Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de la Sociedad Química del Perú
Print version ISSN 1810-634X
Rev. Soc. Quím. Perú vol.84 no.4 Lima Oct./Dic. 2018
ORIGINAL ARTICLES
Xanthones from Hypericum laricifolium Juss., and their antiproliferative activity against HEP3B cells
Xantonas aisladas de Hypericum laricifolium y su actividad antiproliferativa contra las células HEP3B
Marlon Miguel Morales Moiselaa, Orianne Brelb, Geneviève Bourdyb, Mercedes Gonzales de La Cruza, Inés Yolanda Castro Dioniciob,c, Maelle Carrazb, Valérie Jullianbc*
* valerie.jullian@ird.fr, UMR 152 Pharmadev, Université de Toulouse, IRD, UPS, Faculté de Pharmacie,35 chemin des maraichers, 31062 Toulouse Cedex 09, France
a. Universidad Ricardo Palma, Av. Benavides 5440, Santiago de Surco, Lima 33, Perú
b. UMR 152 Pharmadev, Université de Toulouse, IRD, UPS, France. .
c. Laboratorios de Investigación y Desarrollo, Universidad Peruana Cayetano Heredia (UPCH), Lima 34, Perú
ABSTRACT
We describe here the isolation of 6 xanthones and one phenyl-γ-pyrone, together with 3-epibetulinic and quercetine from the aerial parts of Hypericum laricifolium Juss. (Hypericaceae), collected in Peru. Isolated compounds have been evaluated against proliferation of human hepatocellular carcinoma Hep3B cells. The most active compound was xanthone 3, with an inhibitory concentration (IC) of 12 µM. 50
Key words: Xanthones, Hypericum laricifolium, Hep3B cells, hepatocellular carcinoma
RESUMEN
En el presente trabajo se describe el aislamiento de 6 xantonas, una fenil-γ-pirona, el ácido 3-epibetulínico y quercetina a partir de las partes aéreas de Hypericum laricifolium Juss. (Hypericaceae), colectada en Perú. Los compuestos aislados fueron evaluados por su capacidad para inhibir el crecimiento de células de hepatocarcinoma humano Hep3B. El compuesto más activo fue la xantona 3, con una concentración inhibitoria (CI) de 12 µM. 50
Palabras clave: Xantonas, Hypericum laricifolium, células Hep3B, hepatocarcinoma
INTRODUCTION
The genus Hypericum includes 469 species, and belongs to the Hypericaceae family. This genus is present all over the world, with native or introduced species. Hypericum laricifolium Juss. is distributed in South America along the Andes cordillera from Western Venezuela, to the eastern part of Colombia, Ecuador and Peru. In Peru, H. laricifolium is traditionally used in the high Andes for bath against cold and post-partum bath under the name "cipres" or "romerillo"1, its also known as "Chinchango". Two previous chemical investigations have been published and report the presence of hentriacontanyl caffeate, nonacosanyl caffeate. stigmasterol, β-sitosterol, 3-epi-betulinic acid, caffeic acid, ferulic acid, docosanol, p-hydroxybenzoic acid, 3,4-dimethoxy benzoic acid, quercetin, quercetin- 3-O-galactoside, quercetin-3-O-rutinoside, quercetin-3-O-rhamnoside, quercetin-3-O- glucuronide and shikimic acid2, as well as xanthones, 1-hydroxy-7-methoxyxanthone, 1,7-dihydroxyxanthone, 2-hydroxyxanthone, 6-deoxyisojacareubin, 1,3-dihydroxy-6- methoxyxanthone, and 1,5,6-trihydroxy-7-methoxyxanthone3.
In a former publication1, we have described the good activity of an ethanolic extract from Hypericum laricifolium against the growth of human hepatocellular carcinoma Hep3B cells. The lack of effective treatments against human hepatocellular carcinoma (HCC) prompted us to isolated and identify the compounds responsible for this activity.
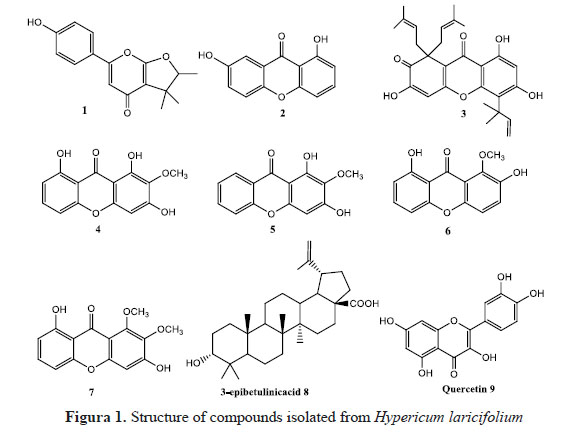
In this work, we report the isolation from Hypericum laricifolium aerial part of one phenyl- γ-pyrone, the hyperbrasilone 1 and 6 xanthones 2-7. Hyperbrasilone 1 and xanthones 3-7 are described for the first time in the species. 3-epi-betulinic acid 8 and quercetine 9 have also been isolated. Eight compounds have been evaluated against proliferation of human hepatocellular carcinoma Hep3B cells.
EXPERIMENTAL PART
Plant material
Aerial parts of Hypericum laricifolium Juss. were collected in Huaraz surroundings (Ancash department, Peru), at the altitude of 3900 m a.s.l. and identified by specialists.
Isolation of compounds
Aerial parts were dried at ambient temperature, away from sun, and then grinded. 650 g of powdered plant were then extracted three times with 5.6 L of ethanol 96o, to give 91.5 g of extract E. Extract E (55 g) was partitioned in 2 L of water and 2 L of ethyl acetate, to give an aqueous phase, and 21.6 g of an extract EA. 1.4 g of extract EA were submitted to two successive medium pressure liquid chromatographies (MPLC) on silica gel to give 6.6 mg of hyperbrasilone 1 and 4.3 mg of 1,7-dihydroxy-xanthone 2.
Then, 9.1 g of EA extract were partitioned in 280 mL of petroleum ether and 280 mL of methanol-water 90-10, to give 2.40 g of extract PE and 6.70 g of extract MW. The extract MW (6.7 g) was absorbed on 70 g of silica gel. This solid was then successively extracted with mixtures of petroleum ether-ethyl acetate of increasing polarity to give four fractions A (1.5 g), B (1.5 g), C (1.7 g), D (1.7 g). Fraction A, extracted with a mixture of petroleum ethe / ethyl acetate 80 / 20, was submitted to column chromatography on silica gel, eluted with mixtures of petroleum ether / ethyl acetate of increasing polarity, from petroleum ether / ethyl acetate 80 / 20 to pure ethyl acetate. 15 fractions were obtained, A1 to A15.
A2 (97.4 mg) was submitted to column chromatography on silica gel, eluted with petroleum ether then petroleum ether / dichloromethane 50 / 50 to give A2-2 (8.0 mg), identified as 3 A4 (115.0 mg) was submitted to column chromatography on silica gel, eluted with mixtures of dichloromethane / methanol of increasing polarity, from pure dichloromethane to dichloromethane 2% methanol, to give A4-1 (3.4 mg), identified as 1,3,8-trihydroxy-2- methoxy-xanthone 4, A4-2 (5.1 mg), A4-3 (5.5 mg) and A4-4 (37.2 mg). A4-2 and A4-3 were submitted to chromatography on Sephadex LH-20, eluted with methanol to give respectively A4-2-1 (3.0 mg) identified as 1,3-dihydroxy-2-methoxyxanthone 5, and A4-3-1 (1.0 mg) identified as 2,8-dihydroxy-1-methoxyxanthone 6. A4-4 was washed with methanol to give A4-4-1 as a white precipitate (11.5 mg) identified as 3-epi-betulinic acid 8.
A7 (19.1 mg) was submitted to successive columns chromatographies on silica gel and Sephadex LH-20 to give 3,8-dihydroxy-1,2-dimethoxyxanthone (0.6 mg) 7.
A13 (50.0 mg) was washed with dichloromethane / methanol 90 / 10 to give 8.2 mg of a yellow solid identified as quercetine 9.
Identification of compounds
Hyperbrasilone 1 was isolated as a colorless oil. 1H-RMN (CDCl ) δ: 1.25 (3H, s), 1.42 (3H, d, J = 6.7 Hz), 1.44 (3H, s), 4.59 (1H, q, J = 6.7 Hz), 6.40 (1H, s), 6.98 (2H, d, J = 4.9 Hz), 6.67 (2H, d, J = 4.9 Hz). 13C-RMN (CDCl ) δ: 14.6, 20.3, 25.5, 42.7, 91.8, 92.3, 108.3, 116.1, 123.9, 127.9, 159.1, 161.7, 163.5, 170.5. These data were compared with those previously published4. The absolute configuration of the chiral center was not determined, neither in this work, nor in the original publication.
1,7-dihydroxyxanthone 2 was isolated as a yellow solid. 1H-RMN (CDCl + 10% CD OD) δ: 6.75 (1H, dd, J = 8.2, 0.9 Hz) , 6.91 (1H, dd, J = 8.4, 0.9 Hz), 7.29 (1H, dd, J = 9.0, 3.0 Hz), 7.36 (1H, d, J = 9.4 Hz), 7.50 (1H, d, J = 2.8 Hz), 7.56 (1H, dd, J = 8.3, 8.3 Hz). 13C-RMN (CDCl + 10% CD3OD) δ: 107.1, 108.2, 109.7, 119.1, 120.8, 125.5, 136.6, 153.7, 156.5, 161.3, 182.5. APCI-MS m/z: 229 (MH+). These data were compared with those previously published5.
Compound 3 was isolated as a yellow-orange oil 1H-RMN (CDCl ) δ: 1.52 (6H, se), 1.53 (6H, se), 1.71 (6H, se), 2.84 (2H, dd, J = 13.7, 8.1 Hz), 3.45 (2H, dd, J = 13.7, 7.0 Hz), 4.68 (m, 2H), 5.42 (1H, dd, J = 10.5, 0.8 Hz), 5.50 (1H, dd, J = 17.8, 0.8 Hz), 6.32 (s, 1H), 6.48 (1H, dd, J = 17.8, 10.5 Hz), 6.50 (s, 1H), 13.44 (s, 1H). 13C-RMN (CDCl ) δ: 18.0, 25.8, 28.2, 37.9, 40.9, 55.8, 101.6, 105.7, 108.2, 109.2, 113.6, 116.2, 117.7, 135.4, 149.0, 151.8, 154.8, 158.6, 161.1, 161.3, 179.5, 201.4. APCI-MS m/z: 465 (MH+). These data were compared with those previously published6. 1,3,8-trihydroxy-2-methoxyxanthone 4 was isolated as a yellow solid. 1H-RMN (CDCl +10% CD OD) δ: 3.91 (s, 3H), 6.44 (s, 1H), 6.73 (1H, d, J = 8.3 Hz) , 6.84 (1H, d, J = 8.4 Hz), 7.52 (1H, t, J = 8.4 Hz), 11.94 (s, 1H), 12.11 (s, 1H). 13C-RMN (CDCl + 10% CD OD) δ: 60.8, 94.4, 102.4, 107.0, 107.0, 110.4, 130.5, 136.6, 153.3, 153.4, 156.1, 158.4, 160.8, 184.8. APCI-MS m/z: 275 (MH+). These data were compared with those previously published7.
1,3-dihydroxy-2-methoxyxanthone 5 was isolated as a yellow solid. 1H-RMN (CDCl ) δ: 4.07 (s, 3H), 6.57 (s, 1H), 7.41 (1H, ddd, J = 8.4, 1.1, 0.5 Hz), 7.47 (1H, ddd, J = 8.1, 7.1, 1.1 Hz), 7.75 (1H, ddd, J = 8.0, 1.7, 0.5 Hz), 8.28 (1H, ddd, J = 8.5, 7.1, 1.7 Hz). 13C-RMN (CDCl ) δ: 61.0, 93.4, 104.2, 117.7, 120.2, 124.0, 125.8, 129.5, 135.0, 153.4, 153.4, 156.1, 156.4, 181.3. APCI-MS m/z: 259 (MH+). These data were compared with those previously published8.
2,8-dihydroxy-1-methoxyxanthone 6 was isolated as a white solid. 1H-RMN (CDCl ) δ: 4.07 (s, 3H), 6.80 (1H, dd, J = 8.2, 1.0 Hz), 6.90 (1H, dd, J = 8.4, 1.0 Hz), 7.24 (1H, d, J = 9.0 Hz), 7.45 (1H, d, J = 9.1 Hz), 7.59 (1H, dd, J = 8.3, 8.3 Hz), 12.91 (s, 1H). 13C-RMN (CDCl3) δ: 62.7, 106.5, 109.0, 110.3, 114.2, 114.7, 123.3, 136.6, 144.2, 145.4, 150.8, 155.8, 161.9, 182.1. APCI-MS m/z: 259 (MH+). These data were compared with those previously published9.
3,8-dihydroxy-1,2-dimethoxyxanthone 7 was isolated as a yellow solid. 1H-RMN (CDCl ) δ: 4.04 (s, 3H), 4.06 (s, 3H), 6.78 (1H, dd, J = 8.2, 1.0 Hz), 6.81 (s, 1H), 6.85 (1H, dd, J = 8.3, 1.0 Hz), 7.54 (1H, dd, J = 8.3, 8.3 Hz), 13.14 (s, 1H). 13C-RMN (CDCl ) δ: 61.8, 62.0, 99.0, 106.2, 108.8, 109.2, 110.6, 136.0, 137.4, 152.2, 154.6, 155.5, 155.8, 162.1, 181.5. APCI-MS m/z: 289 (MH+). These data were compared with those previously published10.
3-epibetulinic acid 8 was isolated as a white solid. 1H-RMN (CDCl + 10% CD3OD) δ: 0.61 (1H, m), 0.68 (3H, s), 0.75 (3H, s), 0.87 (3H, s), 0.88 (3H, s), 0.90 (3H, s), 1.65 (3H, s), 1.03-1.70 (19H, m), 1.87 (2H, m), 2.18 (2H, m), 2.94 (1H, td, J = 10.6-4.5 Hz), 3.09 (1H, dd, J =10.6-5.7 Hz), 4.53 (1H, s), 4.65 (1H, s). 13C-RMN (CDCl + 10% CD3OD) δ: 179.2, 150.5, 109.4, 78.8, 56.3, 55.5, 50.5, 49.2, 46.9, 42.4, 40.6, 38.7, 38.6, 38.2, 37.1, 37.0, 34.2, 32.2, 31.9, 30.6, 27.8, 27.0, 25.4, 20.9, 19.2, 18.3, 16.1, 15.9, 15.3, 14.6. APCI-MS m/z: 439 (M-OH)+. These data were compared with those previously published11.
Quercetin 9 was isolated as a yellow solid. 1H-RMN (DMSO-d ) δ: 6.19 (1H, d, J = 2.0 Hz), 6.41 (1H, d, J = 2.0 Hz), 6.89 (1H, d, J = 8.5 Hz), 7.54 (1H, dd, J = 8.5, 2.2 Hz), 7.68 (1H, d, J = 2.2 Hz). 13C-RMN (DMSO-d ) δ: 93.9, 98.6, 103.4, 115.5, 116.0, 120.4, 122.4, 136.3, 145.4, 147.3, 148.2, 156.5, 161.2, 164.4, 176.3. APCI-MS m/z: 303 (MH+). These data were compared with those previously published12.
Measurement of the antiproliferative activity on Hep3B cells.
An already published protocol was followed1, with slight modifications. The human cancer cell line Hep3B has been cultivated as described and double each 20 h in our culture conditions. In this assay, Hep3B cells were seeded in 96-well plates at a density of 5 x 103 cells per well and after 18 h of adhesion, incubated for 72 h with compounds following the concentration range: 0.1, 1, 10, 25, 50, 100 µg/mL in DMEM supplemented with 0.1% of DMSO. Their growth was measured using an ATP-based luminescence assay (ATPliteTMkit, Perkin Elmer, France). 72 h after cells treatment by extracts, luminescence was read on a Trilux reader (Perkin Elmer). The 50% inhibitory concentration (IC50) of each extract was determined based on 3 independent experiments using the GraphPad Prism software (GraphPad Software Inc., USA).
RESULTS AND DISCUSSION
Compounds have been isolated after successive chromatographies on silica gel and Sephadex LH-20 from an ethanolic extract of Hypericum laricifolium aerial part displaying a good antiproliferative activity on Hep3B cells (IC50 = 11.5 µg/mL). Their structures were established on the basis of 1D and 2D NMR spectra (1H, 13C, COSY, HSQC, HMBC) and mass spectrometry, and verified by comparison with previously published spectroscopic data.
3-epibetulinic acid 8 and quercetin 9 are ubiquitous in plants, and have already been isolated from H. laricifolium2,3. 1-7 dihydroxyxanthone 2 was also reported in Hypericum laricifolium3. Interestingly, all other 6 compounds are reported in Hypericum laricifolium for the first time, and 1,3,8-trihydroxy-2-methoxyxanthone 4 and 2,8-dihydroxy-1-methoxyxanthone 6 have never been reported in the genus Hypericum. Interestingly, the 4 xanthones 4-7 reported here are different from the xanthones reported in a previous work3, describing the chemical composition of Hypericum laricifolium collected in Venezuela, suggesting that xanthones metabolites of this species could vary according to location, or season of collect, and raises the hypothesis of an epigenetic influence of xanthones metabolites production in the H. laricifolium specie.
Concerning the antiproliferative activity on human Hep3B cells, the interesting activity previously observed for an ethanolic extract of H. laricifolium prompted us to test the isolated compounds on Hep3B cells culture and determine their IC . The most active compounds are xanthone 3 and 1,3,8-trihydroxy-2-methoxyxanthone 4 with an IC of 12 µM and 22 µM respectively. These activity are in the same range as the activity of the reference drug Sorafenib (Table 1).
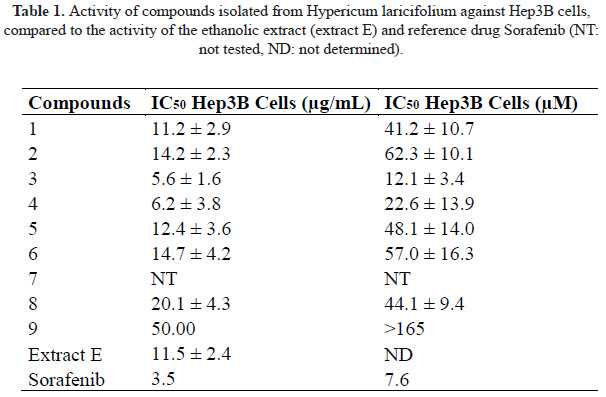
To the best of our knowledges, the only activity of xanthones or xanthone derivatives against the growth of Hep3B cells is reported for gambogic acid, a xanthone derivatives isolated from the tree Garcinia hanburryi. Gambogic acid inhibits the growth of Hep3B through apoptotic pathways with an IC of 1.8 µM13.
Allaxanthone C, structurally close to 3, have been shown to induce a prolongation of survival in a xenograft murine model of human chronic lymphocytic leukemia14.
1,3,8-trihydroxy-2-methoxyxanthone 4 was also tested again different multidrugresistant human tumor cells and was moderately active against taxol resistant SMMC-7721 human liver carcinoma cell line (IC = 42 µM)15. All of this suggest that xanthones structurally close to 3 or 4 should be tested against Hep3B cells lines in order to find a potential lead against hepatocellular carcinoma, however the activity of isolated compounds is rather moderate compared to the activity of the crude ethanolic extract of H. laricifolium (IC = 11.5 µg/mL), suggesting that other compounds with better activity may be present in the plant, or that some synergetic effect may occur between the constituents of this species.
CONCLUSION
In this work, we have identified xanthones inhibiting the growth of Hep3B cells. This highlight the interest of this class of compounds, and especially xanthones structurally related to compound 3, in the search of active compounds against human hepatocellular carcinoma.
ACKNOWLEDGMENTS
We thank the Ando-Amazonian International Mixed Laboratory (LMI LAVI, IRD-UPCH) and its co-directors Dr Michel Sauvain (IRD, France) and Dra. Rosario Rojas (UPCH, Peru), to have hosted in the UPCH the greatest part of the chemical work described in this publication.
REFERENCES
1. Carraz M, Lavergne C, Jullian V, Wright M, Gairin JE, Gonzales de la Cruz M, et al. Antiproliferative activity and phenotypic modification induced by selected Peruvian medicinal plants on human hepatocellular carcinoma Hep3B cells. J Ethnopharmacol. 2015;166:185-99. [ Links ]
2. El-Seedi HR, Ringbom T, Torssell K, Bohlin L. Constituents of Hypericum laricifolium and their cyclooxygenase (COX) enzyme activities. Chem Pharm Bull. 2003;51(12):1439- 40. [ Links ]
3. Ramírez-González I, Amaro-Luis JM, Bahsas A. Xanthones from aerial parts of Hypericum laricifolium Juss. Nat Prod Commun. 2013;8(12):1731-2. [ Links ]
4. Rocha L, Marston A, Kaplan MAC, Stoeckli-Evans H, Thull U, Bernard T, et al. An antifungal γ-pyrone and xanthones with monoamine oxidase inhibitory activity from Hypericum brasiliense. Phytochem. 1994;36(6):1381-5. [ Links ]
5. Kato L, Alves de Oliveira CM, Vencato I, Lauriucci C. Crystal structure of 1,7-dihydroxyxanthone from Weddellina squamulosa Tul. J Chem Crystallogr. 2005;35(1):23-6. [ Links ]
6. An TY, Hu LH, Chen ZL. A new xanthone derivative from Hypericum erectum. Chin Chem lett. 2002;13(7):623-4. [ Links ]
7. Kang W-Y, Ji Z-Q, Wang J-M. A new xanthone from the roots of Securidaca inappendiculata. Chem Pap. 2009;63(1):102-4. [ Links ]
8. Tanaka N, Takaishi Y. Xanthones from Hypericum chinense. Phytochem. 2006;67:2146- 51. [ Links ]
9. Kijjoa A, Joseâ M, Gonzalez TG, Pinto MMM, Damas AM, Mondranondra I-O, et al. Xanthones from Cratoxylum maingayi. Phytochem. 1998;49(7):2159-62. [ Links ]
10. Marston A, Hamburger M, Sordat-Diserens I, Msonthi JD, Hostettmann K. Xanthones from Polygala nyikensis. Phytochem. 1993;33(4):809-12. [ Links ]
11. Pieroni LG, de Rezende FM, Ximenes VF, Dokkedal AL. Antioxidant activity and total phenols from the methanolic extract of Miconia albicans (sw.) Triana leaves. Molecules. 2011;16:9439-50. [ Links ]
11. Fossen T, Pedersen AT, Andersen OM. Flavonoids from red onion (Allium cepa). Phytochem. 1998;47(2):281-5. [ Links ]
12. Lee PN, Ho WS. Antiproliferative activity of gambogic acid isolated from Garcinia hanburyi in Hep3B and Huh7 cancer cells. Oncol Rep. 2013;29:1744-50. [ Links ]
13. Loisel S, Le Ster K, Meyer M, Berthou C, Youinou P, Kolb J-P, et al. Therapeutic activity of two xanthones in a xenograft murine model of human chronic lymphocytic leukemia. J Hematol Oncol. 2010;3(1):49-51. [ Links ]
14. Wang Q, Ma C, Ma Y, Li X, Chen Y, Chen J. Structure–activity relationships of diverse xanthones against multidrug resistant human tumor cells. Biorg Med Chem Lett. 2017;27:447-9. [ Links ]
Recibido el 18-05-18
Aprobado el 29-11-18