Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634On-line version ISSN 1726-4642
Rev. perú. med. exp. salud publica vol.38 no.2 Lima Apr./Jun. 2021 Epub June 10, 2021
http://dx.doi.org/10.17843/rpmesp.2021.382.7158
Original articles
Factors associated with mortality due to COVID-19 in patients from a public hospital in Tacna, Peru
1 Facultad de Ciencias de la Salud, Universidad Privada de Tacna, Tacna, Perú.
2 Hospital III Daniel Alcides Carrión, Tacna, Perú.
3 Centro de Investigación de Estudiantes de Medicina, Tacna, Perú.
4 Universidad San Ignacio de Loyola, Unidad de Investigación para la Generación y Síntesis de Evidencias en Salud, Lima, Perú.
Objective:
To describe the clinical, laboratory and treatment characteristics of COVID-19 patients and to determine the factors associated with mortality during hospital stay. Materials and Methods: Retrospective cohort study of adult COVID-19 inpatients. The clinical, laboratory and treatment data were obtained from the medical records of patients from the Hospital Nivel III Daniel Alcides Carrión in Tacna. For the survival analysis we used the Cox proportional hazards model, and we calculated crude and adjusted hazard ratios (HR) with 95% confidence intervals (95% CI).
Results:
We evaluated 351 patients; 74.1% of them were men. The most common comorbidities were obesity (31.6%), hypertension (27.1%) and diabetes mellitus (24.5%). The median time of hospitalization was 8 days (IQR: 4-15). From the total of patients, 32.9% died during follow-up. The multivariate analysis showed an increased risk of dying associated with the following: age ≥65 years, HR = 3.55 (95% CI: 1.70-7.40); increase in lactate dehydrogenase >720 U/L, HR = 2.08 (95% CI: 1.34-3.22); and oxygen saturation less than 90%, mainly when it was less than 80%, HR = 4.07 (95% CI: 2.10-7.88). In addition, the use of colchicine during treatment showed a protective effect, HR = 0.46 (95% CI: 0.23-0.91).
Conclusions:
Risk factors for mortality due to COVID-19 included being older than 65 years, having oxygen saturation less than 90%, and elevated lactate dehydrogenase >720 U/L; colchicine treatment could improve the prognosis of patients.
Keywords: COVID-19; SARS-CoV-2; Risk Factors; Pandemics; Viral Pneumonia; Coronavirus Infections; Hospitalization; Mortality; Survival; Peru
INTRODUCTION
In December 2019, an outbreak caused by the novel human severe acute respiratory syndrome type 2 (SARS-CoV-2) coronavirus was first detected in Wuhan city, Hubei province, China 1, since then it has spread worldwide. Because of its rapid spread, the World Health Organization declared the 2019 coronavirus disease (COVID-19) pandemic on March 11, 2020 2.
The COVID-19 pandemic, a little more than a year after its appearance in Peru, has caused more than one million infections among Peruvians and has caused the death of more than 40,000. According to the General Comptroller of the Republic of Peru, in 2017, only 57% of a total of 251 hospitals possessed the adequate number of health personnel for proper functioning and only 58% could perform basic laboratory tests in addition to having the minimum equipment for the analysis of samples 3 , 4. The Peruvian health system collapsed; it had the highest mortality rate in the world during the first wave of contagion, with a shortage of health personnel of approximately 24,000 workers 5.
The main risk factors associated with death from COVID-19 are being ≥65 years of age, being male, suffering from hypertension, cardiovascular disease, diabetes, chronic obstructive pulmonary disease, or cancer 6. However, this information was obtained from high-income countries, which differs from low- to middle-income countries. It is known that the burden of critical illness is greater in middle- and low-income countries, which added to the precariousness of the health system 7, could have a greater impact on mortality due to COVID-19 in Peru. Due to this situation and the different behavior of the disease in each region of Peru, it is crucial to know the clinical or laboratory factors associated with mortality in order to be able to identify in a timely manner those patients at greater risk of death.
This study reports the results of an analysis of all hospitalized cases with confirmed diagnosis of SARS-CoV-2 pneumonia at Hospital III Daniel Alcides Carrión, in Tacna. The aim of the study was to identify demographic, clinical, laboratory and treatment characteristics associated with hospital death in patients with COVID-19.
MATERIALS AND METHODS
Study design
A retrospective cohort study was designed and executed in September 2020, and included patients who were hospitalized between March 1 through August 22, 2020.
Population and sample
We analyzed 450 medical records of all patients hospitalized for SARS-CoV-2 infection at the Hospital III Daniel Alcides Carrión; located in the district of Calana in Tacna, southern Peru. This is a level three referral hospital that belongs to the network of the Social Health Insurance in Tacna (EsSalud). It has a capacity of 110 hospital beds and 18 beds in the intensive care units (ICU). Its patients are mainly agricultural workers, fishermen, and construction workers 8.
We included adult patients (≥18 years), with confirmed diagnosis of SARS-CoV-2 infection, based on clinical, radiological criteria and with positive rapid test for detection of IgM and/or reactive IgG antibodies or SARS-CoV-2 detection test by polymerase chain reaction (PCR) in nasopharyngeal swab. These tests were processed in the molecular biology laboratory of the Tacna Regional Health Directorate. Patients without respiratory symptoms, those who withdrew voluntarily during hospitalization and those without confirmed diagnosis were excluded.
To find statistical power, we used the study by Díaz-Vélez et al. 9 and oxygen saturation (SatO2) ≤80% was considered as the main independent (exposure) variable. In that study 91.2% of participants with SatO2 ≤80% died during disease follow-up, while from those with SatO2 >80%, 53% died. Also, an unexposed/exposed ratio of 2.76 (345/125) was reported. With these parameters, a confidence level of 95% and 351 participants in the sample of this study, we calculated a statistical power of 99.9%.
Study variables
Death during hospitalization (yes/no) was considered as the main (dependent) variable. Exposure (independent) variables were age (18-49 / 50-64 / ≥65 years), sex (female/male), length of hospital stay (days), previous hospitalization (yes/no), oxygen saturation on admission (≤80% / 81% - 84% / 85% - 89% / ≥90%) and use of mechanical ventilation (yes/no). The comorbidities that had dichotomous answers (yes/no) were obesity, arterial hypertension, cardiovascular disease, type 2 diabetes mellitus, chronic respiratory disease, chronic kidney disease, oncologic diseases and immunosuppressive diseases; additionally, the number of comorbidities was recategorized (≤2/≥3). Laboratory data were leukocytes (<4000 / 4000-10 000 / >10 000 cells/mm3), lymphocyte percentage (%), aspartate aminotransferase (TGO, ≤40 / >40 U/L), alanine aminotransferase (TGP, ≤40 / >40 U/L), creatinine (≥ 1, <1 mg/ dL), C-reactive protein (CRP, ≤10 / >10 mg/dL), D-dimer (≤500 / >500 ug/L) and lactate dehydrogenase (LDH, ≤720 / >720 U/L); treatments that had dichotomous answers (yes/no) were antibiotics, corticosteroids, colchicine, tocilizumab and ivermectin.
Procedures
We used the general registry of patients who were hospitalized from March 1 to August 22, 2020. Once identified, the physical and electronic medical records were reviewed using the Intelligent Health Service, which is a software used to digitize patients’ medical records, collecting information on care, tests, diagnoses, prescriptions and treatments 10.
Four researchers carried out the double entry of data. A fifth researcher was in charge of the first quality control and, when any variation was found, compared the information with the clinical history.
Patients were treated according to institutional protocols developed specifically for patients with COVID-19, which were updated as new scientific evidence became available. This study did not influence the change of therapy of any patient.
Follow-up of the cases of death due to COVID-19 was done during the hospital stay, with starting time considered to be hospital admission and final time to be the occurrence of death or hospital discharge of the participants.
Statistical analysis
All data were analyzed in the statistical program Stata SE 14 (StataCorp., Texas, USA) in which a second quality control was conducted. In the descriptive analysis, simple frequencies and percentages were calculated for categorical variables and median and interquartile range (IQR) were calculated for numerical variables, since they showed an asymmetric distribution when evaluated. The following tests were used for bivariate analyses: categorical variables were analyzed with the chi-square or Fisher’s exact test, according to their expected values; and for numerical variables the Student’s t-test was used if there was a normal distribution or, in its absence, the Mann-Whitney U test. A value of p < 0.05 was considered statistically significant.
The survival function for the independent variables was carried out with the Kaplan-Meier method and the differences between survival functions were evaluated with the log-rank test. During the crude and adjusted regression analysis, we evaluated the association between baseline characteristics at admission and in-hospital mortality, using the Cox proportional hazards model to find the hazard ratios (HR) and their respective 95% confidence intervals (CI). The variables that entered the adjusted analysis were chosen for statistical significance in the crude Cox regression analysis. Variables with a p value < 0.05 were entered into the adjusted Cox regression analysis. Proportionality assumptions were evaluated using Schoenfeld residuals.
Ethical aspects
This study complies with the Helsinki norms for research on human subjects. The protocol was approved by the ethics committee of the Universidad Privada de Tacna (registration number: 48-FACSA-UI), it was also evaluated and approved by the research committee of the Hospital Daniel Alcides Carrión EsSalud-Tacna (registration number: 00012-EsSalud-CI). For greater transparency, the protocol was entered with code EI00000001619 in the national registry of Health Research Projects (PRISA) developed by the Instituto Nacional de Salud. Informed consent was not requested due to the retrospective observational nature of the study.
RESULTS
Of the 450 medical records reviewed, 99 did not meet the inclusion criteria (80 patients had a negative confirmatory test and 19 were hospitalized for other diagnoses). We included 351 medical records of patients hospitalized with a diagnosis of SARS-CoV-2 pneumonia. The median age was 61 (IQR: 50-69) years, the patients were predominantly male (74.1%) and 12.8% had more than three comorbidities, the most important being obesity (31.6%), arterial hypertension (27%) and diabetes (24.5%). The median hospital follow-up was 8 (IQR: 4-15) days.
Of the patients admitted, 70.1% had an oxygen saturation of less than 90%, of these, 39% had a saturation between 80% and 90%. There were 64 (18.2%) patients who required mechanical ventilation, 47 (73.4%) of them were admitted to the ICU, the median length of stay in the ICU and stay on mechanical ventilation was 13 (IQR: 8-18) and 8.5 (IQR: 5-14.5) days, respectively. Regarding ancillary tests, leukocytosis greater than 10,000 cells/mm3 was observed in 56.1% of patients, and the median percentage of lymphocytes was 10% (IQR 5-16) (Table 1).
Table 1 Description of the clinical and laboratory characteristics of the study population (n = 351).
| Characteristics | n (%) |
|---|---|
| Age (years) a | 61 (50-69) |
| 18-49 | 87 (24.8) |
| 50-64 | 132 (37.6) |
| ≥65 | 132 (37.6) |
| Sex | |
| Female | 91 (25.9) |
| Male | 260 (74.1) |
| Previous hospitalization | 9 (2.6) |
| Death during hospitalization | |
| No | 232 (66.1) |
| Yes | 119 (32.9) |
| Comorbidities | |
| Obesity | 111 (31.62) |
| Arterial Hypertension | 95 (27.1) |
| Cardiopathies | 12 (3.42) |
| Type 2 diabetes mellitus | 86 (24.5) |
| Chronic respiratory disease | 57 (16.2) |
| CKD | 20 (5.7) |
| Oncologic | 12 (3.42) |
| Immunosuppression | 8 (2.28) |
| Number of comorbidities a | 1 (0-2) |
| ≤2 | 306 (87.2) |
| ≥3 | 45 (12.8) |
| Patients admitted to MV | 64 (18.2) |
| Patients admitted to ICU | 47 (13.4) |
| Stay (days) | |
| Hospitalization a | 8 (4-15) |
| ICU a | 13 (8-18) |
| MV a | 8.5 (5-14.5) |
| Laboratory tests | |
| Oxygen saturation (%) a | 86 (79-90) |
| ≥90 | 105 (29.9) |
| 85-89 | 99 (28.2) |
| 81-84 | 38 (10.8) |
| ≤80 | 109 (31.1) |
| Leucocytes (cells/mm3) a | 11,110 (7,710-14,970) |
| <4,000 | 7 (2) |
| 4,000-10,000 | 147 (41.9) |
| >10,000 | 197 (56.1) |
| Lymphocyte percentage a | 10 (5-16) |
| TGO (U/L) a | 54 (36.5-86) |
| ≤40 | 104 (29.6) |
| >40 | 247 (70.4) |
| TGP (U/L) a | 59 (35-100) |
| ≤40 | 108 (30.8) |
| >40 | 243 (69.2) |
| Creatinine (mg/dL) a | 1.02 (0.86-1.21) |
| ≥1 | 166 (47.3) |
| <1 | 185 (52.7) |
| CRP (mg/dL) a | 14.99 (9.89-17.96) |
| ≤10 | 89 (25.4) |
| >10 | 262 (74.6) |
| D dimer (ug/L) a | 255 (190-473) |
| ≤500 | 228 (65) |
| >500 | 123 (35) |
| LDH (U/L) a | 754 (564-974) |
| ≤720 | 157 (44.7) |
| >720 | 194 (55.3) |
| Treatment | |
| Antibiotic | 341 (97.2) |
| Corticoids | 330 (94.0) |
| Colchicine | 50 (14.3) |
| Tocilizumab | 30 (8.6) |
| Ivermectin | 130 (37) |
a Median and interquartile range.
CKD: chronic kidney disease; MV: mechanical ventilation; ICU: intensive care unit; TGO: aspartate aminotransferase; TGP: alanine aminotransferase; CRP: C-reactive protein; LDH: lactate dehydrogenase
During hospital stay, 119 (32.9%) patients died, representing a mortality rate of 3.2 per 100 person-days; 38 (59.4%) patients on mechanical ventilation died. Statistically significant differences in mortality were observed in patients older than 50 years and in patients with comorbidities such as hypertension, diabetes and chronic kidney disease. Oxygen saturation was significantly lower in the group of patients who died (p < 0.001). In addition, we observed that admission to mechanical ventilator, having more than three comorbidities, leukocytosis, lymphopenia, and having increased C-reactive protein, D-dimer and lactate dehydrogenase were significantly more frequent in patients who died. Colchicine administration was significantly more frequent in patients who survived (Table 2).
Table 2 Clinical and laboratory characteristics of the study population according to hospital outcome.
| Variable | Survivors | Deceased | p value |
|---|---|---|---|
| (n = 232) | (n = 119) | ||
| Age (years) a | 57 (47-65) | 66 (59-76) | <0.001 d |
| Age (CT) | <0.001 b | ||
| 18-49 | 78 (89.7) | 9 (10.3) | |
| 50-64 | 90 (68.2) | 42 (31.8) | |
| ≥65 | 64 (48.5) | 68 (51.5) | |
| Sex | 0.185 b | ||
| Female | 55 (60.5) | 36 (39.5) | |
| Male | 177 (68.1) | 83 (31.9) | |
| Previous hospitalization | 0.998 c | ||
| Was hospitalized | 6 (66.7) | 3 (33.3) | |
| Was not hospitalized | 226 (66.1) | 116 (33.9) | |
| Obesity | 0.290 b | ||
| Yes | 69 (62.2) | 42 (37.8) | |
| No | 163 (158.6) | 77 (81.4) | |
| AHT | 0.003 b | ||
| No | 181 (70.7) | 75 (29.3) | |
| Yes | 51 (53.7) | 44 (46.3) | |
| CVD | 0.563 b | ||
| No | 225 (66.4) | 114 (33.6) | |
| Yes | 7 (58.3) | 5 (41.7) | |
| DM2 | 0.020 b | ||
| No | 184 (69.4) | 81 (30.6) | |
| Yes | 48 (55.8) | 38 (44.2) | |
| CRD | 0.413 b | ||
| No | 197 (67) | 97 (33) | |
| Yes | 35 (61.4) | 22 (38.6) | |
| CKD | 0.002 b | ||
| No | 225 (68) | 106 (32) | |
| Yes | 7 (35) | 13 (65) | |
| Oncologic disease | 0.116 c | ||
| No | 227 (67) | 112 (33) | |
| Yes | 5 (41.7) | 7 (58.3) | |
| Immunosuppression | 0.126 c | ||
| No | 229 (66.8) | 114 (33.2) | |
| Yes | 3 (37.5) | 5 (62.5) | |
| Admitted to MV | <0.001 b | ||
| No | 206 (71.8) | 81 (28.2) | |
| Yes | 26 (40.6) | 38 (59.4) | |
| Admitted to ICU | 0.724 b | ||
| No | 202 (66.4) | 102 (33.6) | |
| Yes | 30 (63.8) | 17 (36.2) | |
| Length of hospital stay (days) a | 9 (5-16) | 6 (3-11) | <0.001 d |
| Time in ICU (days) a | 13 (8-18) | 14 (9-17) | 0.782 d |
| Time in MV (days) a | 9.5 (6-16) | 7 (2-14) | 0.141 d |
| Number of comorbidities a | 1 (0-2) | 1 (1-2) | <0.001 d |
| Number of comorbidities (CT) | <0.001 b | ||
| ≤2 | 211 (69) | 95 (31) | |
| ≥3 | 21 (46.7) | 24 (53.3) | |
| Oxygen saturation (%) a | 88 (84-92) | 80 (69-87) | <0.001 d |
| Oxygen saturation (CT) | <0.001 b | ||
| ≥90 | 94 (89.5) | 11 (10.5) | |
| 85-89 | 69 (69.7) | 30 (30.3) | |
| 81-84 | 23 (60.5) | 15 (39.5) | |
| ≤80 | 46 (42.2) | 63 (57.8) | |
| Leucocytes (cells/mm3) a | 10,185 (7,395-14,085) | 13,220 (8,610-16,580) | <0.001 d |
| Leucocytes (CT) | 0.031 c | ||
| <4,000 | 5 (71.4) | 2 (28.6) | |
| 4,000-10,000 | 108 (73.5) | 39 (26.5) | |
| >10,000 | 119 (60.4) | 78 (39.6) | |
| Lymphocyte percentage a | 12 (7-17) | 6 (4-10) | <0.001 d |
| TGO (U/L) a | 55 (38-86) | 54 (36-80) | 0.676 d |
| TGO (CT) | 0.855 b | ||
| ≤40 | 68 (65.4) | 36 (34.6) | |
| >40 | 164 (66.4) | 83 (33.6) | |
| TGP (U/L) a | 69 (37-111) | 49.5 (30-80) | 0.002 d |
| TGP (CT) | 0.071 b | ||
| ≤40 | 64 (59.3) | 44 (40.7) | |
| >40 | 168 (69.1) | 75 (30.9) | |
| Creatinine (mg/dL) a | 1.01 (0.88-1.17) | 1.05 (0.84-1.35) | 0.147 d |
| Creatinine (CT) | 0.334 b | ||
| ≤1 | 114 (68.7) | 52 (31.3) | |
| <1 | 118 (63.8) | 67 (36.2) | |
| CRD (mg/dL) a | 13.9 (8.9-17.7) | 16.4 (11-18.5) | 0.004 d |
| CRD (CT) | 0.017 b | ||
| ≤10 | 68 (76.4) | 21 (23.6) | |
| >10 | 164 (62.6) | 98 (37.4) | |
| D Dimer (ug/L) a | 245 (178-344) | 325 (217-1,331) | <0.001 d |
| D Dimer (CT) | 0.050 b | ||
| ≤500 | 159 (69.7) | 69 (30.3) | |
| >500 | 73 (59.4) | 50 (40.6) | |
| LDH (U/L) a | 704 (532-874) | 846 (699-1,061) | <0.001 d |
| LDH (CT) | <0.001 b | ||
| ≤720 | 124 (78.9) | 33 (21.1) | |
| >720 | 108 (55.7) | 86 (44.3) | |
| Treatment with ATB | 0.505 c | ||
| No | 8 (80) | 2 (20) | |
| Yes | 224 (65.7) | 117 (34.3) | |
| Treatment with corticoids | 0.314 b | ||
| No | 16 (76.2) | 5 (23.8) | |
| Yes | 216 (65.4) | 114 (34.6) | |
| Treatment with colchicine | 0.025 b | ||
| No | 192 (63.8) | 109 (36.2) | |
| Yes | 40 (80) | 10 (20) | |
| Treatment with tocilizumab | 0.093 b | ||
| No | 208 (64.8) | 113 (35.2) | |
| Yes | 24 (80) | 6 (20) | |
| Treatment with ivermectin | 0.829 b | ||
| No | 147 (66.5) | 74 (33.5) | |
| Yes | 85 (65.4) | 45 (34.6) |
a Median and interquartile range, b Chi-square test, c Fisher’s exact test, d Mann Whitney U test.
CT: categorization of numerical variables, AHT: arterial hypertension, CVD: cardiovascular disease, DM2: type 2 diabetes mellitus, CRD: chronic respiratory disease, CKD: chronic kidney disease, MV: mechanical ventilation, ICU: intensive care unit, TGO: aspartate aminotransferase, TGP: alanine aminotransferase, CRP: C-reactive protein, LDH: lactate dehydrogenase, ATB: antibiotic.
Predictors of mortality were evaluated by Cox proportional hazard regression. The crude analysis found that the factors associated with mortality were age older than 50 years, and all oxygen saturation categories less than 90%. Comorbidities associated with mortality in the crude analysis were hypertension, diabetes mellitus, chronic kidney disease, cancer, and having more than three comorbidities. Altered laboratory tests associated with mortality were elevated TGP and LDH. The treatments associated with decreased mortality in the crude analysis were colchicine and tocilizumab. In the adjusted analysis, oxygen saturation less than 90% was independently associated with in-hospital mortality, presenting HRa = 2.20 (95% CI: 1.07-4.51), HRa = 2.54 (95% CI: 1.14-5.70) and HRa = 4.07 (95% CI: 2.10-7.88) times higher risk of death for oxygen saturation of 89% to 85%, 84% to 81% and ≤80%, respectively. Likewise, being older than 65 years was independently associated with mortality with HRa = 3.55 (95% CI 1.70-7.40) as was having lactate dehydrogenase greater than 720 U/L with HRa = 2.08 (95% CI 1.34-3.22). Treatment with colchicine was shown to be a protective factor for death in the multivariate analysis with HRa = 0.46 (95% CI 0.23-0.91) (Table 3).
Table 3 Crude and adjusted regression model for predictors of death due to COVID-19.
| Variable | Crude HR (95% CI) | p value | Adjusted HR (95% CI) | p value |
|---|---|---|---|---|
| Age (years) | ||||
| 18-49 | Reference | - | Reference | - |
| 50-64 | 2.79 (1.35-5.74) | 0.005 | 1.92 (0.90-4.07) | 0.089 |
| ≥65 | 4.95 (2.46-9.93) | <0.001 | 3.55 (1.70-7.40) | 0.001 |
| Sex a | ||||
| Female | Reference | - | - | - |
| Male | 0.69 (0.47-1.02) | 0.073 | - | - |
| Previous hospitalization a | 0.73 (0.23-2.30) | 0.593 | - | - |
| Admission to MV | 1.20 (0.80-1.80) | 0.358 | - | - |
| Comorbidities | ||||
| Obesity a | 1.10 (0.76-1.60) | 0.611 | - | - |
| AHT (mmHg) | 1.77 (1.21-2.57) | 0.004 | 0.92 (0.56-1.52) | 0.752 |
| Cardiovascular disease a | 1.23 (0.50-3.03) | 0.648 | - | - |
| Diabetes mellitus | 1.51 (1.03-2.23) | 0.038 | 0.89 (0.53-1.50) | 0.676 |
| Chronic respiratory disease a | 0.94 (0.59-1.51) | 0.806 | - | - |
| Immunosuppression | 1.79 (0.73-4.41) | 0.213 | ||
| CKD | 2.19 (1.23-3.91) | 0.009 | 1.26 (0.67-2.39) | 0.473 |
| Oncologic disease | 2.21 (1.03-4.77) | 0.048 | 2.12 (0.94-4.80) | 0.069 |
| Number of comorbidities | ||||
| ≤ 2 | Reference | - | Reference | - |
| ≥ 3 | 1.38 (1.17-1.61) | 0.001 | 1.71 (0.86-3.42) | 0.125 |
| Oxygen saturation | ||||
| ≥90 | Reference | - | Reference | - |
| 85-89 | 2.50 (1.25-5.00) | 0.010 | 2.20 (1.07-4.51) | 0.032 |
| 81-84 | 3.34 (1.52-7.28) | 0.003 | 2.54 (1.14-5.70) | 0.023 |
| ≤80 | 5.25 (2.76-9.97) | <0.001 | 4.07 (2.10-7.88) | <0.001 |
| Laboratory tests | ||||
| TGO (U/L) a | ||||
| ≤40 | Reference | - | - | - |
| >40 | 0.91 (0.61-1.34) | 0.635 | - | - |
| TGP (U/L) | ||||
| ≤40 | Reference | - | Reference | - |
| >40 | 0.65 (0.44-0.94) | 0.027 | 0.79 (0.52-1.20) | 0.269 |
| Creatinine (mg/dL) | ||||
| ≤1 | Reference | - | - | - |
| >1 | 1.29 (0.89-1.85) | 0.182 | - | - |
| CRP (mg/dL) a | ||||
| ≤10 | Reference | - | - | - |
| >10 | 1.48 (0.92-2.34) | 0.110 | - | - |
| D dimer (ug/L) a | ||||
| ≤500 | Reference | - | - | - |
| >500 | 1.43 (0.99-2.06) | 0.060 | - | - |
| LDH (U/L) | ||||
| ≤720 | Reference | - | Reference | - |
| >720 | 2.23 (1.49-3.34) | <0.001 | 2.08 (1.34-3.22) | 0.001 |
| Leucocyte cells/mm 3 a | ||||
| 4,000 - 10,000 | Reference | - | - | - |
| ≤4,000 | 0.96 (0.23-4.01) | 0.966 | - | - |
| ≥10,000 | 1.43 (0.97-2.10) | 0.075 | - | - |
| Treatment | ||||
| Antibiotic a | 0.92 (0.23-3.75) | 0.914 | - | - |
| Corticoids a | 0.88 (0.36-2.16) | 0.781 | - | - |
| Colchicine | 0.46 (0.25-0.90) | 0.024 | 0.46 (0.23-0.91) | 0.025 |
| Ivermectin a | 0.91 (0.63-1.33) | 0.678 | - | - |
| Tocilizumab | 0.35 (0.15-0.81) | 0.015 | 0.54 (0.23-1.28) | 0.165 |
a Variables that did not enter the adjusted regression model because they showed a p value > 0.05 in the crude regression model. The proportionality of the multivariate model had a p value = 0.093.
HR: hazard ratio; MV: mechanical ventilation; AHT: hypertension; CKD: chronic kidney disease; TGP: alanine aminotransferase; LDH: lactate dehydrogenase.
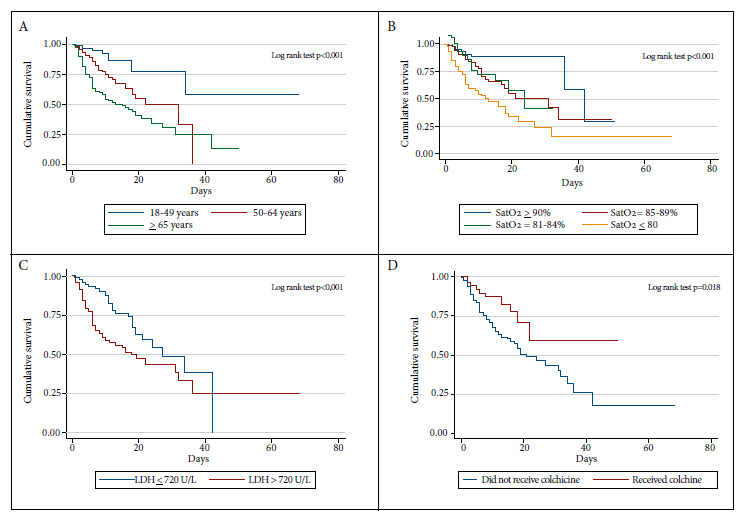
The risk proportionality assumption of the Cox model was evaluated using Schoenfeld residuals, and the assumption was considered to be complied with if the p value of the test was greater than 0.05. In this evaluation, the variable type 2 diabetes mellitus was excluded from the adjusted analysis because it had a p value = 0.023. Subsequently, a p value = 0.093 was obtained for the evaluation of the multivariate model. Figure 1 represents the survival function using Kaplan-Meier curves for the variables that were associated in the adjusted regression analysis.
DISCUSSION
One third of the participants died during the study period and approximately one fifth of them required mechanical ventilation. The risk factors with the greatest impact on in-hospital mortality were age over 65 years, oxygen saturation less than 90% and elevated lactate dehydrogenase. Colchicine administration resulted in a lower risk of death.
The mortality found in this study (32.9%) was higher than that observed in other reports from middle- and high-income countries (6.3% - 21%) 11 - 12. This is probably due to the fact that in these countries the health systems were better prepared to face the pandemic, with a greater number of intensive care beds, health personnel and medical treatment (mainly medical oxygen), as well as standard and updated care protocols. Likewise, the mortality reported in two hospitals from the Peruvian capital (36.9% - 52.3%) exceeded that of this study, as well as the total mortality of three hospitals in the north of the country (60.2%) 9 , 13 - 14. Another important difference was the mortality of patients admitted to intensive care in one hospital in Lima (73.9%) and in three hospitals in Lambayeque (81.3%) compared to that of this study (36.2%) 9 , 13 - 14.
These differences, even at the departmental level, are probably due to the preparation level of the regional health system, as well as the idiosyncrasy of each region of Peru, which led to different responses to the quarantine measures applied by the central government, so that, although the spread of the virus occurred throughout the Peruvian territory, each region had different infection curves, being a little delayed in the southern part of the country. This provided more time to prepare, as well as to develop and update protocols for the management of patients with COVID-19, in addition to the fact that many patients received therapies that were still being evaluated for COVID-19 (corticosteroids, tocilizumab, early pronation) and this may not have been similar in all Peruvian hospitals. These measures are probably reflected in the low mortality rate reported.
On the other hand, the proportion of patients admitted to the ICU was lower than what was described in other studies (14.2% in New York; 19.4% in Detroit; 20.5% in Genoa), but higher than what was observed in a hospital in Lima (10.2%) 12 , 14 - 16. These differences are probably due to the limited resources available in Peruvian hospitals. By March 8, 2020, the date in which patient zero was detected in Peru, there were only approximately 300 ICU beds 17. Although efforts were made to cover this large deficit, they were not sufficient to deal with the large volume of patients requiring intensive care.
We observed that the proportion of patients admitted with oxygen saturation less than 90% was higher than what was described in two level III public referral hospitals in Lima, Peru (64.5% in the Cayetano Heredia Hospital and 34.4% in the Edgardo Rebagliati Martins Hospital) 14 , 18. As in other studies, oxygen saturation less than 90% was associated with an increased risk of death, mainly when oxygen saturation was less than 80%. This high proportion of hypoxemia on admission reflects the precarious prehospital monitoring system, and the lack of adherence to the COVID-19 treatment algorithm at the first level of healthcare, where the measurement of oxygen saturation is most useful 19.
In this study, colchicine treatment was found to be a factor associated with greater survival, and although the small number of participants does not allow definitive conclusions, the initial clinical experience in this hospital showed that the evolution and prognosis improved in those who received this treatment; these data are similar to the results of cohort studies and clinical trials that show that treatment with colchicine could improve the results in patients with COVID-19 20 - 21. It is proposed that colchicine could possess an inhibitory effect on inflammasome activation, destabilization and degradation, and a potential antiviral effect that could be secondary to inhibition of microtubule polymerization 22 - 23. A systematic review and meta-analysis of four published studies reports an odds ratio for mortality of 0.28 (95% CI 0.18-0.44) suggesting a significant survival benefit 23. Although this systematic review has serious methodological deficiencies and its results should be interpreted carefully, it proposes the need for randomized clinical trials with good methodological design in order to demonstrate this potential effect.
Despite conflicting results in some randomized clinical trials of tocilizumab for COVID-19 24 - 27, tocilizumab treatment in this study showed a protective effect, but not statistically significant in the adjusted regression. Although the number of subjects who received it was small (30 patients), these findings coincide with the preliminary report of the RECOVERY clinical trial that included 4,116 participants, which showed that those patients who received tocilizumab had a lower risk of death: rate ratio: 0.86 (95% CI: 0.77-0.99), this benefit was observed in all subgroups and mainly in those who additionally received corticosteroids 26. These findings are similar to those reported in the REMAP-CAP clinical trial, where treatment with tocilizumab in critically ill patients with COVID-19 improved survival 27.
These findings should be interpreted with caution. The main limitation of this study was its retrospective nature, so it was not possible to examine all clinical characteristics (weight, body mass index, nutritional status prior to hospitalization, etc.) or history (smoking, alcohol consumption, physical activity, etc.), because in most patients these data were not recorded at hospital admission. On the other hand, there were unmeasured laboratory data (troponin, interleukin 6, procalcitonin, total and myocardial creatine phosphokinase). It is necessary that future reports should have a single registry card for patients diagnosed with COVID-19 in which all these data are included. Furthermore, the study was carried out in only one health institution, so the results cannot be extrapolated to the general population, and this is reflected in the fact that the results differ from other investigations carried out in Peru, so prospective studies that include a larger number of patients, that are multicenter and that have more detailed information are needed. Finally, it is possible that many of the participants may have self-medicated before admission to the hospital, which could alter the natural history of the disease and some laboratory values due to the adverse effects of self-medication.
In conclusion, this study describes that most of the hospitalized patients were male, the most frequent comorbidities were obesity, hypertension and diabetes, and one third of the hospitalized patients died. The main risk factors for mortality due to COVID-19 were age, being older than 65 years, and clinical alterations at hospital admission, such as elevated lactate dehydrogenase and oxygen saturation less than 90%, especially less than 80%. Early recognition of these risk factors may help to identify patients with a poor prognosis early in the course of the disease and implement better strategies that could reduce COVID-19-related mortality. It should be considered that the behavioral pattern of COVID-19 in Peru is probably different in each region, so the public health strategies should be adapted to the reality of each of these regions.
REFERENCES
. Abdullahi IN, Emeribe AU, Mustapha JO, Fasogbon SA, Ofor IB, Opeyemi IS, et al. Exploring the genetics, ecology of SARS-COV-2 and climatic factors as possible control strategies against COVID-19. Infez Med. 2020; Ahead Of Print1;28(2):166-173. [ Links ]
. WHO. director-general's opening remarks at the media briefing on COVID-19. [Internet] Geneva: World Health Organización; 2020 [citado el 30 de marzo de 2021]. Disponible en: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-themedia-briefing-on-covid-19---11-march-2020. [ Links ]
. Centro John Hopkins de Ciencias e Ingeniería de Sistemas. [Internet] Baltimore 2021 [citado el 30 de marzo del 2021]. Mapa interactivo: la pandemia de coronavirus en tiempo real 2021; Disponible en: https://coronavirus.jhu.edu/map.html. [ Links ]
. W Gianella C, Iguiñiz-Romero R, Romero MJ, Gideon J. Good Health Indicators are Not Enough: Lessons from COVID-19 in Peru. Health Hum Rights. 2020;22(2):317-319. [ Links ]
. Gestión. [Internet]. Lima 2020 [citado el 30 de enero del 2021]. Disponible en: https://gestion.pe/peru/minsa-hay-deficit-de-24000-profesionales-de-la-salud-para-enfrentar-pandemia-noticia/. [ Links ]
. Salomaa S, Bouffler SD, Atkinson MJ, Cardis E, Hamada N. Is there any supportive evidence for low dose radiotherapy for COVID-19 pneumonia?. Int J Radiat Biol. 2020;96(10):1228-1235. doi: 10.1080/09553002.2020.1786609. [ Links ]
. Diaz JV, Riviello ED, Papali A, Adhikari NKJ, Ferreira JC. Global Critical Care: Moving Forward in Resource-Limited Settings. Ann Glob Health. 2019;85(1):3. doi: 10.5334/aogh.2413. [ Links ]
. Seguro Social de Salud del Perú [Internet] Lima: EsSalud; 2020 [citado el 21 de enero del 2021]. Disponible en: http://noticias.essalud.gob. pe/?inno-noticia=essalud-amplia-servicios-y-potencia-equipamiento-para-atencion-covid-19-en-tacna. [ Links ]
. Díaz-Vélez C, Urrunaga-Pastor D, Romero-Cerdán A, Peña-Sánchez E, Fernández JL, Cossio JD, et al. Risk factors for mortality in hospitalized patients with COVID-19 from three hospitals in Peru: a retrospective cohort study [version 1; peer review: awaiting peer review]. F1000Research 2021,10:224. doi: https://doi.org/10.12688/f1000research.51474.1. [ Links ]
. Seguro Social de Salud [Internet] Lima: EsSalud; 2019 [Citado el 21 de enero del 2021]. Disponible en: http://www.essalud.gob.pe/essalud-implementa-historia-clinica-digital-para-atencion-de-asegurados/. [ Links ]
. Sousa GJB, Garces TS, Cestari VRF, Florêncio RS, Moreira TMM, Pereira MLD. Mortality and survival of COVID-19. Epidemiol Infect. 2020;148:e123. doi: 10.1017/S0950268820001405. [ Links ]
. Crawford JM, McGinn T, Davidson KW. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA. 2020;323(20):2052-2059. doi: 10.1001/jama.2020.6775. [ Links ]
. Rodríguez-Zúñiga MJM, Quintana-Aquehua A, Díaz-Lajo VH, Charaja-Coata KS, Becerra-Bonilla WS, Cueva-Tovar K, et al. Factores de riesgo asociados a mortalidad en pacientes adultos con neumonía por SARS-CoV-2 en un hospital público de Lima, Perú. Acta Med Peru. 2020;37(4):437-46. doi: 10.35663/amp.2020.374.1676. [ Links ]
. Mejía F, Medina C, Cornejo E, Morello E, Vásquez S, Alave J, et al. Oxygen saturation as a predictor of mortality in hospitalized adult patients with COVID-19 in a public hospital in Lima, Peru. PLoS One. 2020;15(12):e0244171. doi: 10.1371/journal.pone.0244171. [ Links ]
. Suleyman G, Fadel RA, Malette KM, Hammond C, Abdulla H, Entz A, et al. Clinical Characteristics and Morbidity Associated With Coronavirus Disease 2019 in a Series of Patients in Metropolitan Detroit. JAMA Netw Open. 2020;3(6):e2012270. doi: 10.1001/jamanetworkopen.2020.12270. [ Links ]
. Vena A, Giacobbe DR, Di Biagio A, Mikulska M, Taramasso L, De Maria A, et al. GECOVID study group. Clinical characteristics, management and in-hospital mortality of patients with coronavirus disease 2019 in Genoa, Italy. Clin Microbiol Infect. 2020;26(11):1537-1544. doi: 10.1016/j.cmi.2020.07.049. [ Links ]
. Giraldo EB. COVID-19 in Peru. Indian J Psychiatry. 2020;62(Suppl 3):S498-S501. doi: 10.4103/psychiatry.IndianJPsychiatry_1045_20. [ Links ]
. Benites-Goni H, Vargas-Carrillo E, Peña-Monge E, Taype-Rondan A, Arróspinde-Mormontoy D, Castillo-Córdova M, et al. Características clínicas, manejo y mortalidad de pacientes hospitalizados con COVID-19 en un hospital de referencia en Lima, Perú. Preprint [Internet] 2020 [citado el 21 de enero 2021]. Disponible en: https://preprints.scielo.org/index.php/scielo/preprint/view/905/1266. [ Links ]
. Organización Panamericana de la Salud. Algoritmo de manejo de pacientes con sospecha de infección por COVID-19 en el primer nivel de atención y en zonas remotas de la Región de las Américas, 2020[Internet]. Washington: OPS; 2020 [citado el 12 de septiembre de 2020]. Disponible en: https://iris.paho.org/handle/10665.2/52501. [ Links ]
. Sandhu T, Tieng A, Chilimuri S, Franchin G. A Case Control Study to Evaluate the Impact of Colchicine on Patients Admitted to the Hospital with Moderate to Severe COVID-19 Infection. Can J Infect Dis Med Microbiol. 2020;27;8865954. doi: 10.1155/2020/8865954. [ Links ]
. Deftereos SG, Siasos G, Giannopoulos G, Vrachatis DA, Angelidis C, Giotaki SG, et al. The Greek study in the effects of colchicine in COVID-19 complications prevention (GRECCO-19 study): Rationale and study design. Hellenic J Cardiol. 2020;61(1):42-45. doi: 10.1016/j.hjc.2020.03.002. [ Links ]
. Schlesinger N, Firestein BL, Brunetti L. Colchicine in COVID-19: an Old Drug, New Use. Curr Pharmacol Rep. 2020; 18:1-9. doi: 10.1007/s40495-020-00225-6. [ Links ]
. Vrachatis DA, Giannopoulos GV, Giotaki SG, Raisakis K, Kossyvakis C, Iliodromitis KE, et al. Impact of colchicine on mortality in patients with COVID-19. A meta-analysis. Hellenic J Cardiol. 2021; 6:S11099666(20)30285-2. doi: 10.1016/j.hjc.2020.11.012. [ Links ]
. Stone JH, Frigault MJ, Serling-Boyd NJ, Fernandes AD, Harvey L, Foulkes AS, et al. Tocilizumab Trial Investigators. Efficacy of Tocilizumab in Patients Hospitalized with Covid-19. N Engl J Med. 2020;383(24):2333-2344. doi: 10.1056/NEJMoa2028836. [ Links ]
. Salama C, Mohan SV. Tocilizumab in Patients Hospitalized with Covid-19 Pneumonia. Reply. N Engl J Med. 2021 3:10.1056/NEJMc2100217#sa2. doi: 10.1056/NEJMc2100217. [ Links ]
. Horby W, Pessoa-Amorim G, Peto L, Brightling CE, Sarkar R, Koshy T, et al. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): preliminary results of a randomised, controlled, open-label, platform trial. [version 1; peer review: awaiting peer review]. MedRxiv. doi: https://doi.org/10.1101/2021.02.11.21249258. [ Links ]
. REMAP-CAP Investigators, Gordon AC, Mouncey PR, Al-Beidh F, Rowan KM, Nichol AD, Arabi YM, et al. Interleukin-6 Receptor Antagonists in Critically Ill Patients with Covid-19. N Engl J Med. 2021 NEJMoa2100433. doi: 10.1056/NEJMoa2100433. [ Links ]
Cite as: Hueda-Zavaleta M, Copaja-Corzo C, Bardales-Silva F, Flores-Palacios R, Barreto-Rocchetti L, Benites-Zapata VA. [Factors associated with mortality due to COVID-19 in patients from a public hospital in Tacna, Peru]. Rev Peru Med Exp Salud Publica. 2021;38(2):214-23. doi: https://doi.org/10.17843/rpmesp.2021.382.7158.
Received: January 22, 2021; Accepted: April 16, 2021











 text in
text in