian várzea forests by remote sensing techniques. Journal of Tropical Ecology. 18: 805- 820.
Worbes M., Klinge H., Revilla J.D. & Martius C. 1992. On the dynamics, floristic subdivision and geographical distribution of Várzea forests in Central Amazonia. Journal of Vegetation Science. 3: 553-564.