Introduction: history of Andean pseudocereals (APS) in beer
Beer is the most consumed millenary drink in the world (Cantrell, 2008). In 2021 its production reached 1.86 billion hectoliters, with China, the United States, and Brazil as the leading manufacturing countries (DATAMARK, 2022). Traditionally, beer is prepared from the fermentation of malted cereals (generally barley), hop, yeast, and water, resulting in an alcoholic beverage with a distinctive bitter taste (Harrison & Albanese, 2019; Howe, 2020). However, the formula may vary by region, adapting to consumer preferences and needs. According to Gómez-López (2020) and Salvador (2022), today, the industry is looking for raw materials that produce beverages with unique flavors, and at the same time than consumers seek healthier and more nutritious food options. In this scenario, beers made from Andean pseudocereals (APS) have been gaining popularity both for their protein quality and the health benefits they have associated with (Martínez-Villaluenga et al., 2020; Schoenlechner, 2017). Moreover, APS grains are also suitable for celiacs (gluten intolerant) or those who prefer a gluten-free diet.
It is estimated that APS originated about 5000 years ago in the central region of the Andes, which has the right conditions for their growth, such as cold weather and high altitudes (Di Fabio & Parraga, 2016; Haros & Schonlechner, 2016). Countries in the Andean region, such as Peru, Ecuador, and Bolivia, are today responsible for most of the APS production (90%), however, some crops (5 to 8%) can be found in the US, Denmark, Argentina, and Sweden (FAOSTAT, 2022). The best-known APS are amaranth, cañihua, and quinoa, the latter with the largest area of cultivation and production (Figure 1).
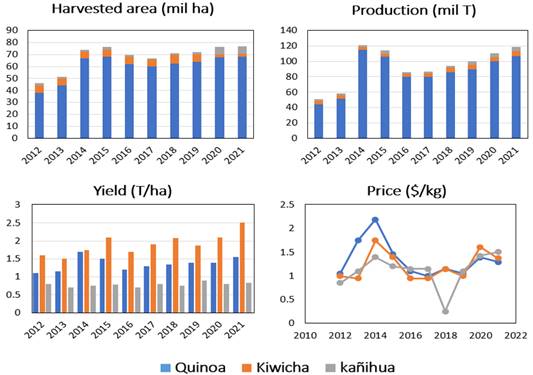
Figure 1 Agricultural production data of the main Peruvian Andean pseudocereals (APS) from 2012 to 2021. Source: Statistical Annual of agricultural production - Integrated System of Agrarian. Statistics of Peru (MINAGRI, 2022).
APS popularity began in the early 2000s, when studies revealed that, unlike cereals and legumes, their seeds provide all the essential amino acids and minerals such as calcium and magnesium (Belton et al., 2002; Gorinstein et al., 2002; Segura-Nieto et al., 1999), in addition to being rich in starch and gluten-free. Later, their bioactive potential would be known due to their high content of phenolic compounds and flavonoids with antioxidant capacity (Campos et al., 2018; Peñarrieta et al., 2008; Repo-Carrasco-Valencia et al., 2010), proving that APS have health benefits as they can help in the control of diabetes and hypertension (Martínez-Villaluenga et al., 2020; Ranilla et al., 2009). Processes such as fermentation, germination, and malting can increase the concentration of bioactive compounds and antioxidant capacity of APS (Abderrahim et al., 2012; Mäkinen et al., 2013; Modgil & Sood, 2017; Paucar-Menacho et al., 2017, 2018; Pilco-Quesada et al., 2020). Likewise, these processes can improve the digestibility and bioavailability of their nutrients (Motta et al., 2019; Najdi Hejazi et al., 2016; Omary et al., 2012), so APS was suggested as an alternative substrate in brewing where it would have an advantage over cereal malt.
Due to the physical characteristics and composition of APS seeds, the malting process is different from conventional cereals (barley, wheat, and corn). By having a small size, their seeds germinate fast; however, their starch presents a higher viscosity, requiring a longer maceration time with more controlled parameters (Meo et al., 2011; Phiarais & Arendt, 2008). In addition, depending on the type (quinoa, amaranth, or cañihua), drinks with an alcoholic strength similar to ordinary beers or low in alcohol can be obtained (Phiarais & Arendt, 2008).
The first applications of APS in producing beers aimed to replace barley malt to obtain gluten-free beverages. For example, Zarnkow et al. (2005) obtained beverages with alcoholic strength similar to barley beer from quinoa malt; however, these had a lower amount of foam, a lighter color, and a bitter taste, so their suitability was limited. Subsequent studies proposed the partial replacement of barley malt with quinoa and amaranth malt to increase the nutritional value and functional activity of beer (Castañeda, 2015; Martínez & Tuano, 2018; Montenegro, 2016; Rodriguez, 2015) and, at the same time decrease their alcohol content (Villamarín, 2017; Zapata, 2016). On the other hand, its incorporation as a grain without malting is also sought to create new flavors and add value to this drink (Bellut et al., 2019; Buiatti et al., 2018; Kordialik-Bogacka et al., 2018).
Currently, most of the production of APS beers belongs to the artisanal sector. However, considering the available literature and studies that support their nutritional and functional potential, it is likely that their commercialization on an industrial scale is not far away. In this sense, this review is a compilation of the studies carried out on the main APS (quinoa, amaranth, and cañihua) and their potential for beer production, focusing on the characteristics that are of interest to this industry, parameters, and processing effects, as well as the different applications that have been carried out in this area.
General characteristics
2.1 Quinoa
Quinoa (Chenopodium quinoa, Amaranthaceae) is a native crop from the Andean region located between Peru and Bolivia. This plant produces resistant to environmental stress seeds with high nutritional. According to Asher et al. (2020), quinoa has high-quality proteins containing all the essential amino acids, moreover, it has a high content of bioactive compounds. As for their morphology, the seeds are characterized by being small (1.5 to 2.5 mm), flat with an oval shape, and presenting a variety of colorations ranging from pale yellow, reaching pink or black tones (Aluwi et al., 2017).
As for quinoa varieties, South America has around 3000 types stored in genebanks (Vega‐Galvez et al., 2010). According to the National Institute of Agricultural Innovation (INIA), within the commercial varieties of Peruvian quinoa there are: INIA 431-Altiplano, INIA 427 - Yellow Sacaca, INIA 420 - Black Collana, INIA 415 - Pasankalla, Illpa INIA, Salcedo INIA, Quillahuaman INIA, Ayacuchana INIA, Amarilla Marangan, Juli White, Junín White, Cheweca, Huacariz, Hualhuas, Huancayo, Kankolla, Mantaro, Junín Pink, Taraco Pink, and Yanamango Pink (Vidal et al, 2015). In a recent study, Valdez-Arana et al. (2020) tested the potential as a food ingredient for several cereal-based products of the Blanca de Hualhuas, Rosada from Huancayo, and Pasankalla varieties, based on their physicochemical and functional properties. Some of these quinoa varieties are shown in Figure 2 (a, b, and c).
The chemical composition of quinoa grains varies depending on the variety and growing conditions (Valdez-Arana et al., 2020). Table 1 shows the proximal composition of some types of Peruvian quinoa. Quinoa has a superior nutritional quality compared to other commercial grains such as rye, barley, wheat, rice, and oats, likewise, it has a higher content of proteins, lipids, and ash (Vilcacundo & Hernández-Ledesma, 2017, Pereira et al., 2019).
The protein content of quinoa (expressed in g/100 g of edible matter) ranges from 13.1% to 16.7% (Vilcacundo & Hernández-Ledesma, 2017). As mentioned above, quinoa seeds have a high biological value since they contain all the essential amino acids for human diet, highlighting lysine (between 2.4 and 7.8 g/100g of protein), methionine (0.3 to 9.1 g/100g of protein), and threonine (2.1 to 8.9 g/100g of protein) which are the limiting amino acids in conventional cereals, such as wheat and corn (Vilcacundo & Hernández-Ledesma, 2017). As for carbohydrates, starch is the element found in greater quantity (32% to 69%), while total dietary fiber is similar to other cereals (7% to 9.7%) (Vilcacundo & Hernández-Ledesma, 2017). Due to the quality and quantity of its lipid fraction, quinoa could be considered an alternative oily seed. According to Filho et al. (2017), quinoa seeds have an oil rate of 2.0% to 9.5%, rich in essential fatty acids such as linoleic and alpha-linolenic acids.
Quinoa is also rich in micronutrients as vitamins and minerals (Navruz-Varli & Sanlier, 2016). Although vitamin research is limited, it is known that seeds contain high concentrations of pyridoxine (B6) and folic acid (James, 2009). The ash content of quinoa (3.4%) is higher than rice (0.5%), wheat (1.8%), and other commercial cereals grains, which could be associated with higher amounts of miner als in its seeds, as calcium and iron (Vega-Gálvez et al. 2010; Repo-Carrasco-Valencia et al., 2011). Furthermore, Paucar-Menacho et al. (2018) research showed that sprouted quinoa contains a high concentration of flavonoid compounds and phenolic acids.
2.2 Amaranth
Amaranth (Amaranthus caudatus), also known as kiwicha, is a native crop from South America. In Peru, amaranth crops show great biodiversity due to the various agroeco logical zones of the country, where it has been consumed for hundreds of years as cooked and roasted whole grains or as flours in traditional foods such as cooked dishes, puddings, and salads (Pilco-Quesada et al., 2020).

Figure 2 Most commercialized Andean pseudocereals (APS) varieties: (a) pearl white quinoa, (b) red quinoa (Pasankalla), (c) black quinoa, (d) amaranth, and (e) cañihua. Note: Seeds scattered on a white background using a ruler graduated in cm to the left.
Amaranth is an APS that belongs to the Amaranthaceae family and has more than 80 species. The seeds are spherical and small (1 to 1.5 mm) (Figure 2c). According to the Ministry of Agriculture of Peru (MINAG, 2016), Amaranthus caudatus, Amaranthus hypocondriacus, and Amaranthus cruentus are the three main species produced in the country.
Interest in APS for formulating healthy, high-quality gluten-free products such as bread and pasta increased considerably (Alvarez-Jubete et al., 2010; Paucar-Menacho et al., 2017). Amaranth is a good source of starch (58% and 66%), proteins (13% and 19%) with an adequate balance of amino acids, fiber-rich in pectic substances, and xyloglucans (14 and 16%), lipids containing significant amounts of squalene (5% and 13%), minerals (Ca, Fe, Mg, Mn, K, P, S, and Na), and complex-B vitamins (Lamothe et al., 2015; Nascimento et al., 2014). Moreover, amaranth contains health-promoting compounds, including bioactive peptides, phenolic compounds, and betacyanins (Montoya-Rodriguez et al., 2015; Repo-Carrasco-Valencia et al., 2010). Researchers also observed that sprouted amaranth generates flours with better nutritional content and phytochemical and antioxidant activity suggesting their use in bakery products (Paucar-Menacho et al., 2017).
In Peru, thanks to the APS genetic improvement program promoted by the Ministry of Agriculture and Irrigation (MINAGRI), some amaranth varieties have been developed as Ayacuchana - INIA (obtained by the National Program of Andean Crops), 10-C, 41-F, San Luis, Otusco, Rojo Cajamarca, E-13 and E-2008 that grow in Cusco, Ayacucho, and Cajamarca regions respectively. Due to its high amaranthine content, the Rojo Cajamarca variety is mainly used in beverages or soft colored drinks (chicha). There is also the Consuelo variety, obtained in Cusco recently, which was introduced as CAC-2074-BA 87 (Juan & Siguas, 2019). In 2020, the Canaán Ayacucho Agricultural Experimental Station, of the National Institute of Agrarian Innovation-Peru presented a new kiwicha variety named INIA 442-La Rendidora, which is adapted adequately to the agroecological conditions of the Ayacucho region, in terms of production and grain quality (INIA, 2020). From a commercial point of view, the most recommended varieties are "Centenario" and "Oscar Blanco" which present the highest yields (Roque, 2019).
2.3 Cañihua
Cañihua (Chenopodium pallidicaule), is native crop from the highlands of Peru and Bolivia characterized by its high nutritional value (Huamaní et al., 2020). Its cultivation has remarkable adaptability to different agroecological regions, growing in semi-desert climates between 3600 and 4400 m of altitude. Cañihua seeds are not demanding in fertilization and irrigation and can develop even in impoverished soils (Peñarrieta et al., 2008). Thus, for people who live from subsistence agriculture in the highlands, cañihua is extremely important as the primary source of calories and good-quality protein food (Repo-Carrasco-Valencia et al., 2009a)
The cañihua is part of the Chenopodiaceae family, the same family as quinoa (Betalleluz-Pallardel et al., 2017), and its seeds are characterized by being polyhedral and very small, measuring less than 1mm (Figure 2e). Cañihua, along with amaranth and quinoa, could be considered a gluten-free, nutritious alternative, being used as a substitute for conventional cereals (Moreno et al., 2016; Rai et al., 2018; Zegarra et al., 2019). Its nutritional value (Table 1) is mainly related to its protein content ranging from 12.7% to 23.0%, which is higher than most commercial cereals (Gross et al., 1989; Repo-Carrasco et al., 2003; Ramos-Diaz et al., 2015; Betalleluz-Pallardel et al., 2017). Likewise, cañihua grains have a great balanced composition of essential amino acids, rich in lysine, the first limiting amino acid of common cereals (Repo-Carrasco et al., 2003). On the other hand, cañihua contains a high content of dietary fiber that has beneficial effects on health (Repo-Carrasco Valencia et al., 2017; Betalleluz-Pallardel et al., 2017).
Cañihua is gluten-free, becoming an attractive alternative for developing celiac disease or gluten-sensitivity products (Ramos-Diaz et al., 2015; Betalleluz-Pallardel et al., 2017). Rastrelli et al. (1996) and Repo-Carrasco-Valencia et al. (2009b) observed that the content of saponins in cañihua grains is low, ideal for a non-bitter food product. Cañihua is relatively rich in oil that contains mainly unsaturated fatty acids. According to Repo-Carrasco (2003), its tocopherol content is higher than corn oil. Its grains also offer various antioxidants, phenolic compounds, and flavonoids (Peñarrieta et al., 2008). Abderrahim et al. (2012) observed that the germination process of cañihua improves its total antioxidant capacity, including removable and non-removable antioxidant compounds (total extractable phenolics and non-extractable flavonoids).
APS malting
Malting is defined as the process of transforming grain starch into sugar chains necessary for the fermentation and formation of alcohol and CO2 (Phiarais & Arendt, 2008). The main objective of the process is to convert the nutritional reserves of the grain into the substrates required for brewing. According to Kalita et al. (2017), altering grain composition by malting can improve organoleptic characteristics such as the taste and color of derived food products.
The traditional malting process consists of three stages: hydration, germination (controlled), and drying; however, the process parameters may vary depending on the grain/raw material. For example, malting can take more than four days in conventional cereals such as barley and wheat (Solano, 2019), while in APS, the process occurs with greater speed due to the reduced size and high enzymatic activity of the seeds (Phiarais & Arendt, 2008). Figure 3 and Table 2 show the diagrams and process parameters of cereal vs. pseudocereal malting, including APS. Studies evidence differences in the process parameters of each stage which are discussed below.
Hydration
Hydration or soaking aims to break the grain's latent state, activating the enzymes for starch and protein degradation for yeast consumption (Molina, 1989). The grain is immersed in water until it reaches 43 to 46% moisture under aerobic conditions (with an oxygen supply).
Table 1 Proximal composition of the main Andean Pseudocereals (APS), including raw quinoa, amaranth, and cañihua (g/100g dry weight)
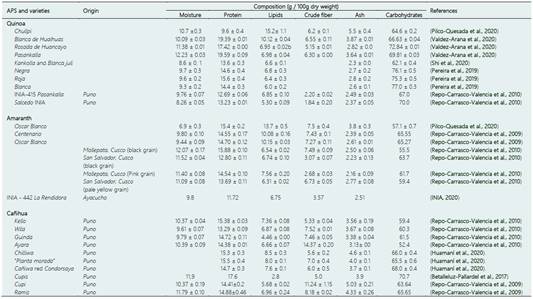
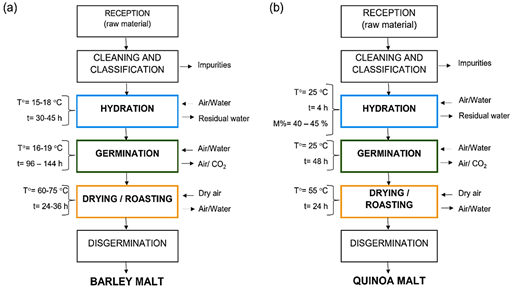
Figure 3 Elaboration process of a) Barley malt (cereal) (Solano, 2019) vs b) Quinoa malt (pseudocereal) (Aguilar et al., 2019).
In cereals soaking temperatures used ranged from 15 to 20 °C, for a time between 36 and 48 hours, using high-quality water such as drinking water (Phiarais & Arendt, 2008), while in pseudocereals such as quinoa, soaking is done at 25° C for 4h (Aguilar et al., 2019). As observed in Figure 3 and Table 2, the hydration time and temperature will depend on the raw material characteristics. According to Bradford (1995), these parameters are determined considering the physical diffusion properties of the grain, such as size, cover permeability, hydratable substances concentration, among others.
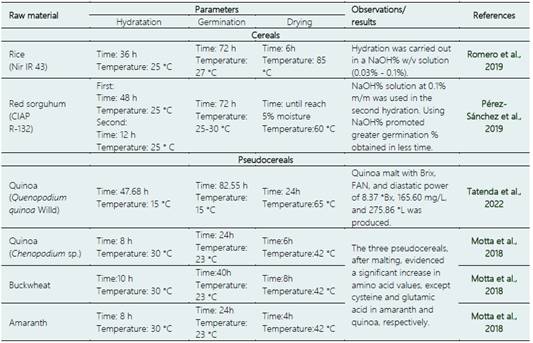
Several authors have reported that using agents, such as NaOH, can help to obtain a higher percentage of germinated grains in less time than just using water during hydration (Table 2).
Pérez-Sánchez et al. (2019) observed that red sorghum germination using NaOH at 0.1% m/m was 76.2%, while it was 74.1% using only water. However, Péres-Guerrero (2017) points out that, although pre-treatment of seeds soaked using NaOH (0.2% m/m) helps to prevent a mi crobial attack during germination, it can affect the enzyme pack of some pseudocereals as amaranth, inhibiting the synthesis of gibberellic acid and stopping the radicle growth. These observations suggest that NaOH use let to obtain contamination-free malt; however, it is necessary to conduct future research to know the appropriate concentration of the alkaline solution, allowing the proper germination and microbial inhibition of grains.
Germination
During the germination process, the physical modification of the endosperm occurs, increasing the bioactive compounds, nutritional value, and stability of the grains (Ha et al., 2016). Then, this process increases the nutrient profile and antioxidant capacity of food products.
In 2016, Hejazi & Orsat evaluated the effects of temperature and germination time on the primary nutrients of malted finger millet and amaranth. The grains were germinated for 24, 36, and 48 hours at 22, 26, and 30 °C. In finger millet grains, germination for 48 hours at 30 °C increased their protein availability (17%) and total energy (10%) and reduced the resistant starch (60%). Previously, Mbithi-Mwikya et al. (2000) have already observed an increase in the protein availability (30%) of finger millet after four days of germination at room temperature, showing that as the germination time increases, the protein availability from the millet is greater.
The recommended germination parameters for amaranth are 48 hours at 26 °C; in those conditions, the protein availability and total energy increase by 8% and 11%, respectively (Motta et al., 2018). Unlike the finger millet cereal, the first 24 hours of germination at 20 °C increased its protein availability by 5.5% (Hejazi & Orsat, 2016). Table 2 presents the optimal parameters established by studies of some pseudocereals, including APS. In general, pseudocereals require less germination time than cereals, resulting from the previous hydration of the grain tissues increasing the specific activity of numerous enzymes that promote the growth and elongation of the radicle (Motta et al., 2019). Likewise, hydrolysis during germination transforms the reserve nutrients into simpler molecules, with proteins as the primary energy reserve source, increasing their availability (Motta et al., 2017).
Drying
The drying stage involves passing the malt by a hot and dry airflow at various speeds and increasing temperature. The enzymes survival is influenced by the temperature and timing of the process regime (Briggs et al., 1981). Grains between 2 to 3 mm in diameter, such as pseudocereals, are commonly dried at 45 or 50 ºC until they reach 5% and 7% moisture, stopping the enzymatic reactions without destroying the enzymes.
As shown in Table 2, drying parameters can vary depend ing on the raw material. Besides interrupting the action of the enzymes in the malt, the drying process contributes to fixing desirable properties in the grain acquired during germination, it increases its fragility facilitating its grinding, modifies the chemical composition, and reduces the enzymatic content (Lopez & Ramirez, 2018).
Effect of malting on APS
Nutritional properties
As mentioned above, the malting process involves the controlled germination of grains that increase the activity of the hydrolytic enzymes affecting their principal nutritional reserves and enhancing the bioavailability of proteins, carbohydrates, vitamins, and minerals (Singh et al., 2015). The details of these effects are described below.
Proteins
During the malting process, a slight increase in the protein content of cereals and pseudocereals is promoted, specifically in amino acids such as glutamic acid, lysine, leucine, and valine. In 2019, Motta et al. studied the behavior of three varieties of malted pseudocereals (amaranth, buckwheat, and quinoa) subjected to different germination parameters (Table 2); 17 amino acids were identified, of which valine presented a significant increase of 35, 4.2, and 34% in the three varieties, respectively. Previously, Montanuci et al. (2017) had shown that protein values could vary by up to 13% using different temperature and hydration conditions of malting. For example, barley grains malted at 30 °C for 12 hours presented 10.9 g/100g of protein, while those malted at 10 °C for 24 hours showed a protein value of 9.58 g/100g, similar to the control without malting (9.64 g/100g).
In APS as amaranth, it was reported that crude protein and digestible protein contents increased by 41% and 22%, respectively, after 72 h of germination at 35 °C (Paredes-Lopez & Mora-Escobedo 1989). Research recommends germinating amaranth for 48 hours at 20 °C, since it increases protein availability by up to 8% without the risk of grain over-fermentation (Hejazi & Orsat, 2016). Protein availability increase is directly related to starch content reduction produced by germination since protein matrices are surrounded by starch granules that restrict their bioavailability. During germination, amylolytic activity (α-amylase) increases, promoting the starch granules breakdown (Elkhalifa & Bernhardt 2010); consequently, protein availability increases. Therefore, germination and malting can improve the composition and digestibility of proteins, suggesting that both are viable alternatives for consumption to take advantage of malted grains as a protein source for food products.
Fiber
Research on fiber content in malted cereals and pseudocereals has shown changes in dietary and crude fiber contents after germination. According to Desai (2004), changes in fiber composition are linked with cell wall degradation produced by germination. In 2016, Hejazi & Orsat sought to optimize the malting process parameters for finger millet using temperature ranges of 22 to 30 °C for 24 to 48 h. The sample increased 9% of dietary fiber at 22 °C for 48 hours, while during the first 24 hours, its content decreased by 0.8%. On the other hand, Colmenares de Ruiz & Bressani (1990) observed how amaranth increased its total dietary fiber content by 6.7% when germinated at 26 °C for 36 minutes while in a process at 30 °C for 48 hours, the fiber content decreased 6.2%. The effect of germination in crude fiber was studied using three varieties of amaranth. In A. caudatus variety, the crude fiber content in raw grains was 2.5% and became 2.4, 2.3, and 2.4% after 24, 48, and 72 h of germination. Similarly, for A. cruentus, crude fiber values were 2.5%, 2.4%, 2.6%, and 2.5%, and for A. hypochondriacus, 2.3%, 2.9%, 2.6%, and 2.4%.
The results of buckwheat germination indicated that the crude fiber content of germinated buckwheat flour was 56.52% higher than the ungerminated flour. This increase can be attributed to the starch breakdown (Shreeja et al., 2021), likewise, it could be linked to temperature and ger mination time. However, no study explains the variation of fiber content during germination using various germination parameters using different grains. Therefore, the study of future research in malting that includes the variation of fiber content among its main objectives is encouraged.
Minerals
Pseudocereals are an excellent source of minerals since their iron, zinc, calcium, magnesium, manganese, and copper content is higher than conventional cereals (Reguera & Haros, 2017). These elements cause chemical reactions and can be part of many tissues. For example, in 2004, FAO established daily intake levels for essential minerals recommending 1300 mg/day of calcium for adolescents and between 1000 to 1200 mg/day for adults. Iron is an oxygen carrier by hemoglobin from red blood cells (FAO, 2004); its daily intake requirements vary according to age and can affect physiological functions such as menstruation and lactation. A study conducted by Gómez-Álvarez (2004) recommends a daily iron intake of 7 to 9 mg for infants, 10 to 15 mg for males, and 10 to 18 mg for females.
Due to the importance of minerals, Zeiter (2016) studied the changes produced by malting in white and red sorghum mineral composition. The study found that after malting, the red sorghum increased its Zn, Fe, Ca, and Zn content by 11%, 16.32%, and 35.5%, respectively, but decreased its K content by 27.3%. On the other hand, in white sorghum Fe and Ca content increased by 18.6% and 44.3%, while Zn and K content decreased by 1.5% and 29.1%, respectively (Zeiter, 2016). These results confirm that malting is an excellent process for increasing Fe and Ca, essential minerals for the organism. Therefore, the consumption of malted grains is recommended since it provides a good percentage of essential minerals for the diet.
Functional properties
Total polyphenols
Pseudocereals present a wide diversity of flavonoids (anthocyanins, flavones, flavanones, isoflavonoids, and flavonols) and phenolic acids that can be presented in free soluble form or conjugated to sugars (Martínez-Villaluenga et al., 2020). According to Rochetti et al. (2019), including these elements in the diet can contribute to maintaining a healthy intestine by modulating the intestinal microbial balance (beneficial bacteria) and helping prevent chronic diseases.
During the malting process, the extraction of flavonoids and phenolic acids is more pronounced since polyphe nolic molecules can be found in the different stages and react with proteins during the boiling of the wort (Habschied et al., 2021). Concerning beer, characteristics such as bitter taste, astringency, body, and fullness are directly influenced by polyphenols from hops (30%) and malt (70%) (Habschied et al., 2021; Bogdan et al., 2016).
In 2016, Carciochi et al. compared the polyphenol content in raw and malted quinoa extracts observing an increase in the total phenolic acids and flavonoids after malting process. Thus, if the roasting temperature increased from 100 to 145 °C, total flavonoids and phenolic compounds increased up to 11 and 20 times, respectively, compared to raw seeds. However, temperatures above 190 °C can decrease phenolic acids and flavonoids due to the degradation of some phenolic compounds.
Montenegro (2016) produced an APS beer including 50% amaranth malt. The beer's consistency presented a higher foam capacity since it is produced by polyphenols (tannins) in suspension that reflect light. Likewise, the beer presented a more intense color than the control (barley beer), which can be associated with the oxidation of compounds that produce a dark color.
Vingrys et al. (2022) analyzed the effect of malting on bioactive compounds of cereals such as wheat and barley, and sorghum, observing that malted cereals are a great source of polyphenol-antioxidants. Malted cereals presented 1.5 to 2.8 times higher concentrations of polyphenols than the control. This change can be attributed to the increased enzymatic activity produced by the germination process and the Maillard reaction during the drying stage that generates endogenous antioxidants (Wannenmacher et al., 2018).
Antioxidants
The antioxidant capacity of pseudocereals can prevent oxidative damage in the human body (Tang et al., 2015). APS have a high level of antioxidants associated with their rich phenolic compound concentration due to their severe growing conditions, such as high altitude and cold climate (Choque-Quispe et al., 2021).
The antioxidant potential of malts plays an essential role in beer conservation during storage controlling its oxidative stability (Carvalho et al., 2014). In a recent study, Shopska (2021) states that the most increased antioxidant activity characterizes malts with a higher degree of heat treatment due to the content of Maillard reaction products with antioxidant capacity. Likewise, the conditions of each malting stage can significantly influence the antioxidant activity of the grains.
Traditional beer generally contains little phenolic acid content, which confers low antioxidant power and nutritional value. However, Scioli et al. (2022) in their research enriched a beer with different flavonoids from pseudocereals (quinoa, sorghum, amaranth), enhancing its nutritional value and energizing properties. During analysis, the enriched beer presented the content of bioactive substances as commercial food supplements, highlighting the possibility of using beer as a vehicle for bioactive compounds.
In a study where quinoa grains were malted, it was determined that there is a maximum increase in the inhibition of stable free radicals (DPPH), with an increase of 5 times more antioxidants in quinoa seeds processed at 100 to 145 °C. However, at higher roasting temperatures, the power of capture and reduction of DPPH radicals is significantly reduced by 25% and 19%, respectively. Then the authors recommend 145 ºC and 30 min as the optimal malting parameters for quinoa that increase the antioxidant compounds concentration, Maillard reaction products, and antioxidant activity (Durga et al., 2022).
Table 3 Bioactive compounds of three varieties of quinoa: INIA Salcedo, Passankalla Roja, and Negra Collana before and after malting
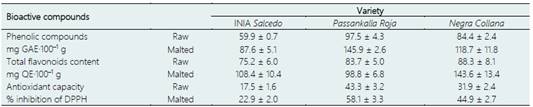
In addition, Aguilar et al. (2019) made a comparison of the values of total phenolic compounds, total flavonoid content, and antioxidant capacity of three varieties of quinoa ("Negra Collana," "Passankalla Roja" and "INIA Salcedo") before and after malting. According to the results (Table 3), the contents of phenolic compounds increased by up to 49% compared to non-malted grains, with The Red Passankalla being the variety with the highest value. Despite this, Carciochi et al. (2016) state that these compounds can increase up to 260% after malting since roasting favors the release of phenolic compounds. As for flavonoids, the values increased to 62.2% due to the germination and roasting time of the malting. Finally, the antioxidant capacity of the three quinoa varieties increased from 34% to 40%, depending on the germination time, since it is one of the metabolic changes that occur during the process (Bathgate,2016).
Folate
Another important group of bioactive compounds is the vitamins such as pyridoxine (B6) and folates (B9) contained in beer. According to Romanini et al. (2021), the vitamin content is variable and depends on malting and processing factors. These micronutrients are necessary for optimal bone health, growth, and development.
Motta et al. (2017) analyzed the effects of malting on the content of five folate vitamers in quinoa, amaranth, and buckwheat obtained by the enzymatic extraction method. The total folate content in malting increased significantly in amaranth by 21% (76 ± 14.2 μg/100 g) and buckwheat by 27% (193 ± 20.0 μg/100 g), while no change was observed in quinoa. However, the folate content presented in malted amaranth and quinoa contributes up to 25% of daily intake, demonstrating that pseudocereals are good sources of folates regardless of the process. It is because folates are found intracellularly, and during malting, they are released from the matrix when the structure of the seeds is destroyed. Hence, as the amaranth grains are small, they have a larger surface area for releasing folates from the seeds.
Ethyl alcohol
Ethyl alcohol is the main product of alcoholic fermentation in alcoholic beverages, such as beer, and its concentration determines the drink's classification by the percentage of alcohol. Moreover, it was proven that small amounts of ethyl alcohol in the daily diet protect against various diseases (Wachełk et al.,2021).
During fermentation stage, the highest rate of alcohol occurs when yeast reacts with glucose forming ethyl alcohol and carbon dioxide (Espinoza, 2016). A conventional beer with barley malt has 4% of alcohol. Beers formulated using pseudocereal malts usually exceed that percentage, Recalde (2017) brewed a beer from quinoa and corn malt with an alcohol content of 4.21%. Phiarais et al. (2010) obtained a beer with 4.53% alcohol using 100% buckwheat and amaranth malt (Montenegro, 2016). This behavior may be related to the rate of glucose degradation, which is faster in pseudocereals compared to commercial cereals such as barley (Teran, 2017; Cela et al, 2022).
Sensory properties
The use of malted pseudocereals can confer different sensory characteristics of barley malt since they allow to achieve a brighter or darker color, good rates of foam, greater body, and add complexity to the flavor and aroma; moreover, pseudocereals allow to obtain gluten-free beers (Suarez, 2013). These effects are detailed below.
Color
Color is one of the most interesting attributes of beer that attracts the consumer's attention. Biochemizing there are several compounds responsible for colors such as mela noidins, caramelization and pyrolysis products, oxidized polyphenols, riboflavin, carotenoids, anthocyanins, and chlorophyll; each one possesses a color spectrum ranging from yellow to amber. It is generated by the Maillard reaction (non-enzymatic browning) that occurs during the heat treatment of malting (Suarez, 2013).
For example, Yao et al. (2015) observed that the use of quinoa malt (50%) directly affected the color parameters of the beer, decreasing its luminosity and giving it a slightly amber-yellow hue. According to the researchers, this is due to the natural pigments presented in quinoa, such as phenolic compounds and betalains, which react during the malting process.
The color of the malts is measured in EBC units, which is the scale to determine their potential styles nowadays. According to this method, the low EBC indices refer to lighter malts, while higher indices are designated to darker malts (Suarez, 2013). In a study by Phiarais et al. (2010), they brewed a beer using 100% buckwheat malt, obtaining a final product with 10.6 EBC color units (amber) due to extensive maceration. The authors decided to adopt an extensive maceration procedure with the enzymes to ensure the complete saccharification of the beer.
Taste
In conventional beers, the taste is influenced by the type of malt (barley or wheat) due to the components such as maltol and isomatol (Bamforth, 2009). Commonly when different grains are used, a bitter taste is obtained. Ale beers are very aromatic, with fruity flavors, and generally have a complex taste on the palate.
Traditionally hops are the ones that impart the thick flavor of beer due to the content of bitter resins and essential oils that it contains, but lately, cereals and fruits are being used to create new flavors. Craft beers have the particularity of use unique ingredients to exalt their flavor. This is the case of Castañeda et al. (2015), who developed a beer based on raw and malted quinoa since the acidity of the last was higher. The mix did not influence the taste of the beer over the first 7 days, however, when adding malted amaranth in more than 50%, the beer presented a bitter flavor (Monteros, 2016).
Finally, it has been reported that brewing beer with up to 50% substitution of barley attachments can produce good flavor stability (Bogdan & Kordialik, 2017).
Foam
The production of foam is one of the most critical factors in the quality of a beer since it transmits the first impression of the product when it is served. The foam is formed mainly by the CO2 dissolved in the liquid and the protein content of the beer (Wallin, 2010).
Its stability depends mainly on the interaction of proteins/polypeptides of malt and acids from hops (Bogdan & Kordialik-Bogacka, 2017). Malt is a good source of foam-promoting proteins and positive-negative foam components, such as b-glucan, melanoidins, polyphenols, and lipids, as they play a precise role in stabilizing long-lasting foam (Combe et al., 2013).
Malted cereals contain proteins that degrade during malting; therefore, the wort contains a high molecular weight of proteins/polypeptides that positively affect the foam production of beer, reflected when the foaming capacity is evaluated when incorporating CO2. In a study by Lopez & Ramírez (2018), they brewed a craft beer based on quinoa malt, obtaining a foaming stability of 288s in their final product, which can be considered acceptable.
Application of APS in craft beers and gluten-free
Beer is the most consumed alcoholic beverage worldwide, highlighting barley in conventional beers. However, thanks to the growing demand for gluten-free (GF) products (Phiarais et al., 2010), many methods have been developed to produce GF beers considering the new raw materials. Among other grains, APS stands out for its excellent nutritional value. The availability of GF beers significantly improves the well-being of celiac or gluten-sensitive people (Hager et al., 2014). Recent studies indicate that the most commonly used APS in brewing are amaranth, and quinoa, accompanied by buckwheat, as substituents for barley (Martínez-Villaluenga et al., 2020).
Methods for producing GF beers can be distinguished mainly by the category of raw materials applied, using cereals naturally GF, APS or adjuncts, and molecular reproduction technologies. That is why to manufacture GF beer in a reproducible way, the product must contain less than 20 mg/kg of gluten (Martínez-Villaluenga et al., 2020). As a result, several authors studied the impact and use of pseudocereals in GF brewing (Table 4).
The use of some cereals and APS allows the production of naturally GF beer, but also differs from conventional beer in quality and sensory attributes. For example, the aroma of GF beer differs from barley beer, which can provide a negative feeling. Although the high carbohydrate concentrations of GF cereals appear to be favorable for brewing, one disadvantage is that most of these grains have a higher gelatinization temperature (Kerpes et al., 2017). Also, another feature is that the malting installation must be adapted, due to the variation in grain size, with corn having the highest weight and amaranth the lowest. Secondly, the parameters of malting differ from those of barley, with a possible need for higher temperatures (millet, rice, sorghum) or lower temperatures (amaranth, quinoa), prolonged germination time (rice, corn), and increased maceration regime (amaranth, quinoa, millet) (Kerpes et al., 2017).
The quality parameters of the beer can vary according to the cereal or pseudocereal used. One of the critical steps in producing beer is heating the wort with hops. At this moment of heating, the Maillard reaction and the associated reactions occur, which will give rise to the color of the beer (Martins et al., 2001). According to Briggs et al. (1981), the color of the beer is related to melanoidin, caramelization compounds, and phenolic compounds in malt. In pseudocereals, it can be affected by the natural pigments present in some grains, such as quinoa which is rich in phenolic compounds and betalains (Abderrahimet al., 2015).
The color and other beer parameters must be subject to the requirements established by Peruvian Technical Standard 213.014:2016, which indicates that the color of light beer must be less than 30 EBC, while dark beer must be above 30 EBC. Thus, some GF beers as buckwheat beer and quinoa-corn beer can be classified as clear beers since they present 10.6 and 18 EBC, respectively (Table 4).
The fermentation of beer is a natural acidification process (Fix, 2000); during the brewing process, it is preferred to obtain a beer with low pH (4.5) to avoid microorganisms attacking (Recalde, 2017). Where the results cited in Table 4 are within the allowed ranges and resemble the reported value. On the other hand, during fermentation, fermentable sugars are transformed into alcohol and CO2, however, a wide variety of other compounds are generated, which can contribute to aroma beer. In conventional barley malt, 57 g/L of total sugars were found, highlighting its maltose content at 37.7 g/L. In comparison, buckwheat malt has low amounts of fermentable sugars except for glucose (Phiarais et al., 2010). Yeasts use only fermentable sugars, such as glucose, fructose, maltose, and maltotriose to produce ethanol. The control of residual fermentable sugars allows us to witness the end of fermentation and possibly dose the addition of sugars for a second fermentation in the bottle. The analysis of fermentable sugars is also a good parameter to determine the fermentative potential of the must and, therefore, the potential alcohol content (Márquez, 2015).
Table 4 Application of APS and other pseudocereals for the production of Gluten-free (GF) beers
| Cereals and pseudocereals | Results | Observations | References |
| Buckwheat malt 100% | The final beer had the following characteristics: • pH of 4.5 • FAN level of 46 mg/L • NRT level of 619 mg/L • Apparent fermentability (ASBC) of 80.1% • Color 10.6 EBC units, due to extensive maceration. • The final alcohol content was 4.53% (v/v). | It was necessary to adopt an extensive mashing procedure and add commercial enzymes to ensure complete saccharification and extract the buckwheat malt. | Phiarais et al. (2010) |
| Quinoa malt (Chenopodium quinoa) Tunkahuan variety and Corn malt (Zea mays) INIAP 111 | The barley-free alcoholic drink resulted: • With an alcohol content of 4.21% • pH 4.56 • With a density of 1.010 g/cm3 • Caloric content of 176.97 Kj/100 ml • About color, the result was a beer with small, suspended particles that affected it while increasing the concentration of quinoa; but still, it corresponded to the American Pilsen type with a minimum color scale of 6 maximum 18 EBC (clear beer) | It was determined that the best ratio of corn malt and quinoa to obtain a beer-type beverage is 60% corn and 40% quinoa. | Recalde (2017) |
| Quinoa malt (Chenopodium quinoa ) and Sorgum malt (Sorghum vulgare) | It turned out a GF beer: • Cloudy amber color • 6% alcohol degree • Ph between 4.2 to 4.5 • IBU Bitter Standard (30) • Turbidity is typical of craft beer and quinoa protein. | This type of GF beers satisfy the need for relaxation innovatively, making them healthier and more competitive. | Bustillo et al. (2018) |
| Organic quinoa (Chenopodium Quinoa) | The resulting beer reached: • Alcoholic Degree of 4.2% and 4.3% • Organoleptically, a very cloudy beer • pH of 4.5 • Acidity of 0.8% m/m, thus, the product will not present problems for its commercialization. | Using pseudocereals with a higher starch content, they can be transformed into fermentable sugars for the production of beverages of this type. | Márquez (2015) |
| Amaranth malt and barley malt | In this experiment, two treatments were carried out, one with germinated amaranth malt and the other without germination, obtaining: • pH 3.5-5 • Density obtained of 1.01 g / cm3 which is like conventional beer • Alcoholic Degree 4.49 º • Total acidity of 0.15% m / m • The beer was darker than the conventional one, with a pleasant smell and a higher foam level. | Including the germinated amaranth in the formulation attributes a higher protein content that provides greater consistency in the quality of the craft beer. | Montenegro (2016) |
| Quinoa malt (Chenopodium quinoa) ILLPA INIA variety and barley malt | The elaboration of this craft beer presented: • Optimal alcoholic degree of 4.4% v/v • Bitterness of 37.67 IBUs • Final PH of 4.44 • I present a red hue complemented with the added caramel malt presenting greater capacity (65%) • Foaming stability (288 s) of said beer. | The best mix to obtain a red color between 24-30 ºEBC units of craft beer is 55% Base Malt, 20% Caramel Malt, and 25% Quinoa Malt. | Lopez & Ramirez, (2018) |
Traditionally, a conventional beer has an alcohol content of 4%, comparing it with beers using APS malt have similar values, for example, Márquez (2015) uses quinoa malt, obtaining 4.2% - 4.3% values, being weak beers (3.5%-4.5%). On the other hand, Montenegro (2016) obtained a degree of 4.9% using amaranth malt, placing it in the range of strong beers (4.8%-5.5%).
However, using high protein APS hinders the hydrolysis of starch and, therefore, the production of alcohol; with this information, Recalde (2017) justifies his results by obtaining a beer with lower alcohol content (1.65%), halfway that increases the proportion of quinoa malt to corn malt.
In general, using cereals and pseudocereals allows the brewing of GF beer. However, because of the different characteristics of grains, the quality parameters and sensory attributes will differ from conventional beer. Therefore, optimizing the parameters for each type of malting is recommended and thus maintaining the values within the requirements established by the Technical Standards of the country where it is marketed.
Economic, social, and cultural impact
Several publications highlight the emergence and growth of the craft beer industry worldwide (Cabras, 2018; Romanini et al., 2016; Duarte Alonso et al., 2020). For example, in the United States, craft breweries increased from 4,803 in 2015 to 8,275 in 2019 (Duarte Alonso et al., 2020). On the other hand, Toro-Gonzalez (2017) indicates opportunities for the craft beer industry to grow in Latin America. Although craft beer is a product for the young public in Peru, it has grown significantly during the last years, from an annual production of 650,000 liters in 2015 to 1 million liters in 2016 (Mamani & Carlos, 2017). This growth could be due to the increase of people opting for less industrialized products with natural characteristics.
Craft beer has components similar to industrial beer (water, malt, yeast hops) but includes a particular additive (attached inputs). The attached additive is included to define the character of the craft beer. These attached inputs can be grains and fruits, which provide flavor and aroma to craft beer, differentiating them from the others (Mamani & Carlos, 2017). Craft brewers can make use of non-traditional ingredients and their unique formulas, "thus developing new styles that are unprecedented" (Duarte Alonso et al., 2020).
Peru has great potential in producing and marketing craft beer, with its own identity, since this country has various raw materials that could be incorporated as adjunct inputs to beer production, such as APS (quinoa, kiwicha, and cañihua). Kordialik‐Bogacka et al., (2018,2019) indicate that using quinoa or kiwicha in brewing could meet the demand of health-conscious consumers and positively affect the composition of the wort for good fermentation. Kordialik-Bogacka et al. (2018) indicate that quinoa is an excellent starchy raw material for brewing, replacing barley malt with up to 30% quinoa, which would offer brewers the opportunity to develop good beers with new sensory characteristics. On the other hand, the low alcohol content and exceptional nutritional properties, such as minerals and amino acids, suggest that the quinoa drink might be suitable for more frequent use (Deželak et al., 2014). De Meo (2011) indicated that APS are grains suitable for producing gluten-free beers, considered fermented and functional beverages.
However, the cost must be overcome for this product to be more widespread and commercialized since craft beer is more expensive than conventional beer. In addition, small craft brewers have the disadvantage of having limited capital, which prevents them from buying raw materials in bulk in larger quantities (Duarte Alonso et al., 2020). An alternative to overcome this problem is that craft breweries operate in metropolitan areas, seeking to serve residents who, on average, may have higher economic incomes (Baginski & Bell, 2011). Furthermore, craft brewers have more flexibility in changing flavors which could be an advantage in the commercial aspect (Woolverton & Parcell, 2008).
Peruvian craft beer is a product made in small batches, unlike traditional beers, no artificial or chemical additives are used, and its process is mostly manual, without the help of machinery, from grinding the malts to bottling. In addition, the brewers use their original recipes, which allow the uniqueness in craft beer's body, aroma and flavor (MINCETUR, 2018).
Craft beer producers are mostly microenterprises and generally face financial difficulties (Gupta et al., 2018). To develop this sector, it is necessary to achieve an early competitive advantage; in the case of Peruvian companies, the already established image of Peruvian cuisine represents an ally and opportunity; but to benefit from such a partnership. The restaurant and artisanal processing industries must be based on the continued excellence of products and services, which could contribute to Peru's destination image and tourism (Duarte Alonso et al., 2020).
Conclusions
For its flavor and nutritional value, beer is the most consumed millenary drink globally and should be considered a food. Unlike industrial beer, craft beer, in addition to water, malt, hops, and yeast, contains a special additive attached that can be grains and/or fruits to provide the flavor and aroma that will differentiate it from others. The brewing process goes through several stages, mainly germination and malting. This is done to improve the composition and digestibility of proteins to take advantage of malted grains as a protein source to formulate food products.
Beers made from Andean pseudocereals (APS) such as quinoa, amaranth, and cañihua, or with partial barley replacement by APS, improve their protein quality, arousing the interest of consumers, especially for their proven health benefits. APS beers are suitable for celiac people (gluten intolerant) or those who prefer a gluten-free diet. The production of craft beers with APS addition becomes economical, culturally, and socially significant in the brewing industry and guides future research on their bioactive compounds and functionality to achieve a better commercial positioning of these ancestral grains.
It has been shown that malting processing improves the techno-functionality of APS, which could be used in conventional industrial protocols for producing alcoholic beverages, promoting the incorporation and consumption of APS in functional foods. At the same time, the explosive growth of the craft beer market and the affiliation of more new breweries are putting pressure on new consumer tastes, creating more exhaustive competition. For this reason, the great versatility of the new formulations allows people to be willing to try the different amounts of nuances, densities, flavors, and aromas, being somewhat different products for the palate.