Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634
Rev. perú. med. exp. salud publica vol.38 no.4 Lima Oct./Dec. 2021 Epub Dec 22, 2021
http://dx.doi.org/10.17843/rpmesp.2021.384.9244
Original articles
Evaluation of the humoral response induced by BBIBP-CorV vaccine by determining neutralizing antibodies in peruvian healthcare personnel
1 Instituto de Investigaciones en Ciencias Biomédicas (INICIB), Facultad de Medicina Humana, Universidad Ricardo Palma, Lima, Perú.
2 Departamento de Medicina Interna, Hospital Nacional Hipólito Unanue, Lima, Perú.
3 Departamento de Patología Clínica y Anatomía Patológica, Hospital Nacional Hipólito Unanue, Lima, Perú.
4 Hospital Nacional Carlos Lanfranco La Hoz, Lima, Perú.
5 Instituto Nacional de Salud, Lima, Perú.
Objective.
To determine the titer of antibodies against the receptor binding domain (RBD) of the spike protein (S) in health personnel between the 4th and 12th week after receiving the BBIBP-CorV vaccine (Sinopharm).
Materials and methods.
We included a total of 168 healthcare workers from two hospitals in the region, who complied with the complete Sinopharm vaccine schedule; serum antibodies were measured using the Elecsys® Anti-SARS-CoV-2 test.
Results.
All participants developed antibodies to the RBD domain. The lowest antibody titer level was 1.78 U/mL. Levels equal to or above 250 were found in 70 (41.7%) participants. The geometric mean was 82.6 (95% CI: 67.8-100.6). Women had higher antibody levels. Participants whose antibodies were measured between 4- and 7-weeks post-vaccination showed significantly higher antibody levels than patients whose antibody levels were measured between 10- and 12-weeks post-vaccination. Among patients with a history of COVID-19, antibody levels were found to be at or above 250 U/mL in 88% of cases, compared to 6% among those without a history of COVID-19, (p<0.001).
Conclusion.
All participants immunized with BBIBPCorV vaccine were positive for antibodies against the SARS-CoV-2 spike protein RBD. The correlation between the titer level and protection against COVID-19, as well as the length of the protection provided by the vaccine, needs to be evaluated.
Keywords: Neutralizing Antibodies; Vaccination; Immunity; COVID-19; SARS-CoV-2
INTRODUCTION
Since the SARS-CoV-2 pandemic began, several research teams worldwide started the development of vaccines against COVID-19, mainly inactivated virus, recombinant protein, vectored and RNA vaccines, especially aimed at producing antibodies against SARS-CoV-2 spike (S) proteins 1 - 6. The efficacy of these vaccines has been demonstrated to be in the range of 70 to 95% 7 - 10, showing immunogenicity comparable to that developed by convalescent patients 1 , 3 , 6 , 11 , 12.
The various antibody tests are a useful tool for identifying subjects who have had prior exposure to COVID-19 13, these antibody titers vary depending on several factors (age, sex, COVID-19 severity, and days since infection) 14 and may decline substantially over time 15 - 17. Reports by Manisty et al. and Long et al. have associated clinical severity of the infection with the magnitude of initial antibody responses and the duration of circulating antibody titers 14 , 18, which would explain the sustained antibody levels in hospitalized patients for 3 to 6 months 19 - 21.
Neutralizing antibodies are considered to be a good marker for measuring humoral responses 22. The plaque reduction method is the gold standard for measuring antibody levels 23. However, this methodology is expensive and requires infrastructure and biosafety conditions that are inaccessible for most health facilities. The measurement of antibodies against the receptor binding domain (RBD) of the virion spike (S) protein is a useful methodology to quantify neutralizing antibodies. This antibody determination has shown good correlation with the plaque reduction method, which is currently considered to be the gold standard 24 - 26.
High mortality rates among health personnel were reported during the pandemic in Peru, mainly due to the lack of biosafety materials and overcrowding 27. By July 14, 502 physicians had died from COVID-19 complications 28, therefore this population group was considered in the first phase of the national vaccination program, which began in February. For this vaccination phase, the inactivated virus vaccine BBIBP-CorV from the Sinopharm laboratory was used, a vaccine with a reported efficacy of 86% 10. By June 30, more than 85% of physicians had received the second dose 30.
Although published phase II clinical trials on the BBIB-CorV vaccine show the production of neutralizing antibodies in 99% of cases 5, there are doubts regarding the immunogenicity in the Peruvian population, which motivated the Peruvian health authorities to modify the vaccination scheme against COVID-19, implementing a booster dose of the vaccine produced by the Pfizer/BioNTech laboratory, in order to achieve a better immune response.
The aim of this study was to evaluate the humoral response determined by the titer of antibodies against the receptor binding domain (RBD) of the spike (S) protein in health personnel between the 4th and 12th week after receiving the BBIBP-CorV vaccine (Sinopharm) as an indicator of the acquired immune response following vaccination.
KEY MESSAGES
Motivation for the study: One of the priorities during the current pandemic is to ensure the efficacy of the preventive measures used to reduce contagion, so we must understand the immunological responses obtained after vaccination, which is important for adequate decision making. This study is a step towards understanding the complex relationship between the virus and the immune system, which should be subsequently studied in greater depth.
Main findings: Vaccination with the BBIBP-CorV vaccine (Sinopharm) provides adequate immunogenicity.
Implications: Further studies should be carried out in order to better understand the behavior of the immune system in response to vaccination.
MATERIALS AND METHODS
Study population and sample
We included health personnel from the Hipólito Unanue and Carlos Lanfranco La Hoz national hospitals, who received the second dose of the COVID-19 vaccine, with and without a history of previous diagnosis of COVID-19. A sample size of 97 participants was calculated based on a 95% confidence level and a 50+/-10% neutralizing antibody positivity ratio. Healthcare workers who received the second dose of BBIBP-CorV vaccine (Sinopharm) against SARS CoV-2, who agreed to be part of the study and signed the informed consent form were included. Participants diagnosed with COVID-19 after vaccination and prior to antibody dosing, as well as those pregnant and those diagnosed with HIV/AIDS, cancer, autoimmune diseases or diseases associated with immunosuppression were excluded.
Procedures
This study was approved by the Institutional Ethics Committee of the Hospital Nacional Hipólito Unanue and by the Hospital Nacional Carlos Lanfranco La Hoz. Informed consent was obtained from all participants before enrollment. Health workers who received the second dose of the BBIBP-CorV vaccine (Sinopharm) between the 4th and 12th post-vaccination week were selected for enrollment. Between the 26th and 30th of May, health workers from different areas of the Hospital Nacional Hipólito Unanue were enrolled as participants; on April 21, health workers from the Hospital Nacional Carlos Lanfranco La Hoz were enrolled. A study form was filled out including information on age, sex, profession, date of vaccination and history of COVID-19.
Venous blood collection
Blood was obtained from a peripheral vein, in 5 mL sample tubes with coagulation activator to obtain serum. Once the sera were obtained by centrifugation, they were stored at 20 °C until processing. Sera that were not processed on the same day were stored at +5 °C (+/- 3 °C) within the first two hours until processing within 14 days of sample collection, as recommended by the reagent manufacturer.
Method verification
Prior to sample analysis, we verified the precision of the measurement procedure, following the recommendations of the guideline for the verification of quantitative analysis procedures of the Instituto Nacional de Calidad (INACAL); the laboratory obtained a variation coefficient (VC) lower than the VC of the reagent manufacturer, so the result was accepted.
Analysis
Samples that met the acceptance criteria were analyzed using the Elecsys® Anti-SARS-CoV-2 S assay, an immunoassay for the quantitative in vitro detection of antibodies to the receptor binding domain (RBD) of the SARS-CoV-2 spike protein (S) in human serum and plasma. The cobas® e601 automated analyzer, which uses the electrochemiluminescence method, was used for measurement. The assay employs a recombinant protein representing the RBD of the S antigen in a dual-antigen sandwich assay format, which detects high-affinity antibodies to SARS-CoV-2. The detected antibody titers showed good correlation with neutralizing antibodies in neutralization assays 24 - 26; however, during the execution of this article the electrochemiluminescence method for in vitro quantitative detection of antibodies against the receptor binding domain (RBD) of the SARS-CoV-2 spicule (S) protein had only emergency use approval from the U.S. Food and Drug Administration (FDA). The linear range is 0.4 to 250 U/mL. A result above 0.8 U/mL is interpreted as reactive 31 , 32; however, suitable cut-off points could not be determined because values above 250 U/mL could not be considered for statistical analysis. Internal quality control was carried out with Preci Control Anti-SARS-CoV-2 S.
Tests were conducted in the laboratory of the Hospital Nacional Hipólito Unanue for the participants from that facility and in the Instituto Nacional de Salud for the participants from the Hospital Carlos Lanfranco La Hoz.
Statistical analysis
The data were entered into a database created in Microsoft Excel and subsequently exported to a dta file. Data processing and analysis were conducted in the statistical program Stata v16.0 (Stata Corporation, College Station, Texas, USA). Numerical variables are presented with the appropriate measures of central tendency and dispersion, according to their distribution. Normality was assessed using histograms and formal tests. Categorical variables are presented as frequencies and percentages. Antibody titers were evaluated by presenting their measures of central tendency and dispersion according to their distribution. The geometric mean concentration is used for antibody titers because titer data generally do not fit a linear scale. When data are skewed and not normally distributed, it is recommended to calculate the geometric mean rather than the arithmetic mean, which may not provide a good representation of the results 33. A 95% confidence interval and a p-value of less than 0.05 were considered significant. Spearman’s correlation was used for comparison of antibody titers against numerical variables, and the Mann Whitney or Kruskall Wallis test was used for the comparison with categorical variables. Due to the different times of vaccination and sample collection techniques in the two hospitals, one group of participants underwent antibody dosing between 4- and 6-weeks post-vaccination while another group of participants underwent dosing between 8- and 12-weeks post-vaccination. The difference in vaccination times was considered as an additional variable. A multivariate analysis was carried out using the robust linear regression method including age, sex, history of COVID-19 and time of vaccination as independent variables. It was not possible to collect pre-vaccination sera in order to compare the increase in pre-vaccination sera in the studied individuals.
Ethical considerations
Our study respected the ethical principles in research according to the Helsinki Principles: autonomy, nonmaleficence, justice and beneficence. The study was authorized by the ethics committees of the Hipólito Unanue National Hospital (038-2021-CIEI- HNHU) and by the Carlos Lanfranco La Hoz Hospital. To participate in the study, patients voluntarily signed an informed consent form. The study protocol has been submitted to the Registry of Health Research Projects (PRISA) under code: EI00000001792.
RESULTS
A total of 168 participants were included, 108 from the Hospital Nacional Hipólito Unanue and 60 from the Hospital Lanfranco La Hoz. Most of the participants (60.1%) were female. The predominant occupation was physician (Table 1). None of the participants developed COVID-19 during the follow-up of up to 3 months after antibody dosing.
Table 1 Participants’ employment and post-vaccination symptomatology.
| Employment | n | % | Symptoms after vaccination | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Headache 31 (18.5%) | Fatigue 22 (13.1%) | Arm pain 9 (5.7%) | Increased sleep 6 (3.6%) | Dizziness 4 (2.4%) | Nausea 6 (3.6%) | Fever 2 (1.2%) | Other symptoms 6 (3.7%) | |||
| Physician | 67 | 39.8 | 13 (19.4) | 10 (14.9) | 5 (7.5) | 2 (3.0) | 3 (4.5) | 1 (1.5) | 0 (0.0) | 1 (1.5) |
| Administrative | 33 | 19.6 | 5 (15.2) | 4 (12.1) | 1 (3.0) | 2 (6.1) | 0 (0.0) | 2 (6.1) | 0 (0.0) | 0 (0.0) |
| Nursing Tech. | 28 | 16.7 | 7 (25.0) | 3 (10.7) | 0 (0.0) | 1 (3.6) | 0 (0.0) | 1 (3.6) | 1 (3.6) | 3 (10.7) |
| Technologist | 17 | 10.1 | 2 (11.8) | 1 (5.9) | 2 (11.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.9) | 1 (5.9) |
| Nurse | 13 | 7.7 | 2 (15.4) | 3 (23.1) | 0 (0.0) | 1 (7.7) | 1 (7.7) | 2 (15.4) | 0 (0.0) | 1 (7.7) |
| Obstetrician | 8 | 4.7 | 2 (25.0) | 1 (12.5) | 1 (12.5) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Pharmacy Tech. | 2 | 1.2 | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
Antibody titers against the receptor binding domain (RBD) of the SARS CoV-2 S protein
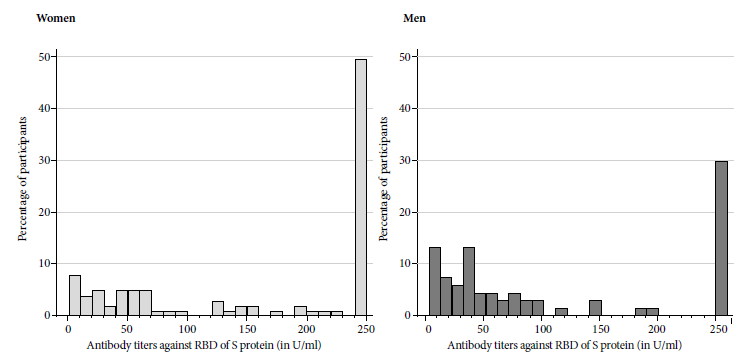
All participants developed antibodies against the RBD domain. The median was 137.05 with an interquartile range between 40.04 and 250. The minimum value was 1.78 U/mL. Seventeen participants (10.1%) had values below 10 U/mL and 70 (41.7%) had values equal to or above 250, the maximum antibody level determined by the test. The geometric mean was 82.6 (95% CI: 67.8-100.6). In patients with a history of COVID-19 the geometric mean was 219.51 (95% CI 195.04-247.06). All patients with a history of hospitalization for COVID-19 had values greater than or equal to 250 U/mL. The overall and by sex distribution of antibodies can be seen in Figure 1 and 2.
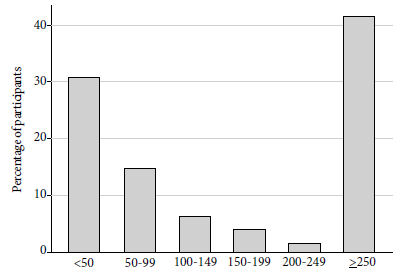
Figure 1 Distribution of antibody titers against the receptor binding domain of S protein in U/L in health personnel from two general hospitals. Lima, Peru.
Bivariate analysis
Females had higher antibody levels (p=0.04) (geometric mean of 100.97; 95% CI: 81.11-125.70) than males (geometric mean of 60.54; 95% CI: 41.90-87.48). The antibody levels of patients in whom antibodies were measured between weeks 4 and 6 post-vaccination were significantly higher (geometric mean 129 U/mL; 95% CI: 100.93-166.78) than those in whom antibodies were determined between 8- and 12-weeks post-vaccination (geometric mean 62.23 U/mL; 95% CI: 47.46-81.59).Patients with a history of COVID had significantly higher levels (geometric mean 219.51; 95% CI: 195.03-247.06) than those without a history of COVID (geometric mean 38.97; 95% CI: 30.39-49.96). Among patients with COVID history, antibody levels were found at or above 250 U/mL in 88% of cases compared to 6% in those without a history of COVID (p<0.001). No association was found between age and antibody titers (Spearman correlation coefficient -0.04; p=0.56). Fourteen participants older than 60 years were included; the geometric mean of this group of participants was 53.44 (95%CI 21.29-134.16), compared to 86.11 (95%CI 70.28- 105.49) with no statistically significant difference (p=0.28). No association was found between working in COVID-19 areas developing post-vaccination symptoms with the antibody titer. Comparison details are shown in Table 2.
Table 2 Medians and geometric means of antibody levels against the S protein receptor binding domain in U/L in health personnel from two general hospitals. Lima, Peru.
| Sex | n = 168 | % | Geometric mean (95% CI) | Median (IQR) | p-value a |
|---|---|---|---|---|---|
| Women | 102 | 60.7 | 100.97 (81.11-125.70) | 150.30 (51.06 - 250) | 0.04 |
| Men | 66 | 39.3 | 60.54 (41.90-87.48) | 77.22 (22.74 -250) | |
| Diabetes mellitus | |||||
| No | 162 | 96.4 | 82.11 (67.50-99.88) | 122.95 (39.88-250) | 0.27 |
| Yes | 6 | 3.6 | 96.80 (9.31-1007.00) | 250 (206.50-250) | |
| Arterial hypertension | |||||
| No | 158 | 94.0 | 79.83 (65.03- 98.00) | 121.40 (39.88-250) | 0.18 |
| Yes | 10 | 6.0 | 141.35 (63.46- 314.83) | 250 (206.50-250) | |
| Asthma | |||||
| No | 160 | 95.2 | 84.00 (68.68- 102.72) | 143.75 (40.04-250) | 0.43 |
| Yes | 8 | 4.8 | 58.96 (16.21-214.44) | 83.91 (24.66-233.5) | |
| Works in COVID-19 area | |||||
| No | 92 | 54.8 | 89.75 (68.28-117.97) | 195.90 (45.08-250) | 0.23 |
| Yes | 76 | 45.2 | 74.68 (55.82-99.92) | 104.39 (31.22-150) | |
| COVID-19 history | |||||
| No | 95 | 56.5 | 38.97 (30.39-49.96) | 47.45(20.52-90.39) | <0.001 |
| Yes | 73 | 43.5 | 219.51 (195.04-247.06) | 250 (250-250) | |
| Time between second dose and antibody measurement | |||||
| 4 to 7 weeks | 62 | 36.9 | 129.74 (100.93-166.78) | 250 (62.30-250) | <0.001 |
| 10 to 12 weeks | 106 | 63.1 | 62.23 (47.46-81.59) | 72.38 (24.09-250) | |
| Symptoms after second vaccine dose | |||||
| No | 90 | 53.6 | 88.36 (68.56-113.88) | 143.75 (41.98-250) | 0.77 |
| Yes | 78 | 46.4 | 76.40 (55.76-104.69) | 122.90 (34.83-250) |
a Mann Whitney U test to evaluate median difference
IQR: interquartile range; 95% CI: 95% confidence intervals.
Multivariate analysis
In the multivariate model (Table 3), history of COVID-19 had a significant association with a higher antibody titer (beta coefficient=213.87). This can be interpreted as the difference (adjusted for sex and time since the second dose) in antibody titer between those participants with history of COVID-19 compared to those without such a history. Male sex was associated with lower antibody titer (beta coefficient=-8.37). Likewise, the time between the second dose and the measurement of antibody titers was also associated with lower antibody titers (beta coefficient=-6.67).
Table 3 Robust regression model for antibody levels against the S protein receptor binding domain in U/L in health personnel from two general hospitals. Lima, Peru.
| Variable | Beta coefficient | 95% CI | p-value |
|---|---|---|---|
| COVID-19 history | 213.87 | 208.24; 219.49 | <0.001 |
| Men | -8.37 | -14.09; -2.65 | 0.004 |
| Dosage 10 to 12 weeks post-vaccination a | -6.67 | -12.51; -0.82 | 0.026 |
a Compared with those vaccinated at 4 to 7 weeks.
95% CI: 95% confidence intervals.
DISCUSSION
Our study demonstrated the presence of antibody responses in all participants. This corroborates the data from other studies and confirms an adequate immunogenicity in the humoral response. Our data are concordant with the clinical trial published by Xia et al. who found adequate immunologic responses in a population of 143 vaccinees, where 100% of participants had increased neutralizing antibody titers compared to pre-immunization baseline 5. Likewise, a recent study carried out by the Instituto Nacional de Salud provides information quite similar to our findings; in this study 95 persons with no history of infection and 34 persons with a history of SARS-CoV-2 infection were evaluated. In the first group, 21 days after the first immunization, 31% of the participants produced IgG antibodies for the B.1.1 lineage, and 15% produced IgG antibodies against the Gamma variant (P.1 ); but 21 days after the second dose with BBIBP-Cor-V vaccine, these percentages increased to 99 and 96%, respectively for each lineage. In the group with history of SARS-CoV-2 infection, 21 days after the first dose, 82% of the participants produced IgG antibodies for the B.1 .1 lineage and 77% produced IgG antibodies against the Gamma variant (P.1), and 21 days after the second dose 100% produced IgG antibodies against the B.1.1 lineage and the Gamma variant 34.
Among the factors associated with a higher antibody titer, history of COVID-19 clearly stands out. This is predictable and to some extent limiting, since it is not possible to distinguish the immune responses to vaccination from those originated by the SARS CoV-2 infection itself. In any case, our data are consistent with a higher production of antibodies in those persons with history of COVID-19. This has also been evidenced by a study published by Xiangyu Chen et al. in which they observed that the neutralizing antibody titers showed a positive relationship with history of the disease and its severity, being higher in those patients with severe disease 22.
The T-cell-mediated immunogenic response appears to be more intense in women, while the levels of several chemokines and innate immune cytokines appear to be higher in male patients. The severity of COVID-19 is known to be lower in women 35, and these differences in the immune response between women and men could explain the differences in disease severity.
Although the sample is small, immunogenicity does not appear to decrease with age. However, studies with a larger population are required to determine whether there are significant differences in this regard. In this regard, it is important to note that data from persons over 60 years of age is scarce and often misinterpreted as vaccine ineffectiveness. However, published studies have found 100% seroconversion rates in people older than 60 years, depending on vaccine doses 34.
On the other hand, history of COVID-19 is associated with increased immune responses. It has been suggested that only one vaccine dose may be necessary in patients with history of COVID-19 and an adequate immune system 36. However, there is a significant number of patients who do not achieve high antibody levels, so we could not make a recommendation in this regard. The correlation between titers and the ability to protect against SARS-CoV-2 infection remains uncertain.
A limitation for our study is the fact that we did not determine the presence of neutralizing antibodies directly, so we cannot necessarily infer that the evidenced immunological responses necessarily imply protection from the clinical point of view; likewise, the test used for the detection of antibodies against the receptor binding domain (RBD) of the spike protein (S) has only been approved for emergency use. However, a good correlation with the neutralization tests by plaque reduction has been observed. Another limitation for our study was having too wide cut-off points, not being able to compare values above 250 U/mL; however, the study seeks to measure seroconversion and found that all participants achieved seroconversion regardless of the titers found. Another limitation was not having pre-vaccination sera to compare the increased effect provided by the vaccine; however, for the purposes of this study this limitation did not interfere with the conclusions. On the other hand, response against the new variants may be different, as has been evidenced by the lower neutralizing capacity of the BBIBP-CorV vaccine against variant B.1.351 37.
Considering that the vaccine we used is based on an inactivated virus, it is reasonable to hypothesize that protection may last for a limited time, given that natural immunity in the case of COVID-19 seems to be diluted after a few months, as suggested by experiences in Manaus where, despite having obtained a prevalence of more than 70% (and thus postulated herd immunity), a second wave of significant proportions emerged. Therefore, it may be reasonable to consider a booster dose after a few months for health personnel, particularly in front-line workers, in accordance with the recommendations issued by regulatory institutions such as the Center for Disease Control and Prevention and the World Health Organization, always prioritizing collective health. However, we consider it is a mistake to state that the chosen vaccine has no utility, which may motivate reluctance to vaccination; such as the refusal of the teachers' union in rural areas who have been offered vaccination with BBIBP-CorV.
In conclusion, our data showed adequate immunogenicity of the BBIBP-CorV vaccine (Sinopharm) as assessed by antibodies against RBD. However, it is necessary to evaluate the correlation between the magnitude of titers and protection against COVID-19 and the time of protection conferred by the vaccine.
REFERENCES
1. Jackson LA, Anderson EJ, Rouphael NG, Roberts PC, Makhene M, Coler RN, et al. An mRNA Vaccine against SARS-CoV-2 - Preliminary Report. N Engl J Med. 2020;383(20):1920-31. doi: 10.1056/NEJMoa2022483. [ Links ]
2. Graham BS. Rapid COVID-19 vaccine development. Science. 2020;368(6494):945-6. doi: 10.1126/science.abb8923. [ Links ]
3. Krammer F. SARS-CoV-2 vaccines in development. Nature. 2020;586(7830):516-27. doi: 10.1038/s41586-020-2798-3. [ Links ]
4. Zhu F-C, Li Y-H, Guan X-H, Hou L-H, Wang W-J, Li J-X, et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. The Lancet. 2020;395(10240):1845-54. doi: 10.1016/S0140-6736(20)31208-3. [ Links ]
5. Xia S, Zhang Y, Wang Y, Wang H, Yang Y, Gao GF, et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBIBP-CorV: a randomized, double-blind, placebo-controlled, phase 1/2 trial. Lancet Infect Dis. 2021;21(1):39-51. doi: 10.1016/S1473-3099(20)30831-8. [ Links ]
6. Folegatti PM, Ewer KJ, Aley PK, Angus B, Becker S, Belij-Rammerstorfer S, et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, single-blind, randomized controlled trial. Lancet. 2020;396(10249):467-478. doi: 10.1016/S0140-6736(20)31604-4. [ Links ]
7. Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020;383(27):2603-2615. doi: 10.1056/NEJMoa2034577. [ Links ]
8. Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384(5):403-16. doi: 10.1056/NEJMoa2035389. [ Links ]
9. Voysey M, Clemens SAC, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99-111. doi: 10.1016/S0140-6736(20)32661-1. [ Links ]
10. Cyranoski D. Arab nations first to approve Chinese COVID vaccine - despite lack of public data. Nature. 2020 Dec;588(7839):548. doi: 10.1038/d41586-020-03563-z. [ Links ]
11. Ramasamy MN, Minassian AM, Ewer KJ, Flaxman AL, Folegatti PM, Owens DR, et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): a single-blind, randomised, controlled, phase 2/3 trial. Lancet. 2021;396(10267):1979-1993. doi: 10.1016/S0140-6736(20)32466-1. [ Links ]
12. Walsh EE, Frenck RW, Falsey AR, Kitchin N, Absalon J, Gurtman A, et al. Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates. N Engl J Med. 2020;383(25):2439-50. doi: 10.1056/NEJMoa2027906. [ Links ]
13. Dillner J, Elfström KM, Blomqvist J, Eklund C, Lagheden C, Nordqvist-Kleppe S, et al. Antibodies to SARS-CoV-2 and risk of past or future sick leave. Sci Rep. 2021;11(1):5160. [ Links ]
14. Long Q-X, Liu B-Z, Deng H-J, Wu G-C, Deng K, Chen Y-K, et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat Med. 2020;26(6):845-848. doi: 10.1038/s41591-020-0897-1. [ Links ]
15. Chen W, Xu Z, Mu J, Yang L, Gan H, Mu F, et al. Antibody response and viraemia during the course of severe acute respiratory syndrome (SARS)-associated coronavirus infection. J Med Microbiol. 2004;53(Pt 5):435-8. [ Links ]
16. Payne DC, Iblan I, Rha B, Alqasrawi S, Haddadin A, Al Nsour M, et al. Persistence of Antibodies against Middle East Respiratory Syndrome Coronavirus. Emerg Infect Dis. 2016;22(10):1824-6. doi: 10.3201/eid2210.160706. [ Links ]
17. Prévost J, Gasser R, Beaudoin-Bussières G, Richard J, Duerr R, Laumaea A, et al. Cross-Sectional Evaluation of Humoral Responses against SARS-CoV-2 Spike. Cell Rep Med. 2020;1(7):100126. doi: 10.1016/j.xcrm.2020.100126. [ Links ]
18. Manisty C, Treibel TA, Jensen M, Semper A, Joy G, Gupta RK, et al. Time series analysis and mechanistic modelling of heterogeneity and sero-reversion in antibody responses to mild SARS CoV-2 infection. EBioMedicine. 2021;65:103259. doi: 10.1016/j.ebiom.2021.103259. [ Links ]
19. Wajnberg A, Amanat F, Firpo A, Altman DR, Bailey MJ, Mansour M, et al. Robust neutralizing antibodies to SARS-CoV-2 infection persist for months. Science. 2020;370(6521):1227-1230. doi: 10.1126/science.abd7728. [ Links ]
20. Iyer AS, Jones FK, Nodoushani A, Kelly M, Becker M, Slater D, et al. Persistence and decay of human antibody responses to the receptor binding domain of SARS-CoV-2 spike protein in COVID-19 patients. Sci Immunol. 2020;5(52):eabe0367. doi: 10.1126/sciimmunol.abe0367. [ Links ]
21. Glück V, Grobecker S, Tydykov L, Salzberger B, Glück T, Weidlich T, et al. SARS-CoV-2-directed antibodies persist for more than six months in a cohort with mild to moderate COVID-19. Infection. Infection. 2021;49(4):739-746. doi: 10.1007/s15010-021-01598-6. [ Links ]
22. Muruato AE, Fontes-Garfias CR, Ren P, Garcia-Blanco MA, Menachery VD, Xie X, et al. A high-throughput neutralizing antibody assay for COVID-19 diagnosis and vaccine evaluation. Nat Commun. 2020;11(1):4059. doi: 10.1038/s41467-020-17892-0. [ Links ]
23. Bewley KR, Coombes NS, Gagnon L, McInroy L, Baker N, Shaik I, et al. Quantification of SARS-CoV-2 neutralizing antibody by wild-type plaque reduction neutralization, microneutralization and pseudotyped virus neutralization assays. Nat Protoc. 2021;16(6):3114-3140. doi: 10.1038/s41596-021-00536-y. [ Links ]
24. Kohmer N, Westhaus S, Rühl C, Ciesek S, Rabenau HF. Brief clinical evaluation of six high-throughput SARS-CoV-2 IgG antibody assays. J Clin Virol. 2020;129:104480. doi: 10.1016/j.jcv.2020.104480. [ Links ]
25. Müller L, Ostermann PN, Walker A, Wienemann T, Mertens A, Adams O, et al. Sensitivity of commercial Anti-SARS-CoV-2 serological assays in a high-prevalence setting. medRxiv. 2020;2020.06.11.20128686. doi: 10.1101/2020.06.11.20128686v2. [ Links ]
26. Rubio-Acero R, Castelletti N, Fingerle V, Olbrich L, Bakuli A, Wölfel R, et al. In Search of the SARS-CoV-2 Protection Correlate: Head-to-Head Comparison of Two Quantitative S1 Assays in Pre-characterized Oligo-/Asymptomatic Patients. Infect Dis Ther. 2021;1-14. doi: 10.1007/s40121-021-00475-x. [ Links ]
27. Raraz-Vidal JG, Allpas-Gomez HL, Torres-Salome FK, Cabrera-Patiño WM, Alcántara-Leyva LM, Ramos-Gómez RP, et al. Condiciones laborales y equipos de protección personal contra el Covid-19 en personal de salud, Lima-Perú. Rev Fac Med Humana. 2021;21(2):335-45. doi: 10.25176/rfmh.v21i2.3608. [ Links ]
28. Colegio Médico del Perú. Médicos fallecidos por COVID-19 en Iberoamérica [Internet]. Colegio Médico del Perú - Consejo Nacional. [citado 19 de julio de 2021]. Disponible en: https://www.cmp.org.pe/medicos-fallecidos-por-covid-19-en-iberoamerica. [ Links ]
29. Galán-Rodas E, Tarazona-Fernández A, Palacios-Celi M. Riesgo y muerte de los médicos a 100 días del estado de emergencia por el COVID-19 en Perú. Acta Med Peru. 2020;37(2):119-21. doi: 10.35663/amp.2020.372.1033. [ Links ]
30. Colegio Médico del Perú. Vacunómetro-CMP [Internet]. Colegio Médico del Perú - Consejo Nacional. [citado 30 de junio de 2021]. Disponible en: https://www.cmp.org.pe/vacunometro-cmp/. [ Links ]
31. ROCHE. Elecsys(r) Anti-SARS-CoV-2 S. Package Insert 2020-09, V1.0; Material Numbers 09289267190 and 09289275190 [Internet]. Diagnostics. 2021 [citado 12 de marzo de 2021]. Disponible en: https://diagnostics.roche.com/global/en/products/params/elecsys-anti-sars-cov-2-s.html. [ Links ]
32. Riester E, Findeisen P, Hegel JK, Kabesch M, Ambrosch A, Rank CM, et al. Performance evaluation of the Roche Elecsys Anti-SARS-CoV-2 S immunoassay. medRxiv. 2021;03.02.21252203. doi: 10.1101/2021.03.02.21252203v1. [ Links ]
33. World Health Organization. Guidelines on clinical evaluation of vaccines: regulatory expectations. Geneva: WHO; 2021, Disponible en: https://www.who.int/publications/m/item/WHO-TRS-1004-web-annex-9. [ Links ]
34. García-Mendoza MP, Fernandez-Navarro M. Informe técnico: Anticuerpos IgG y anticuerpos neutralizantes en personas que han recibido la vacuna BBIBP-CorV [Internet]. Repositorio Científico. Disponible en: https://repositorio.ins.gob.pe/xmlui/bitstream/handle/INS/1311/Rep21-ant.pdf?sequence=1&isAllowed=y. [ Links ]
35. Takahashi T, Ellingson MK, Wong P, Israelow B, Lucas C, Klein J, et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature. 2020;588(7837):315-20. doi: 10.1038/s41586-020-2700-3. [ Links ]
36. Dolgin E. Is one vaccine dose enough if you've had COVID? What the science says. Nature. 2021;595(7866):161-2. doi: 10.1038/d41586-021-01609-4. [ Links ]
37. Wang G-L, Wang Z-Y, Duan L-J, Meng Q-C, Jiang M-D, Cao J, et al. Susceptibility of circulating SARS-CoV-2 variants to neutralization. N Engl J Med. 2021;384(24):2354-6. doi: 10.1056/NEJMc2103022. [ Links ]
Funding: self-funded. The supplies for antibody dosing at the Hospital Hipólito Unanue were donated by the ROCHE laboratory, which had no role in this research.
Cites as: Soto A, Charca-Rodríguez FdM, Pareja-Medina M, Fernández- Navarro M, Altamirano-Cáceres K, Sierra Chávez E, et al. Evaluation of the humoral response induced by BBIBP-CoRV vaccine by determining neutralizing antibodies in Peruvian healthcare personnel. Rev Peru Med Exp Salud Publica. 2021;38(4):493-500. doi: https://doi.org/10.17843/rpmesp.2021.384.9244.
Received: August 09, 2021; Accepted: December 15, 2021











 text in
text in