Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634
Rev. perú. med. exp. salud publica vol.38 no.4 Lima Oct./Dec. 2021 Epub Dec 02, 2021
http://dx.doi.org/10.17843/rpmesp.2021.384.7596
Originales breves
Validation and evaluation of RT-PCR real time in house test to detection of SARS-CoV-2 using specific RdRp gene and GAPDH endogen control
1 Laboratorio de Referencia Nacional de Virus Respiratorios, Centro Nacional de Salud Pública, Instituto Nacional de Salud, Lima, Perú.
2 Laboratorio de Patología Aviar, Facultad de Medicina Veterinaria, Universidad Nacional Mayor de San Marcos, Lima, Perú.
3 Laboratorio de Virología Animal, Instituto de Biología, Universidad de Campinas, Campinas/SP, Brasil.
4 Laboratorio de Referencia Nacional de Biotecnología y Biología Molecular, Centro Nacional de Salud Pública, Instituto Nacional de Salud, Lima, Perú.
The present work validated and evaluated a duplex real-time RT-PCR using specific primers and probes for genes RdRp from SARS-CoV-2 and GAPDH from humans; the latter was used as an endogenous control in all reactions. We evaluated the specificity, the sensitivity, the robustness, the reproducibility, the repeatability, the comparability, and the limit of detection. The predictive positive and negative values (PPV and PNV, respectively) and all the parameters evaluated using our duplex real-time RT-PCR was 100%. The detection limit was 100 copies/µL according to the acceptance criteria established for the validation of this protocol. Our duplex real-time RT-PCR demonstrated to be a good alternative for the diagnosis of COVID-19; in addition, this PCR was used adequately in suspicion of COVID-19, allowing it to control the number of false-negatives.
Keywords: RT-PCR SARS-CoV-2 testing; SARS-CoV-2; Molecular Diagnosis; COVID-19
INTRODUCTION
In December 2019, China reported the presence of several cases of atypical pneumonia in the city of Wuhan 1 resulting in death within a few days. The etiologic agent was identified as a new coronavirus that was named SARS-CoV-2 and the disease was named COVID-19 (Coronavirus Disease 2019). By January 2020, despite containment measures taken by the Chinese government, more cases were reported in bordering countries and in Europe. The increase in COVID-19 cases was so rapid that the World Health Organization (WHO) declared the disease a pandemic and urged countries to adopt control measures to prevent its spread 2. By the end of February 2020, the disease had already reached the Americas. In early March 2020, the first case of COVID-19 was reported in Peru, and the following week the government adopted control measures by closing the borders and declaring a nationwide quarantine.
One of the essential strategies to control the disease is the rapid identification of the SARS-CoV-2 virus. Molecular testing in Peru was implemented by the National Respiratory Virus Reference Laboratory of the National Institute of Health (LRNVR-INS) following recommendations of the Pan American Health Organization (PAHO) and WHO. These recommendations, for molecular diagnosis, were based on the protocol of Corman et al 3. This protocol describes the use of primers and probes for the detection of the E and RdRP genes using the real-time RT-PCR technique in two stages: first the amplification of the E gene and the positive samples would be confirmed in a second reaction amplifying the RdRP gene. In order to improve the detection system to make it faster and simpler without losing sensitivity and specificity, it was proposed to standardize a real-time RT-PCR protocol in duplex format. The present study aimed to validate and evaluate a duplex real-time RT-PCR, which consists of simultaneous amplification (in a single tube) of the diagnostic marker RdRP and an endogenous internal control human GAPDH. In addition, the evaluation of its usefulness in the detection of SARS-CoV-2 virus in clinical samples is presented.
KEY MESSAGES
Motivation for the study: Peru was one of the countries most affected by the COVID-19 pandemic, with a large number of cases and deaths.
Main findings: This standardized test is a good alternative for the diagnosis of COVID-19; in addition, the test was successfully applied in people suspected of having COVID-19, allowing to control the number of false negatives.
Implications: Early and successful diagnosis of COVID-19 will help to implement control measures to reduce the speed of SARS-CoV-2 virus transmission in Peru.
THE STUDY
A study was conducted to validate and evaluate a diagnostic test.
Biological samples
The samples used in this study are nasal and pharyngeal swab (nasal swab and pharyngeal swab (HNF) obtained from the LRNVR-INS virotheque, obtained from symptomatic patients suspected of having COVID-19 in the period from March 2020 to June 2021.
Assay development
Viral RNA was purified using the QIAamp viral RNA Mini kit (QIAGEN, Hilden, Germany) following the manufacturer's instructions. The real time duplex RT-PCR assay was performed using the reported primers and probes (Table 1). The final reaction volume was 20 µL, this contained Mix QPCR Probe 4x (BiotechRabbit), RTase enzyme, PCR water, primers (RdRP_F, RdRP_R, GAPDH_F and GAPDH_R) and probes (RdRp_P2, GAPDH_P). Reaction tubes were placed in a RotorGene Q real-time thermal cycler (QIAGEN, Germany) and cycling was used at 50 °C for 10 minutes for reverse transcription, 95 °C for 3 minutes initial denaturation followed by 45 cycles of 95 °C for 10 seconds and 58 °C for 30 seconds (with acquisition for FAM and ROX), ending with 40 °C for 30 seconds. Positive results were defined with Ct (Cycle Threshold) values < 37 for the RdRP gene (FAM) and Ct < 40 for the GAPDH gene (ROX).
Table 1 Sequence of primers and probes of the duplex RT-qPCR.
| GENE | Primers and probes | 5’->3’ sequence | Concentration by reaction | Volume by reaction |
|---|---|---|---|---|
| RdRP | RdRp_F | GTGARATGGTCATGTGTGGCGG | 0.4 µM | 0.8 µL |
| RdRp_P2 | FAM-CAGGTGGAACCTCATCAGGAGATGC-BBQ | 0.4 µM | 0.8 µL | |
| RdRp_R | CARATGTTAAASACACTATTAGCATA | 0.2 µM | 0.4 µL | |
| GAPDH | GAPDH_F | GTGAAGGTCGGAGTCAACGG | 0.4 µM | 0.8 µL |
| GAPDH_R | TCAATGAAGGGGTCATTGATGG | 0.4 µM | 0.8 µL | |
| GAPDH_P | ROX - CGCCTGGTCACCAGGGCTGC- BHQ2 | 0.2 µM | 0.4 µL |
Assay validation
To evaluate the sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) parameters of the duplex RT-qPCR assay, 55 positive cases diagnosed with the method recommended by PAHO (Corman et al.) 3 ) confirmed by sequencing and 50 negative cases were used. These samples were selected for convenience because it was a new disease and there were not enough samples confirmed by sequencing.
A positive control (quantified SARS-CoV-2 synthetic RNA) donated by PAHO was used to evaluate the detection limit of the assay. To evaluate the diagnostic specificity, clinical samples of FNH positive for influenza A (n=3), influenza B (n=3), respiratory syncytial virus (n=3), adenovirus (n=3), metapneumovirus (n=3), rhinovirus (n=3), dengue virus (n=2) and Streptococcus pneumoniae (n=2) were used; These samples came from the LRNVR-INS virotek.
Repeatability was evaluated with four SARS-CoV-2 positive samples for five days, maintaining the same conditions: analyst, environment, sample and equipment. Reproducibility was evaluated with 10 samples (5 positive and 5 negative) by 2 analysts. Robustness was evaluated with 4 SARS-CoV-2 positive samples using 3 different concentrations (0.6 µM, 0.4 µM and 0.2 µM) of the primers and probes. Comparability was evaluated with 10 samples (5 SARS-CoV-2 positive and 5 negative samples) in two different thermal cyclers. The Ct values obtained were used to obtain the mean, standard deviation and coefficient of variation. The limit of detection was evaluated by performing serial dilutions from 10-1 to 10-7 in triplicate by determining the value of the efficiency coefficient for the RdRP gene, a parameter calculated by RotorGene software (QIAGEN).
Evaluation of clinical samples
These samples were collected between March 17 and 27, 2020 and were sent to the LRNVR-INS for diagnostic confirmation in the framework of the laboratory surveillance of respiratory infections in Peru.
To evaluate whether the test detected variants of concern (VOC) or variants of interest (VOI), 31 samples collected between April and August 2021 and referred to LRNVR-INS for diagnostic confirmation and previously characterized by Next Generation Sequencing (NGS) whole genome sequencing were evaluated: 10 samples of Lambda (C.37 ), 4 from Gamma (P.1), 4 from Alpha (B.1.1.7), 2 from Epsilon (B.1.429), 8 from Delta (AY.12), and 3 from Mu (B.1.621).
Bioinformatic evaluation
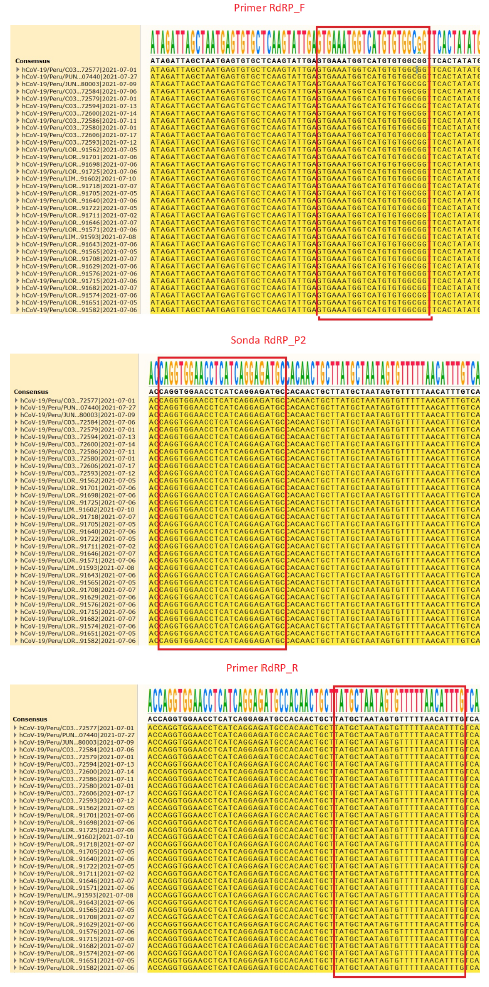
To evaluate the conservation of RdRP marker primer and probe binding sites, an alignment of 5600 whole genome sequences of Peruvian SARS-CoV-2 isolates collected between March 2020 and June 2021 (downloaded from the GISAID database [https://www.gisaid.org/]) was performed using MAFFT software version 7 (https://mafft.cbrc.jp/alignment/server/). This alignment was visualized using SnapGene Viewer software version 5.3.2 (https://www.snapgene.com/snapgene-viewer/).
Statistical analysis
Comparison of Ct values was performed using the Stata program version 16 (Stata Corporation, College Station, Texas, USA). The kappa index was calculated using the Statistical Package for the Social Sciences (SPSS) program. The degree of concordance between duplex RT-qPCR and real-time RT-PCR test results was determined according to Cormann et al. 3. In addition, sensitivity, specificity, NPV and NPV were calculated; and the coefficient less than 1% (close to 0) was considered to indicate the existence of little variability in the data. The measures were obtained through point estimators and 95% confidence intervals (95%CI).
Ethical criteria
Standardization and laboratory validation did not require evaluation by the Institutional Research Ethics Committee (CIEI), since the samples used were obtained in routine activities established within the Action Plan of the National Institute of Health for the prevention, diagnosis and control of COVID-19, approved with RJ-103-2020-J-OP- INS-2020.
FINDINGS
The duplex RT-qPCR assay for the detection of SARS-CoV-2 virus presented results of 100% sensitivity, 100% specificity, 100% PPV and 100% NPV (Table 2), 100% diagnostic specificity (supplementary material, Table S4), 100% concordance in repeatability parameters (CV=0.00975, SD=0.232), reproducibility (CV=0, 638, SD=0.126), robustness (CV=0.00575, SD=0.141) and comparability (CV=0.52, SD=0.124) (supplementary material, table S5-S8), with a detection limit of 100 copies/µL (Figure 1) (supplementary material, table S9) and an efficiency of 93%, a kappa index value of 1 was obtained compared to the method of Corman et al. according to the qualitative results. The Ct values obtained for the specific marker RdRP were not affected when used in the duplex format (supplementary material, Table S1).
Table 2 Determination of sensibilidad, especificidad, valor predictivo positivo y valor predictivo negativo.
| Duplex real time RT-PCR | Reference test (Corman et al.) | Total | |
|---|---|---|---|
| Positive | Negative | ||
| Positive | 55 | 0 | 55 |
| Negative | 0 | 50 | 50 |
| Total | 55 | 50 | 105 |
Sensitivity: 100%
Specificity: 100%
Positive predictive value: 100%
Negative predictive value: 100%
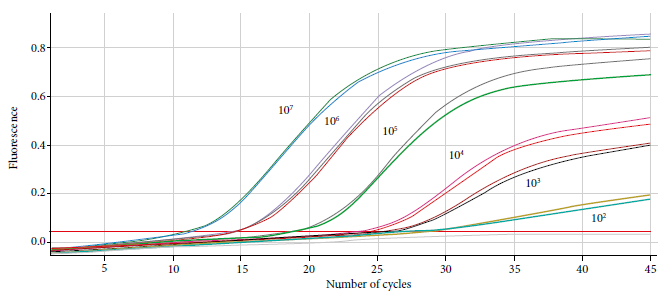
Figure 1 Fluorescence curves obtained during the evaluation of the limit of the duplex real-time RT-PCR test for the detection of SARS-CoV-2.
This test has been used to evaluate 3006 clinical samples obtaining a positivity of 9.2%, the samples belong to all age groups and all regions of the country (Supplementary material, table S3).
Additionally, this test detects VOC Delta and Gamma, and VOI Mu, Epsilon and Lambda reported during 2021 (Supplementary material, table S10). The bioinformatics analysis performed on 5600 genomes of Peruvian SARS-CoV-2 isolates reported in GISAID indicated that the primer and probe binding sites are conserved, except for the first forward of the RdRP gene which presents a nucleotide change at 2.9% of the genomes analyzed (Figure 2).
DISCUSSION
This study standardized and validated a duplex real-time RT-PCR assay using specific primers and probes for the RdRP gene of SARS-CoV-2, and for the human GAPDH gene as an endogenous control. The GAPDH gene, which produces a constitutive protein involved in many cellular functions such as glycolysis, nuclear RNA export, DNA replication and repair, and apoptosis ( 4; this same gene was described by Wong et al. 5 ) as a good internal endogenous control to decrease the possibility of obtaining false-negative results in the diagnosis of SARS-CoV-1. This strategy made it possible to ensure (in a single real-time PCR reaction) the quality of the sample and of the RNA extraction process. The use of internal endogenous control has been recommended by PAHO 6, and is used in different methodologies by the LRN- VR-INS, as in the diagnosis of influenza virus 7.
The validation parameters obtained were 100% in specificity, sensitivity and PPV and NPV when compared with the method reported by Corman et al. 4; additionally, these samples were characterized by whole genome sequencing. The same achievement was obtained for other laboratory validation parameters such as robustness, repeatability and reproducibility. The validation parameters were selected and established based on the experience of the LRNVR-INS research team and according to standards suggested by the WHO, described by Kosak et al. 8.
Given that WHO has suggested that the diagnosis of COVID-19 should be performed considering the amplification of at least two SARS-CoV-2 genes in two consecutive RT-PCR reactions, this strategy would not be widely feasible to implement in developing countries such as Latin America. Other researchers have described protocols or protocol adaptations such as Ishige et al. 9, who included the abl1 gene as an endogenous internal control in SARS-CoV-2 identification reactions. However, this gene produces a tyrosine kinase present in large quantities in the human cell, with the risk of reducing the analytical sensitivity of the test. On the other hand, Pezzi and his team 10 indicated that the MS2 gene (for the expression of the enzyme methionine synthetase 2) could be used as an endogenous control together with the RdRp marker reported by Corman et al. 3 reducing false negatives, presenting results similar to those obtained in this study, without problems with the performance of the reactions.
In Brazil, a group of researchers from the University of São Paulo 11 developed a protocol based on other more economical detection methods using conventional RT-PCR. These methods are not as specific as those using probes; however, they could be an alternative applicable in cases of health emergency and reagent supply crisis.
The results of the present study were successful as the assays met all acceptance criteria. In addition, the efficiency analyses indicated a low coefficient of variation, less than 1%. All these achievements indicated that the criteria for identification of samples as true positive or negative were adequate for a qualitative test. Additionally, this test has been used for the diagnosis of clinical nasal and pharyngeal swab specimens, ensuring consistency of molecular testing of symptomatic patients. Molecular diagnostic tests should be continuously updated to avoid being affected by mutations in viral genomes as in the case of SARS-CoV-2 and other pathogenic viruses, given that a mutation has been detected in one of the primers, we suggest its update to avoid loss of sensitivity of the test.
Among the limitations of the study is the limited availability of confirmed samples for validation due to the fact that this was a new disease, fortunately we had a set of samples with sequencing results. Another limitation is that this system uses only one diagnostic marker; however, bioinformatic analysis indicates that the primers are specific and sensitive for the detection of SAR-CoV-2.
In conclusion, validation of the duplex real-time RT-PCR test indicates that the test is sensitive and specific for the detection of infections caused by SARS-CoV-2 and has been successfully evaluated with clinical samples.
Acknowledgments: We thank PAHO for the donation of reagents and for establishing the collaboration to carry out the validations of the experiment. The authors also acknowledge and thank all the workers of the Microbiology and Biomedicine Laboratory of the National Institute of Health and all the people from other institutions involved in the collection, handling and processing of the samples.
REFERENCES
1. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708-1720. doi: 10.1056/neJMoa2002032. [ Links ]
2. World Health Organization. Statement on the third meeting of the International Health Regulations (2005) Emergency Committee regarding the outbreak of coronavirus disease (COVID-19) [Internet]; 2021. [citado 14 junio 2021]. Disponible en: https://www.who.int/news-room/detail/01-05-2020-statement-on-the-third-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-coronavirus-disease-(covid-19). [ Links ]
3. Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu D, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020;25(3):2000045. doi: 10.2807/1560-7917.ES.2020.25.3.2000045. [ Links ]
4. Sirover MA. New insights into an old protein: the functional diversity of mammalian glyceraldehyde-3-phosphate dehydrogenase. Biochim Biophys Acta. 1999;1432(2):159-184. doi: 10.1016/s0167-4838(99)00119-3. [ Links ]
5. Wong SC, Chan JK, Lee KC, Lo ES, Tsang DN. Development of a quantitative assay for SARS coronavirus and correlation of GAPDH mRNA with SARS coronavirus in clinical specimens. J Clin Pathol. 2005 Mar;58(3):276-280. doi: 10.1136/jcp.2004.016592. [ Links ]
6. Organización Mundial de la Salud. Pruebas diagnósticas para el SARS-CoV-2: orientaciones provisionales, 11 de septiembre de 2020. Washington DC: OMS; 2020. Disponible en: https://apps.who.int/iris/handle/10665/335830. [ Links ]
7. Marcos P, Huaringa M, Rojas N, Gutierrez V, Ruiton S, Gallardo E, et al. Detección de virus influenza A, B y subtipos A (H1N1) pdm09, A (H3N2) por múltiple RT-PCR en muestras clínicas. Rev Peru Med Exp Salud Publica. 2017;34(2):192-200. doi: 10.17843/rpmesp.2017.342.2054. [ Links ]
8. Kosack CS, Page AL, Klatser PR. A guide to aid the selection of diagnostic tests. Bull World Health Organ. 2017;95(9):639-645. doi: 10.2471/BLT.16.187468. [ Links ]
9. Ishige T, Murata S, Taniguchi T, Miyabe A, Kitamura K, Kawasaki K, et al. Highly sensitive detection of SARS-CoV-2 RNA by multiplex rRT-PCR for molecular diagnosis of COVID-19 by clinical laboratories. Clin Chim Acta. 2020;507:139-142. doi: 10.1016/j.cca.2020.04.023. [ Links ]
10. Pezzi L, Charrel RN, Ninove L, Nougairede A, Molle G, Coutard B, et al. Development and Evaluation of a duo SARS-CoV-2 RT-qPCR Assay Combining Two Assays Approved by the World Health Organization Targeting the Envelope and the RNA-Dependant RNA Polymerase (RdRp) Coding Regions. Viruses. 2020;12(6):686. doi: 10.3390/v12060686. [ Links ]
11. Dorlass EG, Monteiro CO, Viana AO, Soares CP, Machado RRG, Thomazelli LM, et al. Lower cost alternatives for molecular diagnosis of COVID-19: conventional RT-PCR and SYBR Green-based RT-qPCR. Braz J Microbiol. 2020;51(3):1117-1123. doi: 10.1007/s42770-020-00347-5. [ Links ]
Cite as: Rojas-Serrano N, Lope-Pari P, Huaringa-Nuñez M, Marques Simas PV, Palacios-Salvatierra R, Balbuena-Torres J, et al. Validation and evaluation of RT-PCR real-time in-house test to detection of SARS-CoV-2 using specific RdRp gene and GAPDH endogen control. Rev Peru Med Exp Salud Publica. 2021;38(4):595-600. doi: https://doi.org/10.17843/rpmesp.2021.384.7596.
Received: March 15, 2021; Accepted: November 03, 2021











 text in
text in