Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634
Rev. perú. med. exp. salud publica vol.37 no.4 Lima Oct-Dec 2020 Epub Oct 06, 2020
http://dx.doi.org/10.17843/rpmesp.2020.374.5491
Originales breves
Early physiological and morphological changes in the remnant kidney of living donors
1 Facultad de Medicina Alberto Hurtado, Universidad Peruana Cayetano Heredia, Lima, Perú.
2 Servicio de Nefrología, Hospital Cayetano Heredia, Lima, Perú.
3 Facultad de Salud Pública y Administración, Universidad Peruana Cayetano Heredia, Lima, Perú
INTRODUCTION
The donation of a kidney represents an abrupt loss of about half of the donor’s kidney function 1. Therefore, the remaining kidney of living donors experiences compensatory changes as early as the first week after nephrectomy and may persist for up to 10 years afterwards 2 , 3. Numerous donor follow-up studies in Europe, the Americas, Africa, and Asia describe that kidney function of a long-term donor, with adequate follow-up, is very similar to that of a person living with both kidneys 4 - 7. The most studied parameters are blood pressure (BP), proteinuria, glomerular filtration rate (GFR), and risk for developing stage 5 chronic kidney disease (CKD5) 4 - 7. Other important aspects to determine the prognosis of patients are the age of donation and the baseline body mass index (BMI). Those under 50 years of age have a better prognosis regarding kidney function recovery after nephrectomy 8, and those who are obese (BMI ≥30) lack renal function reserve after surgery 9.
Variation of kidney function reflects changes in many clinical parameters of living donors. The GFR of the remaining kidney increases from the first week of follow-up 10. BP shows little change in short and long-term follow-up, maintaining values similar to those before surgery with low incidence of high blood pressure (HBP) 6 , 9 , 11. Proteinuria is minimal after nephrectomy, but can increase in the long term and is of greater importance due to its association with hypertension and the decrease in glomerular filtration rate 11 - 13. The remaining kidney undergoes morphological changes from the first week post-nephrectomy. A significant increase in length has been described, as well as the increase in kidney volume up to 15% more at three months 2 , 14.
Although there are studies from Nordic countries, which include Hispanic populations, their authors agree that their results do not necessarily reflect the reality of Latin American countries 15. The aim of this study was to describe the functional and morphological changes in the remaining kidney of healthy donors during the first year after nephrectomy in the Peruvian population, due to the inexistence of follow-up information of kidney donors and the need to know about early morphological and physiological adaptive changes of the remaining kidney of living donors in the local ethnographic context.
KEY MESSAGES
Motivation for the study: To know the renal function variations of living kidney donors during the first year of follow-up after nephrectomy, in Peru.
Main findings: During the first twelve months, glomerular filtration rate, proteinuria, diastolic blood pressure, and kidney length showed significant changes. These results were similar to other international studies.
Implications: This is the first study that explores the physiological adaptation of Peruvian kidney donors and is a starting point for further research.
THE STUDY
This study was a retrospective case series and included people who had donated a kidney between 2007 and 2013, in the Renal Transplant Program of the Hospital Cayetano Heredia (HCH). These individuals were evaluated for co-morbidities before being accepted as donors. According to the program’s specifications, baseline parameters are obtained at most three months before nephrectomy. Once the effective donors and their medical records were identified, we registered the patients’ clinical and demographic information, which was codified in the data collection form specifically developed for the study.
We selected individuals whose medical records contained baseline data and at least one control during post-nephrectomy follow-up. Participants with high blood pressure, diabetes mellitus, pregnancy, or any clinical or physiological condition that might influence the course of renal function were excluded.
The variables of the study were the GFR, measured with creatinine clearance in 24 hours corrected for a of 1.73 m2 body surface; proteinuria measured in urine of 24 hours; the systolic blood pressure (SBP); the diastolic blood pressure (DBP); and the physical measures of the kidney (renal length and parenchymal thickness) obtained by ultrasound performed by an expert radiologist. We investigated the occurrence of mortality due to renal or cardiovascular causes and the development of CKD5 in an exploratory way. Baseline data and data corresponding to the first, second, third, sixth, and twelfth month of follow-up were collected. Half of the baseline GFR was considered to be attributable to a single donor kidney. The data was then entered into a database using Microsoft Excel and analyzed with Stata version 8.2.
Data analysis
We determined the absolute and relative frequencies for the categorical variables, and mean and standard deviation for the continuous ones. In order to describe the evolution of the clinical parameters, we constructed linear regression models, made by multiple observations of each participant. In the particular case of proteinuria, we used a logarithmic transformation to achieve a normal distribution. Linear and quadratic terms were evaluated for each continuous variable. The assumptions of linearity, independence, homoscedasticity and normality were corroborated for each of the final models obtained.
Ethical aspects
This study used only the information available in the patients’ medical records and did not involve sampling or direct interviews with donors. The main researcher filled the data collection sheets on a password-protected computer as a security measure. Each participant was assigned a code in the database so that others did not have access to personal data. The Faculty of Medicine and the Institutional Committee of Ethics of the Universidad Peruana Cayetano Heredia (Registry SIDISI 61396), as well as the Nephrology Service and the Ethics Committee of HCH (Code 096-013) reviewed and approved the protocol.
FINDINGS
Sixty healthy people donated a kidney in the period from 2007 to 2013. In the hospital’s archive, we found the medical records of 56 donors. None of them met the exclusion criteria before 12 months of follow-up. Finally, 55 donors were included, who had baseline data available. The average age was 40.88 years (standard deviation [SD ]: 9.84), 80% were women and the average baseline BMI was 25.68 (SD: 3.5). When constructing the linear and quadratic models, we observed that the GFR and proteinuria had a significant quadratic term. The rest of the variables remained under a linear model.
We observed that the GFR decreased until month 6, then it increased during month 12 (Figure 1A). For the 14 people with available preoperative and annual GFR values, the average 12-month GFR (94.1 mL/min/1.73 m2) was significantly lower than the preoperative (114.5 mL/min/1.73 m2, p = 0.006), but significantly higher than half of this value (GFR attributable to a single kidney) (p < 0.001). In other words, the GFR increases significantly if the GFR of a single kidney is considered as the starting point (Figure 1B).
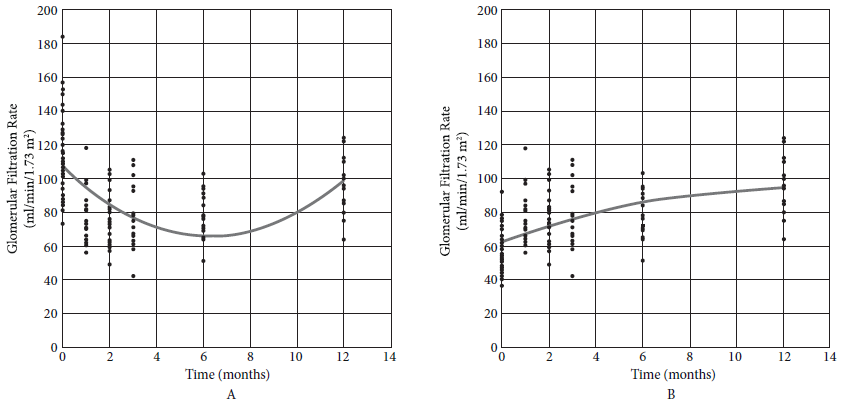
Figure 1 A: Glomerular Filtration Rate (GFR = 106.742 - 13.057 month + 1.032 month2). B: Glomerular filtration rate attributable to the remaining kidney.
The SBP showed no significant change over time (Figure 2A), but the DBP did show a significant increase over the follow-up (Figure 2B).
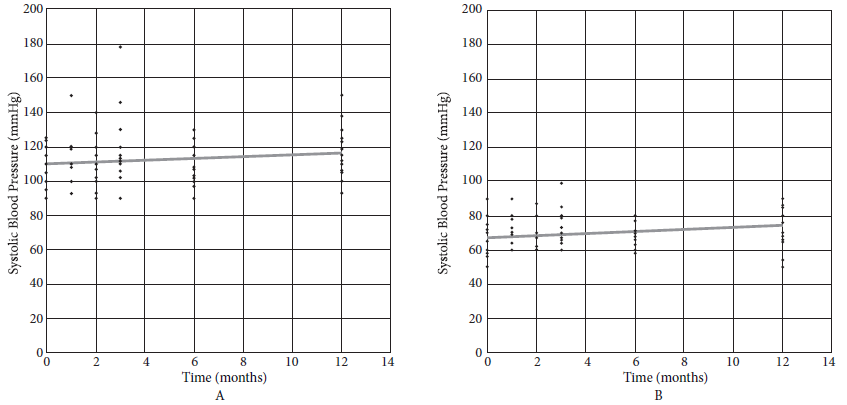
Figure 2 A: Systolic blood pressure (SBP = 109.881 + 0.503 month); B: Diastolic blood pressure (DBP = 67.137 + 0.608 month).
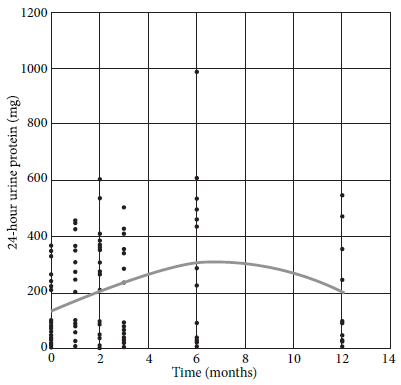
We observed an increase in the values of 24-hour urine protein until month 6, then it decreased until month 12, with significant changes throughout the follow-up (Figure 3).
Kidney length increased significantly during follow-up (Figure 4A). While the renal parenchyma did not show a significant change until 12 months (Figure 4B).
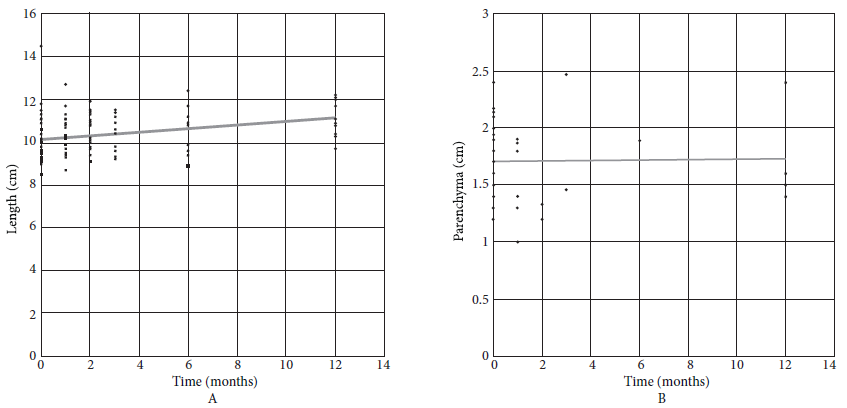
Figure 4 A: Kidney length (Length = 10.149 + 0.081 month); B: Kidney parenchyma (Length = 1.708 + 0.001 month).
No deaths were recorded during follow-up. Of the 14 donors who recorded data up to one year, none developed CKD5. The lowest recorded GFR value at 12 months was 64 mL/min/1.73 m2.
DISCUSSION
In this group of Peruvian donors, GFR, proteinuria, DBP, and remaining kidney length showed significant changes during follow-up. The findings are consistent with the information obtained from the literature. A meta-analysis that included numerous international studies found that global GFR varies during the first six months and is significantly lower than baseline, even 10 years later 16. We observed that global GFR decreases until six months and, although it then increases until the year of follow-up, it remains significantly lower than the preoperative value. However, only taking into account the performance of one kidney, we found that the difference is greater after one year.
The cause for the GFR variation in the remaining kidney is renal hypertrophy after contralateral nephrectomy, which produces hyperfiltration as compensation 17. In this study, by using ultrasound data, we found that the length of the remaining kidney increases significantly, which would be an indicator of the increase in renal mass as a consequence of hypertrophy. Other methods, such as computed tomography (CT), are better than ultrasound and reduce operator bias. These advantages are shown in a study that used CT and found that kidney hypertrophy can occur as early as 3 months and continues for the first year of follow-up 14.
On the other hand, proteinuria increases due to hyperfiltration and may remain elevated in the long term 16. This aspect is important during follow-up, since 24-hour urine protein is an indicator of chronic renal disease progression and a cardiovascular risk factor 16. In this group of donors, proteinuria increased for up to six months and then decreased, although it did not reach levels similar to those from the baseline. Evidence is heterogeneous regarding variations of SBP and DBP. Changes in blood pressure are expected, since the intrarenal renin-angiotensin system is active since the first week after nephrectomy 18.
In our study, we observed that only DBP increases significantly. A meta-analysis conducted in 2005, which evaluated studies about follow-up comparisons between donors and controls, showed that donors increased the SBP in 6 mmHg and the DBP in 4 mmHg over a period of 5 to 10 years when compared to controls. More recent studies show no change in blood pressure at three years 19 or small but significant changes without reaching HBP levels 20. Further study of donor blood pressure variation in larger populations is needed.
Age and BMI stand out among other factors that can influence the recovery of kidney function. Younger donors have a higher probability of developing hyperfiltration after nephrectomy than those older than 50 years 8. The population we studied had an average age of 40 years, so it was expected that recovery would be adequate. On the other hand, BMI is a conditioning factor for the recovery of renal function 9. In this case, the population had an average BMI of 25, which places it at the lower limit of overweight. Although donors with BMIs of up to 35 were found, the study did not have adequate data to carry out exploratory analyses in this aspect.
Overall, living kidney donors have a similar life expectancy and risk of HBP and CKD5 as the general population 6. Most studies of this type have been conducted in high-income countries 16. A study in the United States, which analyzed the likelihood of developing CKD5 in a group of healthy living donors, found that the Hispanic population was at higher risk than the Caucasian population, but lower than the African-American population. However, the authors warn that results cannot necessarily be transferred to other geographic areas 15, and therefore would not reflect the reality of the South American region and Peru because of ethnic diversity and sociodemographic characteristics. For this reason, it is essential for every Peruvian institution that carries out renal transplants to have adequate registry and follow-up of the donors, in order to have accurate information of the local population.
One of the limitations of this study was the lack of information or incomplete data in the medical records, which restricted the exploration of events such as CKD. Ideally, a prospective study should be conducted for a better analysis of the variables that can influence renal adaptation. However, this is the first effort to systematize the information of donors from a public hospital and it is expected to transcend to other institutions that perform organ transplantation from living donors.
In conclusion, in this population, the GFR of the remaining kidney, the proteinuria, the DBP and the length of the remaining kidney showed significant increase throughout the first year after the contralateral nephrectomy.
REFERENCES
1. Delanaye P, Weekers L, Dubois BE, Cavalier E, Detry O, Squifflet J-P, et al. Outcome of the living kidney donor. Nephrol Dial Transplant. 2012;27(1):41-50. doi: 10.1093/ndt/gfr669. [ Links ]
2. Anderson RG, Bueschen AJ, Lloyd LK, Dubovsky EV, Burns JR. Short-Term and Long-Term Changes in Renal Function After Donor Nephrectomy. J Urol. 1991;145(1):11-13. doi: 10.1016/S0022-5347(17)38232-0. [ Links ]
3. Chang S-G, Kim J-H, Lee S-J, Choi J-M, Huh J-S. Factors influencing contralateral renal hypertrophy after living donor nephrectomy. Transplant Proc. 2002;34(4):1139-42. doi: 10.1016/S0041-1345(02)02762-8. [ Links ]
4. El-Agroudy AE, Sabry AA, Wafa EW, Neamatalla AH, Ismail AM, Mohsen M, et al. Long-term follow-up of living kidney donors: a longitudinal study. BJU Int. 2007;100(6):1351-1355. doi: 10.1111/j.1464-410X.2007.07054.x. [ Links ]
5. Fournier C, Pallet N, Cherqaoui Z, Pucheu S, Kreis H, Méjean A, et al. Very long-term follow-up of living kidney donors: Living kidney donors in the very long term. Transpl Int. 2012;25(4):385-390. doi: 10.1111/j.1432-2277.2012.01439.x. [ Links ]
6. Ibrahim HN, Foley R, Tan L, Rogers T, Bailey RF, Guo H, et al. Long-Term Consequences of Kidney Donation. N Engl J Med. 2009;360(5):459-469. doi: 10.1056/NEJMoa0804883. [ Links ]
7. Oppenheimer Salinas F. Short, medium and long-term follow-up of living donors. Nefrologia. 2010;30 Suppl 2:100-5. doi: 10.3265/Nefrologia.pre2010.Nov.10699. [ Links ]
8. Fehrman-Ekholm I, Kvarnström N, Söfteland JM, Lennerling A, Rizell M, Odén A, et al. Post-nephrectomy development of renal function in living kidney donors: a cross-sectional retrospective study. Nephrol Dial Transplant. 2011;26(7):2377-2381. doi: 10.1093/ndt/gfr161. [ Links ]
9. Rook M, Bosma RJ, van Son WJ, Hofker HS, Homan van der Heide JJ, ter Wee PM, et al. Nephrectomy Elicits Impact of Age and BMI on Renal Hemodynamics: Lower Postdonation Reserve Capacity in Older or Overweight Kidney Donors. Am J Transplant. 2008;8(10):2077-2085. doi: 10.1111/j.1600-6143.2008.02355.x. [ Links ]
10. Oh CK, Yoon SN, Lee BM, Kim JH, Kim SJ, Kim K, et al. Beneficial Effects on the Renal Function of Both Recipients and Donors in Living Donor Kidney Transplantation. Transplant Proc. 2008;40(7):2310-2312. doi: 10.1016/j.transproceed.2008.07.001. [ Links ]
11. Borchhardt KA, Yilmaz N, Haas M, Mayer G. Renal function and glomerular permselectivity late after living related donor transplantation. Transplantation. 1996;62(1):47-51. doi: 10.1097/00007890-199607150-00010. [ Links ]
12. Garg AX, Muirhead N, Knoll G, Yang RC, Prasad GVR, Thiessen-Philbrook H, et al. Proteinuria and reduced kidney function in living kidney donors: A systematic review, meta-analysis, and meta-regression. Kidney Int. 2006;70(10):1801-1810. doi: 10.1038/sj.ki.5001819. [ Links ]
13. Goldfarb DA, Matin SF, Braun WE, Schreiber MJ, Mastroianni B, Papajcik D, et al. Renal outcome 25 years after donor nephrectomy. J Urol. 2001;166(6):2043-2047. [ Links ]
14. Shehab AB, Shaheen FA, Fallatah A, Al-Jobori AG, Sheikh IA, Al-Koussi M. Early changes in volume and function of the remaining kidney after unilateral donor nephrectomy. Saudi J Kidney Dis Transpl. 1994;5(4):474-478. [ Links ]
15. Muzaale AD, Massie AB, Wang M-C. Risk of end-stage renal disease following live kidney donation. JAMA. 2014;311(6):579-586. doi: 10.1001/jama.2013.285141. [ Links ]
16. Li S-S, Huang Y-M, Wang M, Shen J, Lin BJ, Sui Y, et al. A meta-analysis of renal outcomes in living kidney donors. Medicine (Baltimore). 2016;95(24):e3847. doi: 10.1097/MD.0000000000003847. [ Links ]
17. Chen KW, Wu MWF, Chen Z, Tai BC, Goh YSB, Lata R, et al. Compensatory Hypertrophy After Living Donor Nephrectomy. Transplant Proc. 2016;48(3):716-719. doi: 10.1016/j.transproceed.2015.12.082. [ Links ]
18. Ohashi N, Isobe S, Matsuyama T, Ishigaki S, Suzuki T, Tsuji T, et al. The Intrarenal Renin-angiotensin System Is Activated Immediately after Kidney Donation in Kidney Transplant Donors. Intern Med Tokyo Jpn. 2019;58(5):643-648. doi: 10.2169/internalmedicine.1756-18. [ Links ]
19. Kasiske BL, Anderson-Haag T, Israni AK, Kalil RS, Kimmel PL, Kraus ES, et al. A prospective controlled study of living kidney donors: three-year follow-up. Am J Kidney Dis. 2015;66(1):114-124. doi: 10.1053/j.ajkd.2015.01.019. [ Links ]
20. Kim SH, Hwang HS, Yoon HE, Kim YK, Choi BS, Moon IS, et al. Long-term risk of hypertension and chronic kidney disease in living kidney donors. Transplant Proc. 2012;44(3):632-634. doi: 10.1016/j.transproceed.2011.12.066. [ Links ]
Received: April 05, 2020; Accepted: July 16, 2020











 text in
text in