Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634
Rev. perú. med. exp. salud publica vol.38 no.1 Lima Jan-Mar 2021 Epub Feb 16, 2021
http://dx.doi.org/10.17843/rpmesp.2021.381.6286
Brief report
Clinical and epidemiological characteristics of cases of acute Chagas disease in the Peruvian Amazon basin, 2009-2016
1 Laboratorio de Chagas, Centro Nacional de Salud Pública, Instituto Nacional de Salud, Lima, Perú.
2 Centro Nacional de Epidemiología, Prevención y Control de Enfermedades, Ministerio de Salud, Lima, Perú.
3Escuela de Medicina, Facultad de Ciencias de la Salud, Universidad Peruana de Ciencias Aplicadas, Lima, Perú.
4 Dirección Regional de Salud Loreto, Iquitos, Perú.
5 Red de Salud San Francisco, Dirección Regional de Salud Ayacucho, San Francisco, La Mar, Perú.
6 Laboratorio de Referencia Regional, Dirección Regional de Salud Ayacucho, Huamanga, Perú.
7 Dirección Regional de Salud Huánuco, Huánuco, Perú.
8 Laboratorio de Referencia Regional, Dirección Regional de Salud Huánuco, Huánuco, Perú.
9 Red de Salud Condorcanqui, Dirección Regional de Salud Amazonas, Perú.
10 Centro de Salud Santa Rosa, Red de Salud San Francisco, Dirección Regional de Salud Ayacucho, Ayacucho, Perú.
INTRODUCTION
Chagas disease is caused by Trypanosoma cruzi, which is transmitted by hematophagous triatomines infected with the parasite, blood transfusion, congenital route, contaminated food or beverages and by other means 1. It has an acute and a chronic phase. People infected in the acute phase may present symptoms such as fever, inoculation chagoma, general malaise, hepatosplenomegaly and lymphadenopathy 1 , 2; 95% are asymptomatic 2. The diagnosis of this phase is based on the detection of the parasite by thick blood smear or microconcentration tests 1 - 2.
In Peru, between 2006 and 2010, seven cases of acute Chagas disease (ACD) had been reported, one in the district of Pevas and the other six in districts of the province of Datem del Marañón, department of Loreto 3 , 4; two of them were related, so the infection could be due to a common source (outbreak) 4. Reports of ACD in the last 20 years have been sporadic 4. Despite being a notifiable disease 5, there are few clinical and epidemiological reports. One of the objectives of surveillance is to detect acute cases and investigate the transmission method for timely treatment 5.
The aim of this report is to describe the clinical and epidemiological characteristics of eight cases of acute Chagas disease in the Peruvian Amazon, detected between 2009 and 2016.
KEY MESSAGES
Motivation for the study: There is limited information on the clinical and epidemiological characteristics of patients with acute Chagas disease (ACD) in Peru.
Main findings: Most cases were boys or girls under 11 years of age, three had, in addition, another disease. Diagnosis is late. Fever and general malaise showed in all patients. Three cases of ACD came from Loreto and two from Ayacucho. One four-month-old girl died due to the disease.
Implications: Chagas disease should be suspected in febrile patients, mainly in those from rural areas of the Peruvian Amazon.
THE STUDY
We carried out a case series study by reviewing medical records and clinical-epidemiological records of cases confirmed by the Instituto Nacional de Salud del Perú (INS) and reported to the epidemiological surveillance system, between 2009 and 2016. ACD cases were identified according to the definition of probable case —patient with fever as the main symptom, with at least one of the following symptoms: hepato-splenomegaly, adenomegaly, chagoma, Romaña sign, malaise, rash, meningoencephalitis, myocarditis, cardiomyopathy 5, coming from the Amazon basin— and were confirmed by the detection of the parasite.
Information was obtained by: a) interviewing cases or relatives 3 , 4 , 5; b) parasitological studies: direct examination, microconcentration and blood culture 6; c) identification of Trypanosoma cruzi by conventional PCR with amplification of a 188 bp nuclear satellite DNA sequence and a 330 bp fragment derived from the mini-exon variable region ( 7 , 8; d) discrete typing units (DTU) were identified by amplification of three regions: intergenic of the mini-exon gene, of the variable domain of the gene coding for 18S rRNA and of the D7 divergent domain of the gene coding for 24S rRNA 9, the nomenclature followed the recommendation described by Zingales et al 10; e) serological surveys of family or community collaterals for detection of IgG anti-T. cruzi antibodies by indirect immunofluorescence, ELISA and immunoblot 3 , 6 (Figure 1) and f) entomological investigation 3.

Figure 1 Flow chart of the detection of acute Chagas disease cases and epidemiological investigation (2009-2016).
Demographic, clinical and epidemiological variables are shown in Table 1. The mean age (years) of the cases and the time between symptom onset and parasitological diagnosis in days were calculated with the MS-Excel program.
Table 1 Demographic, clinical and epidemiological characteristics of acute Chagas disease cases in Peru (2009-2016).
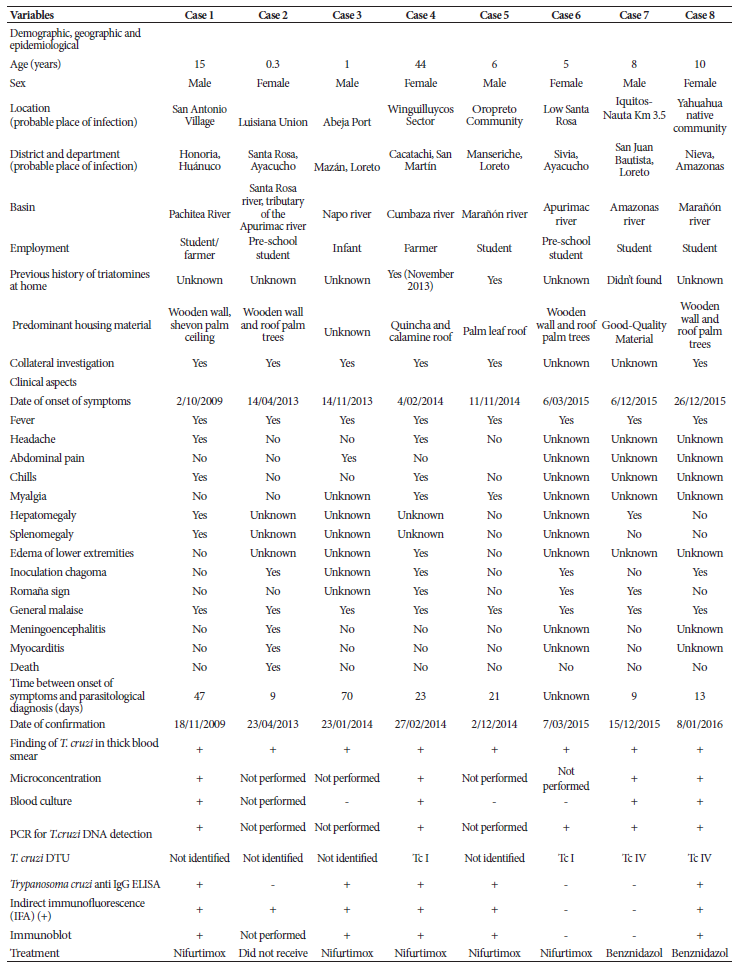
DTU: Discrete Typing Units
PCR: Polymerase Chain Reaction
The data were collected during the epidemiological surveillance of ACD in Peru 5. Patient data have been omitted to avoid identification.
FINDINGS
The mean age was 12.7 ± 13.7 years, range 1 to 44 years, and 4 cases were male. One case of ACD was associated with acute diarrheal disease (ADD), two with urinary tract infection (UTI) and a third with leptospirosis. The overall case fatality rate was 12.5% (1/8). Between 2009 to 2016, three cases of ACD came from Loreto, two from Ayacucho, one from Huánuco, one from San Martín and one from Amazonas. The predominant symptoms and/or signs were: fever and general malaise (8/8), inoculation chagoma (4/8) and Romaña sign (3/8). All were positive to parasitological tests (Table 1).
Case 1
A 15-year-old male, resident of a community one hour away by river from Puerto Inca (Table 1). On his first admission he was diagnosed with urinary tract infection (UTI) and febrile syndrome. Due to persistent symptoms, he was transferred to the Regional Hospital of Pucallpa, where a positive thick blood smear for Plasmodium falciparum was reported, as well as nausea, vomiting and two episodes of epistaxis. Urine examination showed turbidity, red blood cells (0 -1/field), leukocytes (2- 4/field) and bacteria. He started treatment for malaria and UTI. Parasitological controls were negative for the first three days. None of the family members were positive for Plasmodium and no Anopheles were found in the house. Quality control of thick blood smear at the Regional Referral Laboratory of Ucayali was positive for T. cruzi. The seroprevalence of T. cruzi infection among family and community collaterals was 1.5% (1/65) (Figure 1). He did not report blood transfusion. It was reported that the patient slept without a mosquito net in a palm leaf-roofed room tending a pig farm. Hematophagous triatomine nymphs were found in the poultry house (Figure 1). He was treated with nifurtimox (10 mg/kg body weight per day for 60 days) 11 (Table 1).
Case 2
Infant aged 0.3 years, daughter of a 17-year-old adolescent who worked in a brick factory, living in a community at an altitude of 700 meters over sea level (high jungle) (Table 1). She was taken to the health center in Santa Rosa, department of Ayacucho for presenting fever a week ago. She was diagnosed with UTI and received treatment. Due to her irritable state and persistent fever, she was admitted for emergency care. The thick blood smear for Plasmodium and the smear for Bartonella were negative, but Trypanosoma trypomastigotes were found. She was referred to the San Francisco Support Hospital for confirmation and treatment. On admission she had a temperature of 37 ºC, pulse 122/min, respiratory rate of 60/min and oxygen saturation of 95%. In the anamnesis, she was in poor general condition, had subcostal tugging, nasal flaring, irritability, anuria, rapid breathing, dry skin, evidence of tachycardia and nuchal rigidity. Respiratory distress, distal cyanosis and a pupillary diameter of 1.5 cm on both sides were observed. Hemoglobin concentration was 8.9 mg/dL. Radiography showed cardiomegaly. Multiorgan failure occurred and she died. T. cruzi was confirmed in the thick blood smear. The diagnosis was meningoencephalitis due to Chagas disease. She had no history of blood transfusion. The mother reported an erythematous papular lesion (inoculation chagoma) measuring 2 x 2 cm2 on the middle third of the medial side of the right thigh due to a possible triatomine bite, one week before the onset of symptoms. The father was seropositive for antibodies against T. cruzi (Figure 1).
Case 3
A one-year-old boy residing in a community three hours from the district of Mazán, department of Loreto. He went to the health facility for presenting episodes of watery ACD and fever (Table 1). The thick blood smear was negative for Plasmodium and quality control at the Loreto Regional Reference Laboratory reported the presence of T. cruzi. The parents reported a history of fever for almost three months since the onset of symptoms. They also reported that the child was taken to the farming field, and to logging and fishing activities within their community with his family. No family members were positive for T. cruzi (Figure 1). There was no history of transfusion. He was transferred to the Hospital de Apoyo de Iquitos, where the diagnosis was ACD and he received nifurtimox 11 (Table 1).
Case 4
A 44-year-old woman, resident of an urban area in the district of Morales, who was engaged in agricultural activities in Cacatachi, a rural area of the department of San Martín. She reported that when she fell asleep on a tree, she felt facial itching and the characteristic odor of a bedbug. After two days of fever (Table 1), she went to a private doctor’s office. Due to persistent fever, she was admitted to the Tarapoto Hospital with a diagnosis of dengue fever, with no alarm signs. When the fever persisted, she went to a health center, where the rapid test for dengue was negative and the Widal test was reactive, and she was treated for typhoid fever. Due to the persistence of fever, she was treated at the Hospital Nacional Arzobispo Loayza in Lima with a diagnosis of febrile syndrome, associated in the last three days with facial and lower limb edema, rash on the face, chest and legs, cough with hemoptysis and epistaxis, for which she was hospitalized. The hemogram, liver profile, renal profile, chest X-ray and abdominal ultrasound were normal.
The INS reported thick blood smear positive for T. cruzi, and positive for detection of IgG antibodies against T. cruzi (Table 1). She also had positive IgM and microagglutination (MAT) against Leptospira and positive IgG for dengue virus. Cardiological evaluation revealed little pericardial effusion (100 mL) and the electrocardiogram and cardiac enzymes were normal, ruling out myocarditis. Family collaterals were seronegative for T. cruzi (Figure 1). It was reported that the patient raised animals at home and occasionally consumes juices. She received nifurtimox for Chagas disease (Table 1) and oral doxycycline for leptospirosis.
Cases 5, 6, 7 and 8
Case 5 (6-year-old male) was admitted to the Saramiriza Health Center (Loreto), with fever, cough, swollen glands and other symptoms (Table 1). In the thick blood smear taken for malaria, blood forms of T. cruzi were observed (Figure 2). He reported seeing “bedbugs” in his home. Case 6 (5-year-old female) came from a community 15 minutes from Sivia (Ayacucho). She went to the local hospital with fever, loss of appetite, among other symptoms. The blood count showed hemoglobin at 10.8 mg/dL and T. cruzi in the thick blood smear. Case 7 (8-year-old male) went to a health center with fever, decreased appetite, among other symptoms and signs (Table 1), with a suspected diagnosis of dengue fever without alarm signs. Due to the persistence of the symptoms, he was taken to an EsSalud (Social Security) hospital in Iquitos, with a temperature of 40 ºC, respiratory rate of 110/min and oxygen saturation of 98%. Physical examination revealed rhythmic heart sound without murmur, and evidence of hepatomegaly. In the neurological evaluation there was no evidence of meningeal signs. She had a normal hemogram and received treatment. Other data for cases 5, 6, 7 and 8 are shown in Table 1 and Figure 3.
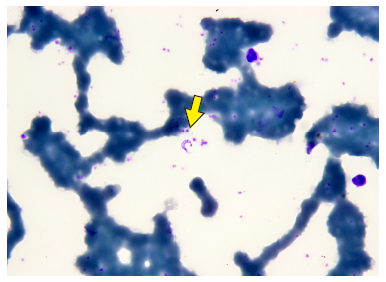
Figure 2 Blood Trypomastigote form of Trypanosoma cruzi in a thick blood smear slide stained with giemsa at 1000 X from case 5 (the yellow arrow indicates the parasite).
DISCUSSION
Eight cases of ACD were identified in this series, the most frequent symptoms and signs were fever and general malaise, the least frequent were inoculation chagoma, Romaña sign, myalgia, hepatomegaly, splenomegaly, meningoencephalitis, lower extremity edema and rash. These are similar to those found in a series in children from native communities of Loreto (Peru), as for fever, myalgia and splenomegaly, but differ in hepatomegaly, edema of lower extremities and general malaise ( 4. These differences could be explained by the incomplete collection of data in this study and probably, by the lineage of the parasite. The Romaña sign and inoculation chagoma were also infrequent in the series of ACD in Brazil 12 and Colombia 13.
In six cases of this report fever was observed for more than one week, as previously described 3 - 5 , 14 - 16. Only case 4, despite having coinfection with Leptospira, showed similarity with the clinical pattern of oral transmission of ACD because she was an adult. Generally, cases of ACD due to vector transmission are children 1. In addition, we observed lower limb edema and rash, which is more frequent in oral transmission 12. More than 57% and 27% of the 233 cases of ACD from the Brazilian Amazon had edema of limbs and face, and rash, respectively 12. Edema and rash in leptospirosis are infrequent 17. A characteristic of cases of ACD by oral transmission is the infrequent presentation of gateway signs 12 or the absence of gateway signs. In this series, 5 of 8 cases had gateway signs and in the epidemiological investigation of case 1, there was no evidence of oral transmission; therefore, 6 of 8 cases were probably vector-borne.
It is likely that the first case of ACD acquired dengue in Puerto Inca, since in addition to fever, the patient had alarm signs (vomiting and bleeding). At that time the incidence of dengue was more than 30 cases/100,000 inhabitants 18. One of the cases of ACD was misdiagnosed as P. falciparum malaria, in two cases typhoid fever was suspected, one was associated with CDD and another was associated with leptospirosis. It is necessary to strengthen the recognition of T. cruzi against the misdiagnosis of Plasmodium at the local level 3 , 4 , 14, incorporating it into the Zero Malaria Plan 19. Likewise, UTI was confirmed in two cases. The same was for a case of ACD reported in the central jungle that was initially suspected of UTI 15.
The time from symptom onset to parasitological confirmation averaged 27.4 days. This could be due to the fact that Chagas disease in areas of sporadic or active transmission is not a diagnostic first option in febrile patients; likewise, some microscopists at the first level of healthcare do not recognize the parasite in the thick blood smear slides. This could also be explained by the low sensitivity of the technique, which varies according to the date the sample is obtained in relation to the onset of infection and the delay in accessing health services, 2 to 23 days after the onset of symptoms. The mean time to diagnosis of ACD in this series is lower than that described in children from native communities in Peru 4.
In this series, the overall case fatality was 12.5%. The only death was probably associated with myocarditis by chagas and occurred rapidly —nine days from the onset of symptoms— as described 1. Late detection could be a risk factor for death in native Amazonian children 4.
There is an increasing trend in the number of cases of ACD reported in Peru, similar to what has been described in other Amazonian countries 20; besides to underreporting. The seroprevalence for IgG antibodies against T. cruzi among collaterals was higher than 1%; however, the sample is not reresentative and has not been randomly selected.
For two cases of ACD, the etiological agent was from the TcI lineage and for the other two it was from the TcIV lineage. The first is associated with wild reservoirs such as rodents, didelphids and wild vectors such as Rhodnius, Panstrongylus and the second is a sporadic agent of Chagas disease 21. This is the first time that the T. cruzi lineage has been identified in cases of ACD in the Amazonian areas where they were detected. It has been described that TcI infects P. geniculatus, a wild vector, and TcIV circulates in P. herrreri 22.
Among the limitations of this study, we should mention that it was not possible to access all the clinical files and clinical-epidemiological records of the cases; in addition, the interviews with some patients were conducted several days/weeks after the date of symptom onset, which may have caused some memory bias. Also, the results of the vectors are not included in this series.
Eight new cases of ACD have been reported in the Peruvian Amazon, mostly associated with other co-infections. The districts of Santa Rosa and Sivia (Ayacucho), appear to be new areas of active transmission, in which wild vectors could be involved.
Acknowledgements:
Dr. Mario Grandez-Armas of the Hospital Nacional Arzobispo Loayza and Dr. Omar Rodriguez for participating in the identification or follow-up of some of the cases presented in this series.
REFERENCES
. Pérez-Molina JA, Molina I. Chagas disease. Lancet. 2018;391(10115):82-94. doi: 10.1016/S0140-6736(17)31612-4. [ Links ]
. Teixeira AR, Nitz N, Guimaro MC, Gomes C, Santos-Buch CA. Chagas disease. Postgrad Med J. 2006;82(974):788-98. doi: 10.1136/pgmj.2006.047357. [ Links ]
. Cabrera R, Vega S, Cáceres AG, Ramal CA, Álvarez C, Ladera P, et al. Epidemiological investigation of an acute case of Chagas disease in an area of active transmission in Peruvian Amazon region. Rev Inst Med Trop. São Paulo 2010; 52:269-272. [ Links ]
. Cabrera R, Vega S, Valderrama Y, Cabanillas CK, Fernández C, Rodríguez O, et al. New focus of active transmission of Chagas disease in indigenous populations in Peruvian Amazon Basin. Rev Soc Bras Med Trop. 2013;46(3):367-72. doi: 10.1590/0037-8682-1195-2013. [ Links ]
. Cabrera CR. Enfermedad de Chagas o Trypanosomiasis Americana. En: Suárez OL, Beingolea ML, Nakamoto TI, Cabrera CR, eds. Protocolos de vigilancia epidemiológica. Parte I. Segunda edición. Lima: Dirección General de Epidemiología, Ministerio de Salud. 89-114; 2006. [ Links ]
. Vega CS. Manual de procedimientos de laboratorio para el diagnóstico de la trypanosomiasis americana (enfermedad de Chagas). Lima: Instituto Nacional de Salud; 1999. Serie de Normas Técnicas Nº 26. [ Links ]
. Moser DR, Kirchhoff LV, Donelson JE. Detection of Trypanosoma cruzi by DNA amplification using the polymerase chain reaction. J Clin Microbiol. 1989;27(7):1477-1482. doi:10.1128/JCM.27.7.1477-1482.1989. [ Links ]
. Vallejo GA, Guhl F, Chiari E, Macedo AM. Species specific detection of Trypanosoma cruzi and Trypanosoma rangeli in vector and mammalian hosts by polymerase chain reaction amplification of kinetoplast minicircle DNA. Acta Trop. 1999;72(2):203-12. doi: 10.1016/s0001-706x(98)00085-0. [ Links ]
. Brisse S, Verhoef J, Tibayrenc M. Characterisation of large and small subunit rRNA and mini-exon genes further supports the distinction of six Trypanosoma cruzi lineages. Int J Parasitol. 2001;31(11):1218-1226. doi:10.1016/s0020-7519(01)00238-7. [ Links ]
. Zingales B, Andrade SG, Briones MR, Campbell DA, Chiari E, Fernandes O, et al. Second Satellite Meeting. A new consensus for Trypanosoma cruzi intraspecific nomenclature: second revision meeting recommends TcI to TcVI. Mem Inst Oswaldo Cruz. 2009;104(7):1051-4. doi: 10.1590/s0074-02762009000700021. [ Links ]
. Ministerio de Salud. Doctrina, normas y procedimientos para el control de la Tripanosomiasis o enfermedad de Chagas en el Perú. Lima: Dirección General de Salud de las Personas, MINSA; 1998. [ Links ]
. Pinto AYN, Valente SAS, Valente VC, Ferreira GAJR, Rogrigues CJ. Fase aguda da doença de Chagas na Amazônia brasileira. Estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008;41:602-14. [ Links ]
. Nichols RS, Cucunubá ZM, Knudson A, Flores AC, Montilla M. Enfermedad de Chagas aguda en Colombia, una entidad poco sospechada. Informe de 10 casos presentados en el periodo 2002 a 2005. Biomédica.2007;27(Supl.1):8-17. [ Links ]
. Asayag CR, Garay CR, Sanchez GM, Angeles CC, Baca CJ, Evans C, et al. Eight year old with fever, hepatomegaly and positive thick smear. Am J Trop Med Hyg. 2008;79:473. [ Links ]
. Vega S, Mendoza A, Cabrera R, Cáceres GA, Campos E, Ancca J, et al. Primer caso de enfermedad de Chagas aguda en la selva central del Perú: Investigación de colaterales, vectores y reservorios. Rev Peru Med Exp Salud Pública 2006; 23(4):288-292. [ Links ]
. Lumbreras H, Arrarte J, Guevara B. Primeros casos de la enfermedad de Chagas en el Departamento de San Martín. Rev Med Per. 1955; 26:42-47. [ Links ]
. Toyokawa T, Ohnishi M, Koizumi N. Diagnosis of acute leptospirosis. Expert Rev Anti Infect Ther. 2011;9(1):111-21. doi: 10.1586/eri.10.151. [ Links ]
. Ministerio de Salud. Sala de situación de salud. Semana epidemiológica Nº 41- 2014. [Internet]. Lima: Dirección General de Epidemiología, MINSA; 2014 (citado el 26 de mayo de 2020). Disponible en: https://www.dge.gob.pe/portal/index.php?option=com_content&view=article&id=44. [ Links ]
. Cabrera R, Valderrama Y, Meza JR. Percepción sobre la enfermedad de Chagas y el riesgo de transmisión oral en Andoas, Loreto, Perú. Rev Peru Med Exp Salud Publica. 2019;37(1):174-5. Doi: https://doi.org/10.17843/rpmesp.2020.371.4875. [ Links ]
. Andrade DV, Gollob KJ, Dutra WO. Acute hagas disease: new global challenges for an old neglected disease. PloS Negl Trop Dis. 2014;8(7):e3010. doi: 10.1371/journal.pntd.0003010. [ Links ]
. Zingales B, Miles MA, Campbell DA, Tibayrenc M, Macedo AM, Teixeira MM, et al. The revised Trypanosoma cruzi subspecific nomenclature: rationale, epidemiological relevance and research applications. Infect Genet Evol. 2012;12(2):240-53. doi: 10.1016/j.meegid.2011.12.009. [ Links ]
. Padilla CP, Alvarado U, Ventura G, Luna-Caipo D, Suárez M, Tuñoque JR, et al. Detección de unidades discretas de tipificación de Trypanosoma cruzi en triatominos recolectados en diferentes regiones naturales de Perú. Biomédica. 2017;37(Supl.2):167-79. doi: https://doi.org/10.7705/biomedica.v34i2.3559. [ Links ]
Funding: Instituto Nacional de Salud de Perú, Centro Nacional de Epidemiología, Prevención y Control de Enfermedades (CDC), Ministerio de Salud, Dirección Regional de Salud de Loreto, Dirección Regional de Salud de Huánuco, Dirección Regional de Salud de Ayacucho and Dirección Regional de Salud de Amazonas.
Cite as: Vega S, Cabrera R, Álvarez CA, Uribe-Vilca I, Guerrero-Quincho S, Ancca-Juárez J, et al. Clinical and epidemiological characteristics of cases of acute Chagas disease in the Peruvian Amazon basin, 2009-2016. Rev Peru Med Exp Salud Publica. 2021;38(1):70-6. doi: https://doi.org/10.17843/rpmesp.2021.381.6286.
Received: August 05, 2020; Accepted: January 20, 2021











 text in
text in 




