Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634
Rev. perú. med. exp. salud publica vol.37 no.1 Lima Jan./Mar 2020
http://dx.doi.org/10.17843/rpmesp.2020.371.4514
Brief report
Identification of Leishmania species in patients derived to the National Institute of Health, Peru
1 Laboratorio de Referencia Nacional de Leishmaniasis, Instituto Nacional de Salud, Lima, Perú.
2 Laboratorio de Biotecnología y Biología Molecular, Instituto Nacional de Salud, Lima, Perú.
3 Escuela de Medicina Humana, Facultad de Ciencias de la Vida, Universidad Científica del Sur, Lima, Perú.
INTRODUCTION
Leishmaniasis is a neglected vector-borne disease. In the last five years, approximately one million cases of cutaneous leish maniasis have been reported worldwide, with an estimated 220,000 cases per year and more than 399 million people at risk of infection 1,2. In Peru, American tegumentary leishmaniasis (ATL) is considered a public health problem because it predomi nantly affects the poorest population, due to its wide geographical distribution 3) and the diversity of circulating species. The most prevalent species are Leishmania (Viannia) braziliensis and Leishmania (V.) guyanensis, mainly in the regions of Madre de Dios, Ucayali and San Martín; Leishmania (V.) peruviana in Lima, Ancash, La Libertad and Cajamarca; while Leishmania (L.) amazonensis has been less reported, mainly in the regions of Junin, Amazonas and Ucayali. Other species such as Leishmania (V.) lainsoni, Leishmania (V.) shawi, Leishmania (V.) panamensis, Leishmania (V.) colombiensis and the hybrid species Leishma nia braziliensis/peruviana have been sporadically reported 4-7.
Between 2000 and 2018, the Peruvian Ministry of Health (MINSA) reported 135,233 confirmed cases, an average of 7,117 cases per year, of which, approximately 94% (6,685 cases) was cutaneous and 6% (432) had the mucocutaneous form. In 2018, the regions of Madre de Dios and Cusco accounted for 25% of the total number of reported cases 3.
The identification of Leishmania species is done by multiple locus sequence typing (MLST), multilocus enzyme electro phoresis (MLEE), the use of monoclonal antibodies, sequencing of specific molecular markers 8,9 and by amplification of multiple markers in the conventional polymerase chain reaction (PCR) 10. On the other hand, high resolution melting analysis (HRMA) is a molecular method following real-time PCR that allows, through the analysis of DNA dissociation curves, the molecular identification of each species 11, population, as well as single-nucleotide polymorphism (SNP) 12.
KEY MESSAGES
Motivation for the study: To contribute to the description, updating and geographical distribution of Leishmania species in Peru.
Main findings: The identification of L. (V.) braziliensis, L. (V.) guyanensis, L. (V.) peruviana, as species with higher prevalence in the country, in addition to Leishmania (V.) lainsoni and L. (L.) amazonensis, the latter identified in a patient from Huanuco, an area with no history of previous cases.
Implications: Rapid and timely identification of the infecting Leishmania species by the analysis of the dissociation curves by HRMA, is important to determine the prognosis of the disease and for the development of future studies about differentiating species of Leishmania circulating in Peru.
Patients suspicious of leishmaniasis are referred, mainly from national hospitals and military and police institutions lo cated in different endemic areas of Peru to the National Health Institute (INS) which is part of the Health Ministry. The INS carries out the confirmatory diagnosis of the disease. In recently evolved skin cases, diagnosis is mainly achieved by parasito logical methods, such as direct microscopic examination or smear, which are widely used due to their low cost, and in vitro culture. The indirect immunofluorescence (IFI) and the Montenegro intradermal test (IDRM) are used for the immunosero logical diagnosis of the chronic mucocutaneous form of the disease. On the other hand, despite the efficiency of molecular methods such as PCR for the detection of the parasite, their implementation is restricted to specialized centers with adequate equipment, infrastructure and budget, as well as specialized personnel to guarantee the continuity of the service.
The objective of the present study was to identify the infecting Leishmania species in patients referred to the INS from 2006 to 2011, through the analysis of DNA dissociation curves.
THE STUDY
Observational, descriptive and retrospective study. The population studied were patients with clinical suspicion of ATL who attended the INS between 2006 and 2011, referred from national hospitals because they had ulcerous skin lesions with: raised edges, nodules, plaques or that were at mucosal level, The diagnosis was confirmed by parasitological methods at the INS Na tional Leishmaniasis Reference Laboratory.
The identification of Leishmania was made from genomic DNA extracted from amastigotes achieved by scraping the skin lesion with a sterile lancet, preserved in 70° alcohol and from promastigotes from in vitro culture. The samples were obtained as part of the development of the diagnostic routine for case confirmation. DNA extraction was performed using the recom mendations of the commercial PureLink Genomic DNA kit (Invitrogen, Life-technologies). The DNA samples were eluted in a volume of 60 µl and stored frozen until later use.
The study was performed by amplifying a region of the kinetoplast DNA (kDNA) in a RotorGene Q thermocycler (Qiagen) , in a reaction volume of 10 µl; 2 μl (5 ng/μl) of parasitic DNA were used, as well as 0.7 μM of the A1 primers: 5’-CCG CCC CTA TTT TAC ACC AAC CCC-3 and A2: 5’-GGG GAG GGG CGT TCT GCG AA-3’ 13) and 1X of the HRM-PCR Master Mix solution (Tipe-it HRM PCR Kit, Qiagen).
The identification of Leishmania species was developed through the analysis of dissociation curves using reference strains as control samples, the WHO codes are detailed in Annex 1. The HRMA identification of a test sample was achie ved by comparing the obtained profile with a control, and was expressed in percentage of similarity by the Rotor Gene Q Series Software version 2.3.1.
Likewise, with the purpose of confirming the cases found in the identification of Leishmania by HRMA, the concordance with 14 samples randomly selected by sequencing of the Leishmania spp, cytochrome B gene was determined according to the methodolo gy described by Foulet et al. 8 The primers used were Lei-CytB09: 5’-TTATGGTGTAGGTTTTAG-TYTAGGTT-3’ and Lei-CytB12: 5’-TGCTAAAACCACTCA-TAAATATACT-3’. The sequences obtained were analyzed and compared with the GenBank database (ht tps://www.ncbi.nlm.nih.gov/genbank).
The frequency of the variables age, sex, occupation, origin, duration of illness, number of lesions, type of lesions, location and total surface area of the lesion were determined. In addition, using the variables of the probable site of infection (at the regional level) and the identified Leishmania species, a distribution map was developed with the Quantum GIS version 3.8 geographic information system.
The study was approved by the INS Research Ethics Committee. Authorization for the use and analysis of the strains was also requested from the National Reference Laboratory for Leishmaniasis
RESULTS
Between January 2006 and December 2011, 262 patients with clinical suspicion of leishmaniasis were referred to the INS. Only 101 patients met the criteria for the parasitological diagnosis of the disease, 69 (68.3%) of which were confirmed for ATL and 32 (31.7%) were negative in parasitological and immunoserological diagnosis of the disease.
Regarding confirmed patients, by means of HRMA and by comparison with the control samples, the identification of Leish mania was achieved in 45 patients; while, in the remaining 24 it was not possible to determine the species of infecting Leishmania, due to the similarity found against the controls below the established cut-off value.
From the 45 patient samples confirmed by HRMA, 40% were identified as Leishmania (V.) braziliensis, 28.9% as Leishmania (V.) guyanensis, 17.8% as Leishmania (V.) peruviana, 6.7% as Leishmania (L.) amazonensis, and 6.7% as Leishmania (V.) lainsoni (Figure 1).
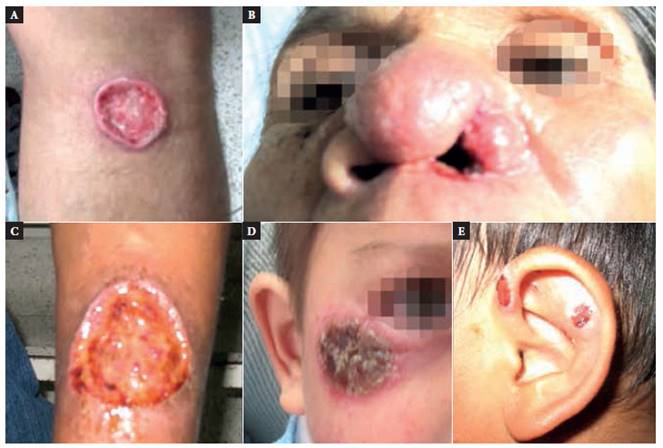
Figure 1 Lesions caused by (A) Leishmania (V.) guyanensis, (B) Leishmania (V.) braziliensis, (C) Leishmania (L.) amazonensis, (D) Leishmania (V.) lainsoni, and (E) Leishmania (V.) peruviana
For the identification of the species, a similarity was assumed to be ≥98% as the cut-off value, with the exception of the sam ples identified as Leishmania (V.) lainsoni which cut-off value was ≥78%. With this criterion, the infecting Leishmania species was identified in a total of 45 patients, 42 of which presented a similarity of ≥98% and the remaining three a similarity of ≥78%.
From the 45 patients identified, 43 (96%) presented the cutaneous form, of which 17 (38%) were caused by Leishmania (V.) braziliensis from Amazonas, Cajamarca, Cusco, Huanuco, Loreto, Madre de Dios and San Martin; 12 (27%) for Leishmania (V.) guyanensis from Amazonas, Cusco, Huanuco, La Libertad, Lima and San Martin 8 (18%) for Leishmania (V.) peruviana from Amazonas, Ayacucho, Huanuco, Junin, Lambayeque and Lima; 3 (7%) for Leishmania (V.) ) lainsoni from the provinces of La Convencion, Satipo and Huaura in the regions of Cusco, Junin and Lima, respectively; and 3 (7.0%) for Leishmania (L.) amazonensis from the provinces of La Mar and Huanta in the region of Ayacucho and Churubamba in Huanuco (Annex 2).
In the latter locality, the described species was identified from a patient with the disseminated cutaneous form. Finally, 4% of the population were two patients from the provinces of Puerto Inca and Huanuco affected by the mucocutaneous form, in whom we were able to identify Leishmania (V.) braziliensis and Leishmania (V.) guyanensis, respectively.
Likewise, the results identified by HRMA and the 14 randomly sequenced samples determined a 100% concordance.
The clinical-epidemiological characteristics of the 45 patients, in whom the identification of the infecting Leishmania spe cies was achieved are detailed in Table 1.
Table 1 Sociodemographic and epidemiological characteristics of confirmed cases of leishmaniasis, 2006-2011 (n=45)

a Merchant, bricklayer, engineer, teacher, civil employee, builder; b data includes a case of disseminated cutaneous leishmaniasis; c nostrils, lips, ears, cheeks, forehead, chin, head, neck; d chest, back, hips; e leg, calf, foot, knee, ankle, thigh, hand, finger, arm, forearm, elbow
DISCUSSION
The identification of Leishmania was conducted in 45 patients confirmed for ATL through the analysis of the kDNA dissocia tion curves, achieving identification of Leishmania (V.) braziliensis, Leishmania (V.) guyanensis, Leishmania (V.) peruviana, Leishmania (V.) lainsoni and Leishmania (L.) amazonensis, species reported in previous studies 4,14.
For the identification of the Leishmania species, a cut-off value was established related to the degree of genetic divergence, the close relationship between Leishmania (L.) amazonensis and Leishmania (L.) mexicana in the subgenus Leishmania and Leishmania (V.) braziliensis, Leishmania (V.) peruviana, Leishmania (V.) guyanensis and Leishmania (V. ) panamensis in the subgenus Viannia has led to the selection of the ≥98% similarity as the cut-off value, unlike the ≥78% similarity assumed for Leishmania (V.) lainsoni, because this species presents particular characteristics at the kDNA level, which differs from the other species in the subgenus Viannia15,16. In our study, this species was identified in samples from three patients from Cusco, Junin and Lima. In the latter, confirmation was achieved by sequencing the cytochrome B gene, while the samples from Cusco and Junin were confirmed by polymorphic length restriction fragment analysis (PCR-RFLP) 17. Conversely, samples from patients who did not reach the cut-off value were not considered because identification might not be correct.
Likewise, from the 45 samples identified, 61% were isolated from patients from the jungle region and 39% from patients from the sierra, mainly from the western slopes of the Andes and inter-Andean valleys, which allowed us to determine Leish mania (V.) braziliensis followed by Leishmania (V.) guyanensis and Leishmania (V.) peruviana as the most prevalent species, widely reported in our country 7,18. However, their ordinal position may vary depending on the origin of the population, as evidenced in the study conducted by Lucas et al. in which Leishmania (V.) braziliensis was most frequent in the jungle region, followed by Leishmania (V.) peruviana and Leishmania (V.) guyanensis.
Similarly, studies by Arevalo et al. and Kato et al. in predominantly sierra populations, where Leishmania (V.) peruviana mainly circulates, determined this species as the most prevalent, followed by Leishmania (V.) braziliensis and Leishmania (V.) gu yanensis. Other species reported from the western slopes of the Andes and inter-Andean valleys were Leishmania (V.) braziliensis in Ancash and Lima 18, and Leishmania (V.) guyanensis in Lambayeque, Lima 19 and La Libertad 14.
With respect to the mucocutaneous type, parasites obtained from two patients from Huanuco, Leishmania (V.) braziliensis and Leishmania (V.) guyanensis were identified as the infecting species, both with recognized capacity to develop metastasis and to evolve into aggressive forms of the disease 20,21. For the clinical mucocutaneous form, between 5% and 20% of total cases of leishmaniasis have been reported, depending on the geographical region 21. In Peru, approximately 6% of the mucocutaneous form of the total annual cases is reported 3. Leishmania (L.) amazonensis was also identified as the causal agent of disseminated cutaneous leishmaniasis in a patient from Huanuco. Cases of leishmaniasis with this clinically disseminated form are unusual and are generally investigated as case reports.
The 100% concordance obtained between the Sanger method sequencing and HRMA allows us to reaffirm the capacity of the latter, and of kDNA used as a marker in the identification of the infecting Leishmania species and, unlike sequencing, with less consumption of time and economic resources.
The limitations of the present study are related to the period in which the samples were taken (2006-2011), to the unequal geographical distribution of the patients, predominantly from the jungle region, to the lack of identification of the total of confirmed samples due to the low parasitic load which did not made possible to reach the cut-off value established for identi fication by HRMA, to the high costs of identification by sequencing using the Sanger method. It is because to these limitations that it was not possible to confirm the remaining 31 identified samples.
In conclusion, from the analysis of the DNA dissociation curves or HRMA of 45 patients, we were able to identify the species of Leishmania (V.) guyanensis, Leishmania (V.) peruviana, Leishmania (V.) braziliensis, Leishmania (V.) lainsoni and Leishmania (L.) amazonensis circulating in 13 regions of Peru with indigenous transmission of leishmaniasis. Likewise, we reported for the first time the circulation of Leishmania (V.) amazonensis in the Huanuco region, identified from a case with disseminated cutaneous leishmaniasis. Continuing studies related to the identification of Leishmania species with a greater number of samples will allow us to have a better knowledge of the distribution and dispersion of Leishmania spp species, con sidering the displacement of the vector, climate variations, migrations, mining activities, among other factors.
REFERENCES
1. Alvar J, Vélez ID, Bern C, Herrero M, Desjeux P, Cano J, et al. Leishmaniasis worldwide and global estimates of its incidence. PLoS One. 2012;7(5):e35671. doi: 10.1371/journal.pone.0035671. [ Links ]
2. The World Health Organization. Leishmaniasis in high-burden countries: an epidemiological update based on data reported in 2014. Relev épidémiologique Hebd. 2016;22(91):285-96. [ Links ]
3. Centro Nacional de Epidemiologia, Prevencion y control de Enfermedades. Sala de Situación de Salud. Semana Epidemiológica No 04- 2019 [Internet]. Lima: Ministerio de Salud; 2019. Disponible en: https://www.dge.gob.pe/portal/index.php?option=com_content&view=article&id=664:sala-situacional-2019&catid=2&Itemid=197. [ Links ]
4. Kato H, Cáceres AG, Mimori T, Ishimaru Y, Sayed ASM, Fujita M, et al. Use of FTA cards for direct sampling of patients' lesions in the ecological study of cutaneous leishmaniasis. J Clin Microbiol. 2010;48(10):3661-5 doi: 10.1128/JCM.00498-10. [ Links ]
5. Urbano J, Minaya-Gómez GS, Sanchez-Moreno M, Gutiérrez-Sánchez R, Marín C. Molecular characterization and geographical distribution of leishmaniasis aethiological agents in Peru. Rev Ibero-Latinoam Parasitol. 2011;70(2):145-56. [ Links ]
6. Tsukayama P, Lucas CM, Bacon DJ. Typing of four genetic loci discriminates among closely related species of New World Leishmania. Int J Parasitol. 2009;39(3):355-62. doi: 10.1016/j.ijpara.2008.08.004. [ Links ]
7. Lucas CM, Franke ED, Cachay MI, Tejada A, Cruz ME, Kreutzer RD, et al. Geographic distribution and clinical description of leishmaniasis cases in Peru. Am J Trop Med Hyg. 1998;59(2):312-7. doi: 10.4269/ajtmh.1998.59.312. [ Links ]
8. Foulet F, Botterel F, Buffet P, Morizot G, Rivollet D, Deniau M, et al. Detection and identification of Leishmania species from clinical specimens by using a real-time PCR assay and sequencing of the cytochrome b gene. J Clin Microbiol. 2007;45(7):2110-5 doi: 10.1128/JCM.02555-06. [ Links ]
9. Van der Auwera G, Dujardin J-C. Species typing in dermal leishmaniasis. Clin Microbiol Rev. 2015;28(2):265-94. [ Links ]
10. Castilho TM, Shaw JJ, Lucile M, Floeter-Winter LM. New PCR Assay Using Glucose-6-Phosphate Dehydrogenase for Identification of Leishmania Species. J Clin Microbiol. 2003;41(2):540-6. [ Links ]
11. Hernandez C, Álvarez C, Gonzales C, Ayala MS, León CM, Ramirez JD. Identification of Six New World Leishmania species through the implementation of a High-Resolution Melting (HRM) genotyping assay. Parasit Vectors. 2014;7:501-7. doi:10.1186/s13071-014-0501-y. [ Links ]
12. Galarza M, Fasabi M, Levano KS, Castillo E, Barreda N, Rodriguez M, et al. High-resolution melting analysis for molecular detection of multidrug resistance tuberculosis in Peruvian isolates. BMC Infect Dis. 2016;16(1):260. doi: 10.1186/s12879-016-1615-y. [ Links ]
13. Sierra Romero GA, Guerra MV., Paes M, Cupolillo E, Bentin Toaldo C, Macêdo VO, et al. Sensitivity of the polymerase chain reaction for the diagnosis of cutaneous leishmaniasis due to Leishmania (Viannia) guyanensis. Acta Trop 2001; 79 (3): 225-9. doi: 10.1016/s0001-706x(01)00140-1. [ Links ]
14. Córdova O, Vargas F, Hashiguchi Y, Kato H, Gómez E. Identificación de especies de Leishmania en pacientes y flebotominos en áreas de transmisión en una región del Perú. Rev Peru Med Exp Salud Pública. 2011;28(3):446-53. [ Links ]
15. Corrêa JR, Brazil RP, Soares MJ. Leishmania (Viannia) lainsoni (Kinetoplastida: Trypanosomatidae), a divergent Leishmania of the Viannia subgenus - A mini review. Mem Inst Oswaldo Cruz. 2005;100(6):587-592. doi: 10.1590/S0074-02762005000600014. [ Links ]
16. Kocher A, Valière S, Bañuls AL, Murienne J. High-throughput sequencing of kDNA amplicons for the analysis of Leishmania minicircles and identification of Neotropical species. Parasitology. 2017;1-8. doi: 10.1017/S0031182017002013. [ Links ]
17. Fraga J, Veland N, Montalvo Alvarez AM, Praet N, Boggild AK, Valencia BM, et al. Accurate and rapid species typing from cutaneous and mucocutaneous leishmaniasis lesions of the New World. Diagn Microbiol Infect Dis. 2012;74(2):142-50. doi: 10.1016/j.diagmicrobio.2012.06.010. [ Links ]
18. Arevalo J, Ramirez L, Adaui V, Zimic M, Tulliano G, Miranda-Verástegui C, et al. Influence of Leishmania (Viannia) Species on the Response to Antimonial Treatment in Patients with American Tegumentary Leishmaniasis. J Infect Dis. 2007;195(12):1846-51. doi: 10.1086/518041. [ Links ]
19. Victoir K, De Doncker S, Cabrera L, Alvarez E, Arévalo J, Llanos-Cuentas A, et al. Direct identification of Leishmania species in biopsies from patients with American tegumentary leishmaniasis. Trans R Soc Trop Med Hyg. 2003;97(1):80-7 doi:10.1016/S0035-9203(03)90031-9. [ Links ]
20. de Oliveira Guerra JA, Prestes SR, Silveira H, Raposo Camara Coelho LI de A, Gama P, Moura A, et al. Mucosal Leishmaniasis Caused by Leishmania (Viannia) braziliensis and Leishmania (Viannia) guyanensis in the Brazilian Amazon. Carvalho EM, editor. PLoS Negl Trop Dis. 2011;5(3):doi: 10.1371/journal.pntd.0000980. [ Links ]
21. Di Lella F, Vincenti V, Zennaro D, Afeltra A, Baldi A, Giordano D, et al. Mucocutaneous leishmaniasis: report of a case with massive involvement of nasal, pharyngeal and laryngeal mucosa. Int J Oral Maxillofac Surg. 2006;35(9):870-872 doi:10.1016/j.ijom.2006.02.015. [ Links ]
Funding: This work was funded by the National Public Health Center of the National Institute of Health of Peru.
Received: May 03, 2019; Accepted: December 15, 2019











 text in
text in 


