Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de la Sociedad Química del Perú
versión impresa ISSN 1810-634X
Rev. Soc. Quím. Perú v.71 n.1 Lima ene./mar. 2005
Domino reactions with 5-azido- and 5-amino-4- trifluoromethyl-1,3-azoles.
Klaus Burger1; Eva Höß; Lothar Hennig and Lothar Beyer2
1 Department of Organic Chemistry, University of Leipzig, Johannisallee 29, D-04103 Leipzig, Germany
2 Department of Inorganic Chemistry, University of Leipzig, Johannisallee 29, D-04103 Leipzig, Germany
RESUMEN
Reacciones dominó con 5-azido- y 5-amino-4-trifluorometil-1,3 azoles.
Los compuestos con sus sub-estructuras como CF 3 -C=C-NH-R, son considerablemente reactivos frente a aminas primarias. Ellos se comportan ortho-fluoruros y son fácilmente susceptibles respecto a la eliminación / adición de restos. Éstos pueden ser enlazados uno con otro dando así reacciones tipo dominó.
Palabras clave: 5-Fluoro-4-trifluormetil-1,3-azoles; 5-azido-4-trifluormetil-1,3-azoles; 5-amino-4-trifluor-metil-1,3-azoles, reacciones tipo dominó; [1.4] eliminación HF; adición Michael.
ABSTRACT
Compounds with substructures like CF 3 -C=C-NH-R are remarkable reactive towards primary amines. They behave like ortho-fluorides and are readily susceptible to elimination / addition sequences which can be linked together and performed as domino reactions.
Keywords: 5-Fluoro-4-trifluoromethyl-1,3-azoles, 5-azido-4-trifluoromethyl-1,3-azoles, 5-amino-4-trifluoromethyl-1,3-azoles, domino reaction, [1.4] HF-elimination, Michael addition
INTRODUCTION
Domino reactions offer preparatively simple and elegant solutions of complex synthetic problems.1 The efficiency of this concept relies on linking together single reaction steps, thus avoiding separation and purification procedures after each step. When certain structural requirements are met, perfluoroalkyl substituted compounds readily undergo domino reactions.2
RESULTS AND DISCUSSION
Although the trifluoromethyl group is often considered to be chemically inert,3,4 it is known to undergo a variety of reactions. The hydrolytic behavior of a trifluoromethyl group is very much dependent on its position in a molecule.
Trifluoromethyl groups attached to carbon atoms possessing acidic hydrogen atoms are susceptible to hydrolysis in basic media.5 For this reason 3,3,3-trifluoroalanine and its derivatives are unstable above pH 8.5 even at room temperature. The trifluoromethyl group undergoes hydrolysis to give a carboxylate moiety.6 Likewise, trifluoromethyl groups attached to certain positions of heterocyclic systems undergo facile base-induced hydrolysis, e.g. 2-trifluoromethylimidazole.7
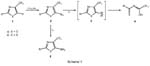
5-Amino-4-trifluoromethyl-1,3-azoles (5)8 are readily available from 5- fluoro-4-trifluoro-methyl-1,3-azoles (1)9 via 5-azido-4-trifluoromethyl-1,3-azoles (2).10 While compounds 2a decompose already at room temperature within 1-2 hours to give 4-cyano-4-trifluoromethyl-1-oxa-3-aza-1,3-butadienes (4) with eliminationofnitrogen,11 the corresponding 5-azido-4-trifluoromethyl-1,3-thiazoles (5)areconsiderable morestable.Therefore,5-amino-4-trifluoromethyl-1,3-oxazoles-(5a),5-azido-(2b)and5-amino-4-trifluoromethyl-1,3-thiazoles(5b)representuseful model compounds to study new types of domino reactions.
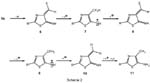
Undersurprisinglymild conditionsacompleteF/H-exchangeofatrifluoromethyl group can be achieved when compounds 2b and 5b are treated with LiAlH 4 in diethyl ether at room temperature. Starting from 5b, three cycles, each consisting of twosteps,namely [ 1.4 ] -eliminationfollowed byadditionofa hydrideion,arelinked together to give a six-step domino reaction (5 → 6 → 7 → 8 → 9 → 10 → 11).
Thisreactionsequencecan beappliedfora concisedecorationofheterocyclic systems bearing subunits like CF 3 CH=CHNH- with interesting substituent patterns.
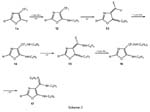
Forexample,5-fluoro-4-trifluoromethyl-1,3-oxazole(1a)was heatedwithanexcess of freshly distilled aniline (bath temperature: 40 o C) until 19 F NMR spectroscopy indicatescomplete consumptionoftheorganic fluorinecompound.Elementalanalysis and mass spectrometry indicate that the compound obtained after work-up and recrystallization from chloroform/hexanes (3:1) has a molecular weight of 430.51 which is in agreement with the formula C 28 H 22 N 4 O. Consequently, four equivalents of HF have been eliminated from the starting material 1a, while three equivalents of aniline have been added during the reaction sequence.
The first step of the domino reaction is a nucleophilic displacement reaction of the single fluorine bound to C-(5) (1 → 12). The subunit CF 3 C=C-NH-C 6 H 5 is capable for a 1.4-HF-elimination to give a highly reactive Michael system (13), since the consecutive addition is driven by rearomatization (13 → 14). The newly formedsubunitH 5 C 6 NH-CF 2 C=C-NH-C 6 H 5 againundergoesa1.4-HF-elimination (14 → 15) followed by a Michael addition (15 → 16). The final step of the sequence is a HF-elimination (16 → 17) to form an amidine moiety. When the core unit CF 3 C=C-NH-R is present twice in a molecule, it should be possible to run two domino reactions – with six steps each – parallel in a one-pot procedure.12
EXPERIMENTAL
General
Solvents were purified and dried prior to use. Reagents were used as purchased. Flash chromatography was performed using silica gel (32-63 ìm) with solvent systems given in the text. Melting points (uncorrected) were determined with a Tottoli apparatus (Fa. Büchi). 1 H (200 MHz, 360 MHz), 13 C (50 MHz, 75 MHz) and 19 F (188 MHz, 282 MHz) NMR spectra were recorded on Bruker WP 200, Bruker AM 360, Jeol C 60 HL and Jeol FX 90 Q spectrometers. TMS was used as reference for 1 H and 13 C NMR spectra (internal), and CF 3 COOH for 19 F NMR spectra (external). IR spectra were obtained on Perkin Elmer 157 G and 257 spectrometers. Mass spectra were recorded on a Varian MAT CH 5 spectrometer at 70 eV.
5-Amino-4-methyl-2-(4-methylphenyl)thiazole (11)
Method A: To a suspension of LiAlH 4 (0.76 g, 20 mmol) in dry ether (25 mL) at -50 o C a solution of 5-azido-2-(4-methylphenyl)-4-trifluoromethylthiazole (2b)10 (2.84 g, 10 mmol) in dry ether (25 mL) was added with stirring. The reaction mixture was slowly warmed up to room temperature and stirred for 16 h. Unreacted LiAlH 4 was destroyed on addition ice/water (20 mL). After filtration the organic phase was twice extracted with water (20 mL) and dried with MgSO 4 . After removal of the solvent in vacuo the residue was purified by column chromatography (eluent: chloroform).
Method B: To a suspension of LiAlH 4 (0.76 g, 20 mmol) in dry ether (10 mL) a solution of 5-amino-2-(4-methylphenyl)-4-trifluoromethylthiazole (5b)8 (2.58 g, 10 mmol) in dry ether (10 mL) was added at -50 o C with stirring. After stirring at -50 o C for 2 h the reaction mixture was slowly warmed up to room temperature and stirred for further 16 h. After careful treatment with ice/water (20 mL), the mixture was filtrated and the organic phase extracted twice with water (20 mL). The organic phase was dried with MgSO 4 , evaporated to dryness and the residue purified by column chromatography (eluent: chloroform).
Yield: 0.62 g, 30% (method A); 0.80 g 39% (method B); mp 113 o C; IR (KBr). δ = 3440, 3260, 3170, 2940, 1620, 1565, 1535, 1465 cm -1 ; 1 H NMR (acetone-d 6 ): ? = 2.32 (s, 3H), 2.38 (s, 3H), 7.25 (m, 2H), 7.82 (m, 2H); 13 C NMR (acetone-d 6 ): δ = 14.9, 21.3, 126.4, 130.4, 132.4, 140.3, 140.5, 144.7, 161.5; MS (70 eV) m/z = 204 [M] + , 171 [M – HS] + , 135 [C 8 H 7 S] + , 118 [C 8 H 8 N] + ; anal. cald for C 11 H 12 N 2 S (204.27): C 64.68, H 5.92, N 13.71; found: C 64.47, H 5.40, N 13.80.
5-[(N-Phenylamino)-2-phenyloxazol-4-yl]-N,N´-diphenyl-formamidine (17): 5-Fluoro-4-trifluoromethyl-1,3-oxazole (1a)9 (1.15 g, 5 mmol) was added to freshly distilled aniline (15 mL), then the stirred mixture was heated (bath temperature: 40 o C) for ca. 6 d. The progress of the reaction was monitored by 19 F NMR spectroscopy. When the conversion of the starting material was complete, the reaction mixture was partitioned between chloroform (25 mL) and water (25 mL). The organic phase was separated and twice treated with water (25 mL), dried with MgSO 4 and evaporated to dryness in vacuo. The remaining residue was recrystallized from chloroform/hexanes (3:1).
Yiel d: 1.05 g (49%); mp 154 – 156 o C; IR (KBr): δ = 1700, 1590, 1490, 1430 cm -1 ; 1 H NMR (CDCl 3 ): δ = 6.80 – 7.43 (m, 20H), 8.30 (s, br., 1H), 10.45 (s, 1H); 13 C NMR (CDCl 3 ): ? = 107.2, 121.6, 122.0, 124.2, 124.4, 127.4, 127.6, 127.9, 128.2, 128.4, 128.5, 129.1, 130.5, 135.6, 137.3, 137.5, 142.6, 149.9, 165.4; MS (70 eV) m/z = 430 [M] + , 337 [M – C 6 H 5 NH 2 ] + , 105 [C 6 H 5 CO] + , 93 [C 6 H 5 NH 2 ] + , 77 [C 6 H 5 ] + ; anal. calcd for C 28 H 22 N 4 O (430.51): C 78.11, H 5.15, N 13.00; found: C 77.64, H 5.75, N 13.41.
ACKNOWLEDGEMENTS
We thank Deutsche Forschungsgemeinschaft and Fonds der Chemischen Industrie for financial support.
Dedicated: A la «Revista de la Sociedad Química del Perú» por los 70 años de su publicación trimestral ininterrumpida.
REFERENCES
1. a) Tietze LF., Beifuss U. Angew. Chem.. 1993, 105, 137; b) Tietze LF. Chem. Rev. 1996, 96, 115. [ Links ]
2. a) Knunyants IL., Bargamova MD. Izv. Akad. Nauk SSSR, Ser. Khim. (Engl. Transl.) 1977, 1776; b) Bargamova MD., Motsishkite SM., Knunyants IL. Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.) 1990, 39, 2338. [ Links ]
3. Mc Bee ET., Pierce OR., Kilbourne HW. J. Am. Chem. Soc. 1953, 75, 4091. [ Links ]
4. Haszeldine RN. J. Chem. Soc. 1953, 922. [ Links ]
5. Kitazume T., Ohuogi T. Synthesis 1988, 614. [ Links ]
6. a) Weygand F., Steglich W., Lengyel I., Fraunberger F., Maierhofer H, Oettmeier W. Chem. Ber. 1966, 99, 1944; b) Burger K., Mütze K., Hollweck W., Koksch B., Kuhl P., Jakubke H-D., Riede J., Schier A. J. prakt. Chem. / Chem. Ztg.1993, 335, 321. [ Links ]
7. a) Kimoto H., Cohen LA. J. Org. Chem. 1979, 44, 2902; b) Kimoto H., Cohen LA. J. Org. Chem.1980, 45, 3831. [ Links ]
8. a) Burger K., Höß E., Chem. Ztg. 1989, 113, 385; b) Burger K., Höß E., Geith K. Synthesis 1990, 360. [ Links ]
9. a) Burger K., Ottlinger R., Goth H., Firl J., Chem. Ber. 1982, 115, 2494; b) Burger K., Geith K., Sewald N. J. Fluorine Chem. 1990, 46, 105; c) Burger K., Helmreich B. J. prakt. Chem. / Chem. Ztg. 1992, 334, 311. [ Links ]
10. a) Burger K., Höß E., Sewald N., Geith K., Riede J., Bissinger P., Z. Naturforsch. 1990, 45b, 1695; b) Burger K., Geith K., Höß E. Synthesis 1990, 352. [ Links ]
11. Höß E. PhD, Techn. University Munich, 1990. [ Links ]
12. work in progress.