Introduction
The accelerated increasing of energy demand, depletion of oil resources, and the need to reduce the emission of greenhouse gases are the main reasons for developing sustainable and renewable energy and chemical resources (Putro et al. 2016). In this context, current research efforts are being focused on the production of biofuels and other useful chemicals from through lignocellulosic biomass conversion. Biofuels derived from lignocellulosic biomass, particularly from agricultural crops residues, are receiving attention around the world to attain multiple strategic objectives, such as mitigating climate change, energy security and the development of the rural economy (Nanda et al. 2015). In addition, agriculture wastes are abundant, renewable and profitable resources, constituting an attractive source of carbon for the production of sugars and other chemicals (Cortes-Tolalpa et al. 2017, Sarkar et al. 2012). Globally, the main crops are wheat, maize, rice, and sugarcane, which produce most of the lignocellulosic biomass (Carrillo-Nieves et al. 2019). After rice straw, wheat straw is the second largest lignocellulosic source in the world (Sarkar et al. 2012, Tian et al. 2018). In the other hand, production of quinoa is increasing due to its high nutritional value, as a consequence, large quantities of stalks accumulate as an unused byproduct (Gil-Ramirez et al. 2018).
Agricultural waste is composed of cellulose (30 - 50%), hemicellulose (20 - 40%) and lignin (15 - 25%) (Chen et al. 2013). Then, cellulose and hemicellulose are fermentable polysaccharides, which can be converted into fermentable sugars, while lignin is a non-fermentable polyphenolic compound (Arora et al. 2015). Cellulose is the major structural component of the plant cell wall, it is formed by linear chains of β-1,4-glucan that adhere to one another forming higher-order fibrous crystalline structures (Kont et al. 2013). Whereas, hemicellulose includes repetitive polymers of pentoses and hexoses, with different types of bonds, branches, and substitutions (Anwar et al. 2014, Kont et al. 2013). Cellulose fibrils are associated with hemicellulose, forming a complex network of polysaccharides, which in turn is embedded in the matrix of lignin (Ding et al. 2012). In order to obtain the linked sugars, it is necessary to effectively break the blocked polysaccharides of the recalcitrant lignocellulose (Bhalla et al. 2015), due that the efficient conversion of lignocellulosic biomass to fermentable sugars is an essential step for its subsequent conversion into value-added products such as bioethanol (Balat 2011, Wanmolee et al. 2014). In that sense, enzymes such as cellulases and xylanases have been investigated (Blumer-Schuette et al. 2008); especially those that are produced by thermophilic bacteria present in hot environments - such as thermal sources- because they can be used to develop more efficient and cost-effective processes to conversion of lignocellulosic biomass (Bhalla et al. 2013). These enzymes are characterized by their robustness, efficiency and conservation of their activity at high temperatures for long periods, which allow them to degrade carbohydrates under harsh bioprocessing conditions that reflect their natural biotopes (Blumer-Schuette et al. 2014). In this context, the objective of this research was to explore the hot spring Chancos from Carhuaz (Perú) for the isolation and selection of thermotolerant native bacteria with cellulolytic and xylanolytic activities which allow them to degrade lignocellulose from agriculture wastes of the region of Ancash, Peru.
Material and methods
Sampling and isolation.- Sampling and isolation were performed as described by Tamariz-Angeles et al. (2014). The water samples were collected in a secondary and rustic pool (9°19’09.39”S, 77°34’24.59”W) from Chancos hot spring, province of Carhuaz, Ancash, Peru. The temperature and pH during the sampling were 47.3 ± 3.2 °C, and 6.5 ± 0.2, respectively.
For isolation was performed three types of samples: fresh water for direct isolation, ex situ enrichment culture in the laboratory and in situ baiting-left for 14 days in the pool. Sugar cane bagasse, filter paper, microgranular cellulose or beechwood xylan were used for in situ and ex situ enrichment. All samples were diluted, and each dilution was platting by duplicate on basal medium salt (BMS1) supplemented with glucose (1% w/v) and Agar-agar (1.5% w/v). Incubation was at 50 °C by 15 days. Colonies with different morphology were isolated and purified. These strains were kept in the slanted tube with Trypticase Soy Agar (TSA) at 4 °C.
Selection of cellulolytic and xylanolytic strains.- Each strain isolated was cultivated in 5 mL of Trypticase Soy Broth (TSB). Fresh culture (5µL) was inoculated on Petri dishes containing BMS1 supplemented with Carboxymethyl cellulose (1% w/v) or beechwood xylan (1% w/v) and incubated at 50°C during 5 days (Tamariz-Angeles et al. 2014) by duplicate. Qualitative cellulolytic and xylanolytic activity was tested using Congo Red methodology (Sirisena & Manamendra 1995). The clear zone around the colony was measured as hydrolysis activities.
Microscopy analysis and biochemical test.- Strains of bacteria were grown in 5 mL of TSB (Trypticase Soy Broth) by 16 hours, to follow according to the Gram stain protocol. Stained samples were observed under the immersion objective of light microscope (Primo star, Zeiss).
Biochemical characterization of strains included the following tests: catalase production, urea metabolism, acid production using glucose (methyl red test), citrate metabolism, and amylase activity following universal protocols.
Taxonomic identification by 16S rDNA.- DNA of selected strains was extracted with AxyPrep Bacterial Genomic Miniprep Kit (Axygen) following the manufacturer protocol. Primers used to amplify were 27F/1492R (Reinsenbach & Longnecker 2000). PCR was performed as described by (Tamariz-Angeles et al. 2014) and amplification products were evaluated with agarose gel electrophoresis (1% w/v) with molecular marker GeneRuler 1 kb DNA (Thermo Scientific). PCR products were sent for sequencing to Macrogen Korea Inc. Primers used in sequencing reactions were 518F and 800R. Contigs of edited and assembled sequences were compared with similar sequences of NCBI GenBank (The National Center for Biotechnology Information U. S. http://www.ncbi.nlm.nih.gov/) using BLASTN. They were alignment with CLUSTALX v.2.0 and analyzed with MEGA5 using Neighbor-Joining methodology and Kimura 2-parameter with 1000 Bootstraps to make the taxonomic identification.
Optimum growth temperature and metabolic characterization.- Trypticase Soy Broth (5 mL) was inoculated with 250 µL of fresh culture (8 hours) diluted to 0.1 OD620nm; and incubated in an orbital shaker at 180 rpm at 30, 35, 40, 45, 50, 55 or 60 °C. Two replicates of each treatment were prepared. The growth was measured with a UV-VIS spectrophotometer (Spectroquant® Pharo 300, Merck) at 620 nm each 2.5 hours up to 12.5 hours. For each temperature evaluated a growth curve was obtained to calculate the growth rate during the exponential phase. A correlation between growth rate and temperature were obtained(Madigan et al. 1998) to find the optimum temperature.
Quantification of enzymatic activity.- Crude extracts were used for the determination of endoglucanase activity (or CMCase), total cellulase (FPA) and xylanases.
To prepare enzymatic crude extracts, 25 mL of Luria broth (LB) supplemented with CMC or beechwood xylan (1%) was inoculated with 5% (v/v) of fresh culture (8h) diluted to 0.1 OD620nm, and was incubated for 20 hours at 50ºC and 180 rpm. The crude extract was obtained by centrifugation at 10.595 g, and filtration with bacteriological membrane (0.22 µm sterile membrane, Millipore, Merck). All samples were prepared with three replicates.
Enzymatic activities were quantified with the micro-well 3,5-dinitrosalicylic acid (DNS) method proposed by King et al. (2009) and modified by Tamariz-angeles et al. (2014). Total cellulase activity and endoglucanase were calculated from a standard curve with anhydrous glucose and with anhydrous xylose for xylanase activity. These curves were prepared at 7, 8, 9 and 10 min. of reaction time to get a better DNS colorimetric reaction at 91.5 °C, which is the boiling temperature of water in the laboratory condition (3160 m of altitude). Enzymatic activity was expressed in enzyme unit (U), which is the amount of enzyme necessary to release 1μmol of product (reducing sugar) per minute under determined conditions (temperature, pH).
Optimum pH and temperature, and thermal stability.- The crude extract with the highest enzymatic activity was selected to evaluate temperature and optimum pH, and its thermal stability following the methodology described by Tamariz-Angeles et al. (2014). For thermal stability, the crude extract was incubated at 50, 60, 70, and 80 °C during 1 hour previously to evaluate the residual enzymatic activity.
Thermal-alkaline pretreatment of agriculture waste.- Agricultural waste (300 g) washed with tap water, was dried at room temperature for 48 hours. For its complete drying, it was placed in an oven at 65 °C for approximately 4 hours to obtain a constant weight. For the chemical-thermal pretreatment, 100 g of sample was mixed with 1 L of NaOH (24 g.L-1) and autoclaved at 121 °C, 1 atm, 20 minutes.
Next, it was washed with tap water until pH 7 was obtained, and it was allowed to dry at room temperature until constant weight. It was ground and sieved with AST meshes at 2 mm diameter for submerged fermentation, and 150 - 75 μm diameter for saccharification using crude enzymatic extract.
Agriculture waste degradation and enzymatic activity.- The strain with the high enzymatic activity was evaluated in its ability to degrade pretreated substrates (sugar cane bagasse, quinoa stalk and wheat straw) It included: (i) degradation/saccharification during submerged fermentation, (ii) enzyme production with agriculture waste as carbon source, and (iii) saccharification of agriculture waste using the crude enzyme extract. For (i) and (ii), it was used substrates sieved at 2 mm, and for (iii) substrates sieved at 75 μm of diameter.
Submerged fermentation was performed using a basal salt medium (BMS2) with the following composition (g·L-1): NaCl (5), MgSO4.7H2O (0.1), CaCl2.2H2O (0.6), KH2PO4 (0.1), FeCl3.6H2O (0.01), nitrogen source (5) (peptone or ammonium nitrate), carbon source (0.5 of agriculture waste, or 0.25 of CMC or 0.25 beechwood xylan), and glycerol (0.5% v/v). The pH was adjusted to pH 6.5.
For saccharification during submerged fermentation (i), Flasks containing 50 mL of BMS2 were inoculated with 2.5 mL fresh culture of 8 hours (0.08 - 0.1 OD620nm) and incubated at the optimum growth temperature of selected strain at 180 rpm. Cultures were performed by duplicate for each strain.
Cultures of 72, 144 or 216 hours were used to evaluate the degradation or saccharification ability during the submerged fermentation. Reducing sugars release in the submerged fermentations were measure by DNS method described previously. Results were expressed as reducing sugar release per 1 mL of extract, and it was used the glucose standard curve. Moreover, xylan and CMC were considering as standard substrates, their values of reducing sugar release were taken as 100% to compare them with values obtained for agriculture wastes.
To evaluate the enzyme production using agriculture waste as carbon source (ii), BMS2 supplemented with agriculture waste (2% w/v) and the better nitrogen source found in the previous assay was used. Culture conditions were the same as above. Samples of 2 mL were collected by duplicate each 4 hours until 28 hours. Endoglucanase and xylanase activity were quantified.
For saccharification assay using the diluted crude enzyme (iii), two enzymatic extracts were selected according to their enzymatic titers. 500 mL of crude enzymatic extract was concentrated and partially purified using centrifugal filters Ultra-15 3K (Amicon®, Merck Millipore, Ireland). Endoglucanase and xylanase activities of the concentrated extract were quantified and dilute with phosphate buffer (50 mM pH 6) to obtain 120 U/L-1 of endoglucanase activity (DE). Immediately, 35 mL of diluted DE was mixed with 1.4 g of substrate (agriculture waste, CMC or beechwood xylan) and incubated in agitation at 180 rpm and optimal temperature for 12, 24, 36, 48, 60 and 72 hours. Negative control without the crude enzyme extracts were prepared. Reducing sugar release (RSR) was quantified using the DNS method Results were expressed as RSR (mg) per gram of substrate (g).
Amplification of endoglucanase and xylanase genes.- DNA was used for the amplification of endoglucanase and xylanase genes. Primers CelF/CelR were used for endoglucanases of Bacillus (Nurachman et al. 2010), CbgF/CbglR for endoglucanase bgl C of B. licheniformis (Aftab et al. 2012), xynAoli1/xynAoli2 for xylanase xynA gene of B. subtilis (Wolf et al. 1995), and XtF/XtR for cellulase free alkaline xylanase gene of Bacillus sp. (Kumar et al. 2011). The master mix of 50 µL was prepared in 1X Dream Taq reaction buffer with 0.2 mM dNTP, 0.1 mM of each primer, 0.5 U Dream Taq polymerase (Thermo Scientific), and 5 ng of template DNA. PCR protocol for CelF/CelR, xynAoli1/xynAoli2, and XtF/XtR includes 2 min of initial denaturation step at 94 °C, followed by 30 cycles of 1 min at 94 °C for denaturation, 1 min at 53 °C for annealing, and 2 min at 72 °C for extension; and 8 min at 72 °C for final extension. PCR protocol for CbglF/CbglR consists of 5 min of initial denaturation at 94 °C, followed by 30 cycles of 30 s at 94 °C for denaturation, 30s at 54 °C for annealing, and 90s at 72 °C for extension; and 10 min at 72 °C for final extension.
PCR products were evaluated by agarose gel electrophoresis (1% w/v), and sequenced in Macrogen Korea Inc. Both, consensus DNA sequence and translated amino acid residues chain were prepared with Codoncode aligner v.8.0.2. The consensus DNA sequences were analyzed with BlastN; adding similar sequences of Genbank, sequences were aligned with ClustralX v.2.0, and the phylogenetic tree was prepared with MEGA7 using Neighbor-Joining, Kimura 2-parameter, and 1000 Bootstraps methodology. The translated amino acid residues chains were analyzing in Protein Blast and Protein SmartBlast online platform of NCBI (http://www.ncbi.nlm.nih.gov/). According to Yang et al. (2009), the model of protein structure was predictive using Swiss-Model Server online [http://swissmodel. (https://swissmodel.expasy.org).
Statistical analyses.- All qualitative and quantitative data were prepared with four or six replicates, means and standard deviation were calculated. ANOVA to Linear Models, T-student, and Duncan’s analyses were used to evaluate significant differences between treatments (p < 0.05 or p < 0.01).
Results and discussion
Isolation and selection.- Water samples from Chancos hot springs were used for isolation cellulase producing bacteria. From 31 bacterial strains isolated from three different types of samples, 14 strains were screened and selected as endoglucanase and xylanase producers (Figure 1). Direct culture from freshwater samples and in situ enrichment bait of sugar cane bagasse yield more isolated and selected strains (Figure 1A). Tamariz-Angeles et al. (2014) found similar results for in situ enrichment with sugar cane bagasse, but they did not isolate any strains from direct isolation from Huancarhuaz hot spring water samples (near 70°C). Sugar cane bagasse is known as a suitable substrate for production of cellulases and xylanases (Adsul et al. 2004, Camassola & Dillon 2007, Ajijolakewu et al. 2013, Saha 2003).
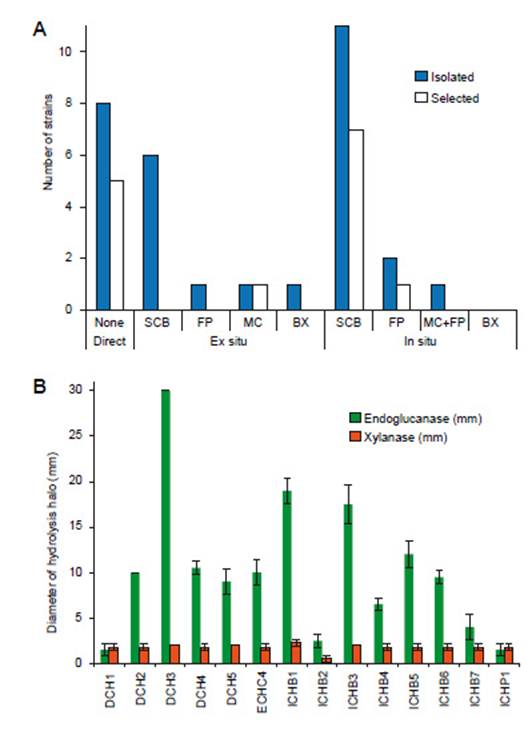
Figure 1: Isolation and selection of thermotolerant bacterial strains of Chancos hot spring. A. Isolated and selected bacterial strains (SCB, sugar cane bagasse; FP, filter paper; MC, microgranular cellulose; BX, birchwood xylan); B. Diameter of the clear zone around selected strains. Values represent the mean of four replicates ± SD.
Molecular identification and biochemical characterization test.- Edited ribosomal 16S sequences were assembled until 1537 bp size corresponding to the complete gen. From BlastN analyses, 99.14 - 99.87% range of identity with sequences of type collection of Genbank was found. According to the phylogenetic tree (data not showed), 12 strains were identified as B. licheniformis and only two strains were B. subtilis (Table. 1). DNA sequences were submitted to Genbank to get the accession numbers MK524697 - MK564710 (Table. 1). In addition, microscopic analysis showed that all selected strains were rod shape and spore-forming gram-positive bacteria. Similar studies found that some species of Bacillus are natural inhabitants of hot springs around the word (Acharya & Chaudhary 2011, Tamariz-Angeles et al. 2014) with useful applications in industry (Mehta & Tulasi 2013).
Table 1 Molecular and biochemical test characterization of selected strains from Chancos hot spring.
| Strain | 16S DNAr identification | GenBank Accession number 16S DNAr | Growth temperature (C°) | Metabolic characteristics | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Specie | Identity (%)* | Optimum | Interval | Citrate | Urea | Catalase | RM | Amylase | ||||
| ICHB1 | B. subtilis | 99.33 | MK564697 | 45 | 30-55 | + | + | + | + | + | ||
| ICHB2 | B. licheniformis | 99.8 | MK564698 | 50 | 30-55 | + | + | - | - | - | ||
| ICHB3 | B. licheniformis | 99.46 | MK564699 | 50 | 30-55 | - | + | + | - | - | ||
| ICHB4 | B. licheniformis | 99.53 | MK564700 | 50 | 30-55 | - | + | + | - | - | ||
| ICHB5 | B. licheniformis | 99.87 | MK564701 | 50 | 30-55 | - | + | + | - | - | ||
| ICHB6 | B. licheniformis | 99.66 | MK564702 | 50 | 30-55 | - | + | + | - | - | ||
| ICHB7 | B. licheniformis | 99.06 | MK564703 | 50 | 30-55 | - | + | + | - | - | ||
| ICHP1 | B. licheniformis | 99.87 | MK564704 | 50 | 30-55 | - | + | - | - | - | ||
| DCH1 | B. licheniformis | 99.87 | MK564705 | 50 | 30-55 | - | + | + | + | - | ||
| DCH2 | B. licheniformis | 99.66 | MK564706 | 50 | 30-55 | - | + | + | - | + | ||
| DCH3 | B. licheniformis | 99.87 | MK564707 | 50 | 30-55 | - | + | + | - | - | ||
| DCH4 | B. subtilis | 99.19 | MK564708 | 45 | 30-50 | + | + | + | + | + | ||
| DCH5 | B. licheniformis | 99.53 | MK564709 | 45 | 30-55 | - | + | + | - | - | ||
| ECHC4 | B. licheniformis | 99.14 | MK564710 | 45 | 30-50 | - | + | - | - | - | ||
*, it shows the highest percentage obtained using BlastN platform and type culture collection of Genbank (www.ncbi.nlm.nih.gov). RM, red methyl test.
The optimum growth temperatures found of selected strains (Table 1) suggest that most of B. licheniformis strains are moderate or facultative thermophiles according to the classification proposed by Mehta and Tulasi (2013). Likewise, B. subtilis strains exhibited optimum growth temperature at 45 °C, showing their thermotolerant condition. A similar result has been reported for a strain of B. subtilis isolated from YangLing hot springs in China (Li et al. 2008). According to biochemical tests performed, all strains were urease producers and most of them were catalase positive. A reduced number of strains were citrate positive, and acid producers. Finally, only two strains showed amylolytic activity.
Determination of enzymatic activity.- Both endoglucanase and xylanase activities of selected strains were tested using DNS method (Figure 2A-B). From 31 isolates, only six strains showed endoglucanase activity, and 14 selected strains displayed xylanase activity. It was observed that endoglucanase activity was significantly induced by xylan (Fig 2A) while xylanase activity promoted by CMC was higher with xylan in most of the strains (Fig 2B). According to Jonnadula et al. (2018), xylan or intermediates produced during xylan degradation might be involved in cross-induction of endoglucanase, and some of the heterologous oligosaccharides structurally related may cause cross-induction of unrelated carbohydrolases.
In submerged fermentation, enzymatic production could be influenced by nutritional (especially carbon and nitrogen sources) and environmental culture factors (Acharya & Chaudhary 2011). In some species of Bacillus cultured with organic source of nitrogen increased the production of xylanases, while (NH4)2SO4 could inhibit secretion and synthesis of xylanases (Sepahy et al. 2011). According to the quantitative method, B. subtilis DCH4 showed better results for both endoglucanase and xylanase activities with tryptone and yeast extract as a nitrogen source. Additionally, the extract LB-CMC of B. subtilis DCH4 showed the best total cellulase activity reaching 108 ±14 UL-1. This strain was selected for agriculture waste degradation assays.
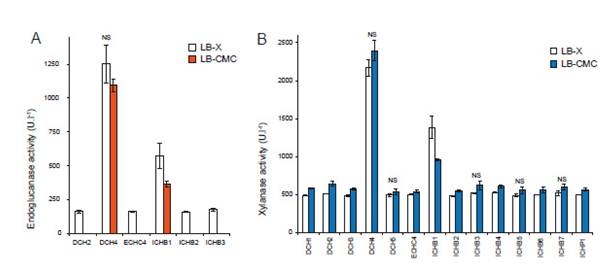
Figure 2: Hydrolytic activities of selected strains. A. Endoglucanase activity of crude extract; and B. Xylanase activity of the crude extract. LB-X, Luria broth-xylan; LB-CMC, Luria broth-Carboxymethyl cellulose; NS, none significant t-student with 99% of confidence. Values represent the mean of six replicates ± SD.
Optimum pH and temperature; and thermal stability.- According to quantitative enzymatic assay, the strain DCH4 was selected to evaluated hydrolytic activities using agriculture waste. Due that enzymes require optimum environmental conditions such as temperature and pH (Anwar et al. 2014), these parameters were determined for endoglucanase and xylanase of B. subtilis DCH4 as well as thermal stability (Fig. 3). Both, temperature fluctuation and pH changes can affect the integrity of the secondary, tertiary and quaternary structure of the enzyme protein, denaturing and inactivating it, thus affecting enzymatic activity (Chen et al. 2016). According to the results, the optimal temperatures for both enzymes were around 55 oC while optimal pH was in the range 5-6 (Fig. 3A and 3B). But endoglucanase activity of DCH4 showed more thermal stability (Fig. 3C).
Specifically, endoglucanase activity of crude extract of B. subtilis DCH4 showed optimum endoglucanase activity between 45 - 55°C and retaining more than 70% of activity when the hydrolysis was performed at 70°C (Fig. 3A). This feature shows that DCH4 has endoglucanase activity useful for some industrial bioprocess, especially in lignocellulose conversions that has several limitations when being carried out at ≤50 °C (Bhalla et al., 2013). Li et al. (2008) purified endoglucanase of family 5 (CelDR) from B. subtilis DR isolated from a hot spring, which showed 40°C optimum temperature and retained more than 80% of its maximum activity at temperatures between 45°C and 70°C. Another condition to get optimum enzymatic activity is the pH. Adaptability of thermophilic microbes and thermostable enzymes to a wide range of pH also makes them suitable candidates bioprocessing (Bhalla et al. 2013). Hence, DCH4 showed optimum endoglucanase activity at pH 5, but retained more than 52% activity between 5 and 7.8 of pH (Fig. 3B) DCH4 and certain stability at pH range 4 - 7. For lignocellulose bioconversion, acidophilic and alkalophilic thermotolerant enzymes have several advantages because could avoid the neutralization step after acid or alkali pre-treatment of biomass (Bhalla et al. 2013).
In the other hand, thermal stability is the capacity of an enzyme to retain its active structural conformation at a selected high temperature for a prolonged period (Bhalla et al. 2013). According to figure 3C, crude enzymatic extract of DCH4 retained 37 %, 25, 25% and 25% of endoglucanase activity after 1 h of incubation to 60, 70 and 80°C respectively. However, CelDR −cataloged with high thermal stability retained 70% of its cellulase activity after incubation at 75°C for only 30 min. (Li et al. 2008). Our results suggest that endoglucanase DCH4 could be exhibited moderate thermal stability.
About xylanase activity, thermostable xylanases which can tolerate extreme conditions pH, and high salt concentrations are of great interest for bioprocessing (Bhalla et al. 2013). The optimum xylanase activity was 55°C, retaining 87% and 74% of its activity at 50°C and 60 °C, respectively (Fig. 3A). The optimum pH was 6, retaining 51, 72 and 40 % of activity at pH of 5, 7and 8 respectively (Fig. 3B). But the crude extract of DCH4 lost its xylanase activity after 1 hour of incubation at 60°C. Similar results were described for xylanase of a mesophyll B. subtilis R5 (Jalal et al., 2009). However, thermostable xylanases from thermo-alkaliphilic, thermo-acidophilic, and thermo-halophilic bacteria have been reported (Bhalla et al. 2013).
Agriculture waste degradation and enzymatic activity.- Biochemical conversion of lignocellulosic substrates through saccharification and fermentation is a major pathway for bioethanol production (Balat 2011). Then, three lignocellulosic agriculture wastes (sugarcane bagasse, wheat straw, quinoa stalks) were selected to evaluate the degradation ability of B. subtilis DCH4. Then, saccharification and enzyme production assays were performed to evaluate the potential of B. subtilis DCH4.
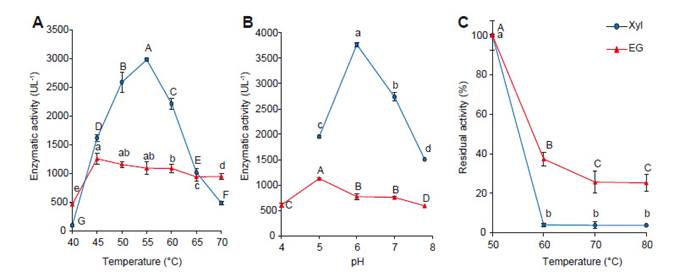
Figure 3 Temperature and pH condition for enzymatic activity of crude extract of B. subtilis DCH4. A. optimum temperature, B. optimum pH, and C. thermal stability (Residual activity after incubation for 1 hour at 50, 60, 70°C). Values represent mean of six replicates ± SD, and means with different letters show significance difference (p<0.05).
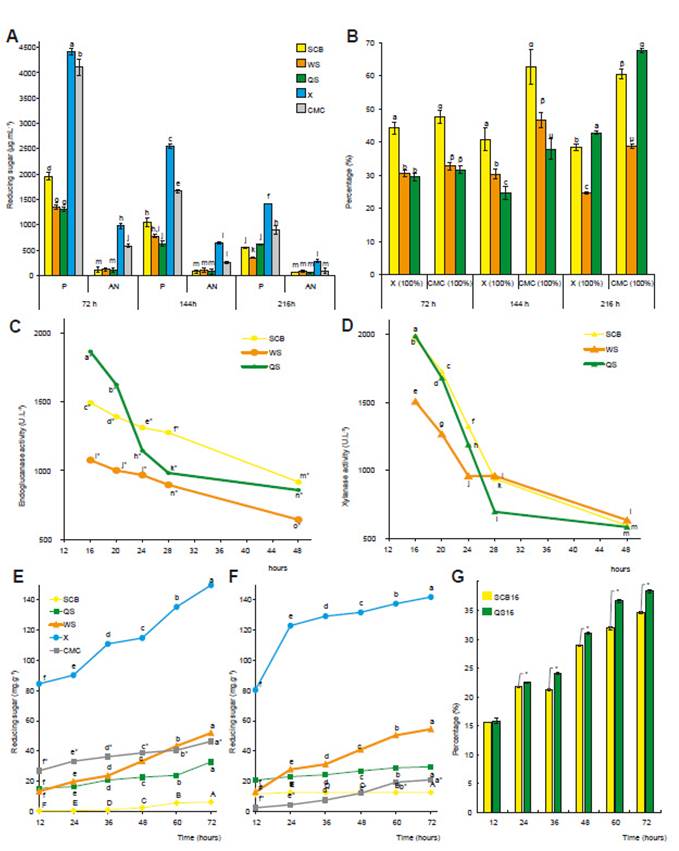
Figure 4: Agriculture waste degradation and enzymatic activity of a selected strain. Submerged fermentation: A. Reducing sugars release; B. Percentage of reducing sugars released from agriculture (considering 100% RSR of CMC or beechwood xylan). Enzyme production by submerged fermentation in medium with agriculture wastes: C. Endoglunacase activity; D. Xylanase activity. Saccharification using diluted crude enzyme: E. Reducing sugar released using crude enzyme of sugar cane bagasse fermentation; F. Reducing sugar released using crude enzyme of quinoa stalk fermentation; and G. Percentage of saccharification of wheat straw related to xylan (100%). P, peptone; AN, ammonium nitrate; SCB, sugar cane bagasse; QS, quinoa stalks; WS, wheat straw; X, xylan of beechwood; CMC, carboxymethyl cellulose. Values represent mean of six replicates ± SD, and means with different letters show significance difference (p<0.01). *, significance difference (p<0.01)
First, saccharification during submerged cultures was measure as reducing sugars release of different lignocelluloses substrates (sugarcane bagasse, wheat straw, quinoa stalks, CMC and xylan). It was found that B. subtilis DCH4 hydrolyzed all carbon sources substrates assayed (Fig.4A). Due that xylan and CMC are common substrates for cellulase and xylanase assays (Acharya & Chaudhary 2011, Lynd et al. 2002, Sharma & Kumar 2013) and are less recalcitrant than agriculture wastes, they have been used as a reference substrate to evaluate the level of degradation ability calculating an index expressed yield of reducing sugar released as percentage (Fig. 4B). It was found that sugar cane bagasse released 44% of reducing sugar compared with xylan during the first 72 hours, and the other substrates attained percentages at around to 30%. These results showed that B. subtilis DCH4 not only have the ability to hydrolyze commercial substrates, furthermore, it can degrade natural pre-treated substrates such as sugar cane bagasse, quinoa stalks, and wheat straw. Additionally, the influence of nitrogen source in the saccharification yield was evaluated using organic and inorganic source. Peptone allows the higher yield of reducing sugars while ammonium nitrate produced the lower release of reducing sugars. Nitrogen source used in the production medium is one of the major factors affecting the level of enzyme production (Kumar et al. 2016). Organic nitrogen influences the formation of extracellular enzymes as a precursor for protein synthesis (Bajaj et al. 2011). Furthermore, peptone has been reported as a good supplement for microbial growth and xylanase production (Raj et al. 2013, Shanthi & Roymon 2018). The largest amount of reducing sugars released occurred at 72 hours and decreased after that time probably by the depletion of the macro and micronutrient in the growth medium (Bibi et al. 2014). Moreover, hydrolysis of carbon substrates could provide an easily accessible source of simple sugars for bacterial growth, which decreased in the culture medium in the resting 144 and 216 hours. Bibi et al. (2014), suggest that high yield of the final product (xylose) produced after the degradation of xylan during fermentation could be responsible of enzyme inhibition due to feedback regulation.
The use of inexpensive agriculture residues as substrates for the production of industrial enzymes is a significant way to reduce the cost of the overall process and literature shows that several microbial species have been reported to use lignocellulose for xylanase production (Bhalla et al. 2015). Related with that, it was evaluated the production of hydrolytic enzymes (cellulases and xylanases) using a culture medium supplemented with agricultural waste as the sole carbon source, finding that sugar cane bagasse, wheat straw and quinoa stalk induced the production of endoglucanase and xylanase (Fig. 4C, 4D). The highest production in all cases was attained at 16 hours of culture, and quinoa stalk produces the highest endoglucanase and xylanase activities (1865.67 U·L-1 and 1986.43 U·L-1, respectively) and sugar cane bagasse reached similar xylanase activity (1963.82 U·L-1). Enzyme production is dependent on the nature of carbon source, favorable degradability, chemical composition, physical associations, accessibility of substrate, and presence of some nutrients (Kumar et al. 2016). Thus, when microorganisms degrade different polysaccharides, they released small, medium or large sized oligosaccharides that enter the cell and induce the expression of enzymes responsible for respective polysaccharide degradation (Jonnadula et al. 2018). In consequence, the chemical composition of each type of agriculture waste evaluated may influence on the induction and production of hydrolytic enzymes; but others micro and macronutrients could be necessary to evaluate for gain better cellulase and xylanase production.
In addition, enzymatic crude extracts of quinoa stalks and sugar cane bagasse cultures by 16 hours were used to saccharification of SCB, QS, and WS. Beechwood xylan and CMC was used as standard substrates. After 72 hours of saccharification, low yield of reducing sugar were obtained for all substrates including standard ones (Fig. 4E, 4F). Wheat straw yields 35 and 38% of reducing sugars with quinoa or bagasse extract, respectively (Fig. 4G). Miyazaki et al. (2005) found that during enzymatic hydrolysis, wheat straw releases more sugars than other lignocellulosic substrates probably by its higher content of cellulose and xylan. Enzymatic hydrolysis is the critical step for bioethanol production because various factors influence yields of monomer sugars from lignocellulose such as temperature, pH, enzyme doses, substrate concentration (Sarkar et al. 2012).
Endoglucanase and xylanase genes.- For improving enzymatic lignocellulose hydrolysis it is necessary to know which enzymes are involved in the bioprocess. Some researches of hydrolytic B. subtilis have been focused on fermentation optimization to obtain higher yields, and increase recovery rates employing genetic engineering (Sarkar et al. 2012). In this sense, there are some reports of characterization of complete genomes, genes and proteins and cloning to get more efficient enzymes (Yang et al. 2009, Nurachman et al. 2010, Vallenet et al., 2017, Chang et al. 2018).
Then, some genes involved in the enzymatic activity of DCH4 were explored. Four sets of primers proposed for Bacillus genus were used but only two of them allowed the amplification at performed PCR conditions. Primer set that amplified endoglucanase gene was CelF/CelR, which was designed using conserved sequences from GenBank of endoglucanase of genus Bacillus (Nurachman et al. 2010). Primer set that amplified xylanase gene was xynAoli1/xynAoli2 designed for xynA gene from Bacillus subtilis (Wolf et al. 1995). CbgF/CbglR and XtF/XtR primer sets did not produce any amplification products.
The DNA sequence of the cellulase gene was edited to 1401 pb, deposited to Genbank as AN MK644599, and compared with GenBank data using BlastN. It was found 99.57% of identity with CDS of completed genome of B. subtilis (AN: CP0237555, CP011534), 98.86% with cellulase gene of B. subtilis (EF070194, JQ346089), 98.79 and 98.72% with endo-beta-1,4-glucanase gene of B. subtilis (MH923517, KC477685, respectively), and 98.50% with endo-beta-1,4-glucanase gene of B. amyloliquefaciens strain IARI-SP-2 (KF240848). Phylogenetic tree obtained is shown in figure 5A. The translated chain had 463 amino acid residues and was compared with other proteins using Standard Blast Protein. It was found that 35 - 281 fragment showed high identity with the cellulase - glycosyl hydrolase domain family 5 (AN pfam0150); while the interval of 341 to 422 has a high identity with cellulose binding domain family 3 (CBM-3) (AN pfam0042). This information was confirmed with protein SmartBlast analysis (Fig 5B.), where DCH4 cellulase chain earned 98% of identity with an endo-beta-1,4-glucanase of B. subtilis subsp. subtilis 168 (NP_389695). According to protein structural analysis, translated chain contains two monomers, a catalytic domain which has 99.34% identity with endo-1,4-beta-glucanase with manganese (II) ion from Bacillus subtilis 168, and other non-catalytic domain with 98.45% of identity to CBM3 lacking the calcium-binding site from Bacillus subtilis 168. Both structures were used for the model of cellulase of DCH4 (Fig. 5C-E). The endoglucanase of DCH4 model comprised 14-326 residual amino acids, it includes a ligand for manganese (Mn+2) shaped by four residues (142, 180, 182, 183) where 180 and 182 residues are metal complexes. The cellulose binding CBM3 model used 339-467 residues. Bhalla et al. (2013) refer that endoglucanases containing carbohydrate-binding modules (CBM) − also called primary cellulases − are the most crucial in enabling efficient utilization of crystalline cellulose. This type of cellulase is bifunctional, where both two domains interact together in cellulose recognition increasing the enzyme efficiency. In this case, DCH4 endoglucanase is supposed to have a ligand to Mn+2. Studies of endoglucanase of B. subtilis 168 (BsCel5A) found that Mn+2 improved drastically the thermal stability and lifetime at high temperature, did not change of substrate preference affinity (Santos et al. 2012). In the same way, studies of the role of CMB3 of Bacillus sp. KD1014 suggest that it contribute in thermostability, and affinity and substrate specificity for small substrates (Lee et al. 2018).
Endo-β-1,4-glucanases are the major enzymes responsible for the breakdown of internal glycosidic bonds of cellulose chains producing oligosaccharides, cellobiose, and glucose (Bhalla et al. 2013, Santos et al. 2012).
Xylanase consense sequence was edited to 651 pb and deposited in Genbank with MK644600 accession number. From BlastN analysis, a 99.69 % of identity with CDS sequences of complete genome B. subtilis (AN CP007173), 99.07% with xylanase gene of B. subtilis (Z34519, KJ540228), 99.38% endo-1,4-beta-xylanase of B. subtilis MW10 (DQ100307), and 98.92% with xlnA gene of xylanase EC 3.2.1.8 from B. circulans strain (XO7723) were obtained. Figure 6A shows the corresponding phylogenetic tree. Its translated chain containing 217 amino acids was trimmed to 214 and compared with other protein chains of NCBI database using Standard Blast Protein tool. It was found 100% of identity with a secreted endo-1,4-beta-xylanase of B. subtilis 168 (NP389765) and 99.53% with an endo-1,4-beta-xylanase of B. pumilus (AAZ17390). Concordantly, Protein SmartBlast analysis found 100% of identity with a multispecies endo-1,4-beta-xylanase A for Bacillales (WP003231377), and endo-1,4-beta-xylanase of B. subtilis subsp. subtilis str. 168 (Fig. 6B). Structural protein analysis showed that the protein is a monomer similar to endo-1,4-beta-xylanase from Bacillus subtilis 168 (1A1) (98.36%), which was used as a template to predict a structural model for xylanase DCH4 (Fig. 6C) with 30-212 residues. This model included a ligand to meso-tartaric acid shaped by six residues (131, 132, 133, 161, 162, 163) where 133, 162 and 163 residues form hydrogen bonds, and 161 form salt bridges.
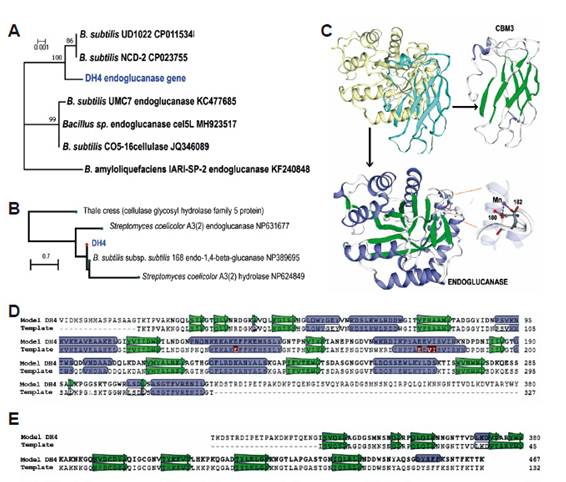
Figure 5 Analysis of endoglucanase gene of B. subtilis DCH4. A. Phylogenetic tree of endoglucanase gene using Neighbor-Joinning, Kimura 2-parameter, and 1000 bootstraps; B. Protein SmartBlast of the translated endoglucanase gene (NCBI, Standard protocol); C. Structural model of endoglucanase with cellulose binding molecule (CMB3); D. Alignment of DCH4 translated chain with endoglucanase of B. subtilis 168; and E. Alignment of DCH4 translated chain with CMB3 of B. subtilis 168. C, D, E were prepared with (https://swissmodel.expasy.org).
Enzymatic hydrolysis of xylan involves a multi-enzyme system where the endoxylanase attack the main chain of xylan (Balat 2011). Xylanase of DCH4 showed adequate optimum temperature (55 °C), but after incubation at 60 °C by 1 hour, its activity has lost completely. Protein structure studies of endoxylanase rXynA from Bacillus subtilis 168 (1A1) ‒ similar to xylanase DCH4 ‒ revels that the transition from the native to the denatured enzyme starts at a temperature of 55 ° C and for higher temperatures, both the catalytic activity and β-sheet secondary structure were lost (Murakami et al. 2005). Then, some studies to improve thermal stability have been performed by other researchers (Miyazaki et al. 2006).
Finally, it was found the presence of interesting results obtained for isolation of cellulolytic and xylanolytic microorganisms from Chancos hot spring, indicating that the strain DCH4, which showed the better performance is a B. subtilis thermotolerant strain. Its enzymatic activities could be related with bifunctional thermostable endoglucanase of family 5, and an endoxylanase of family 11 identified by molecular analysis from genomic DNA. This results, suggest that the strain B. subtilis DCH4 is a suitable candidate to perform biochemical and genetic studies in order to improve its enzymatic activity for biotechnological applications related to lignocellulose conversion process.
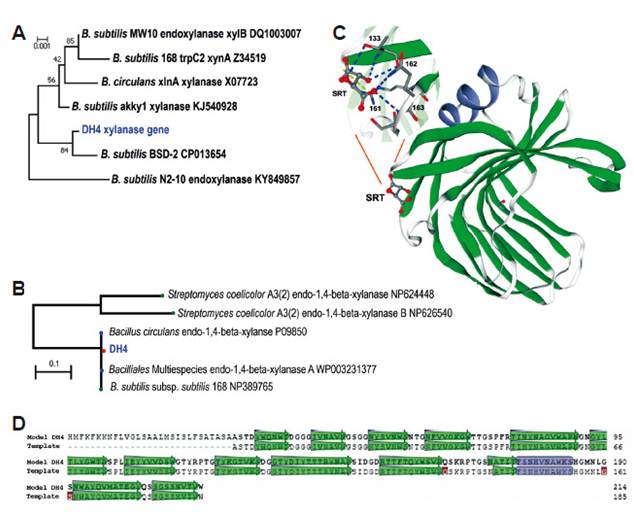
Figure 6 Analysis of xylanase gene of B. subtilis DCH4. A. Phylogenetic tree of xylanase gene using Neighbor-Joining, Kimura 2-parameter, and 1000 bootstraps; B. Protein SmartBlast of the translated xylanase gene (NCBI, Standard protocol); C. Model of xylanase of DCH4; D. Alignment of DCH4 translated gene with the closer template (xylanase of B. subtilis 168 (1A1). SRT, meso-tartaric acid. C and D were prepared with (https://swissmodel.expasy.org).











 uBio
uBio