INTRODUCTION
Dopamine (DA, acontraction of 3,4 - dihydroxyphenethylamine) with C8H11NO2 molecular formula and 153.18 (g/mol) molecular weight, is an organic chemical of Catecholamine and Phenethylamine families. The structure of Dopamine is consist of a Catechol structure with substitution of hydrogen via 2-aminoethyl group(1).
Dopamine acts both as human hormone and a neurotransmitter and apply its effects by binding and activating cell surface receptors(2), meaning that the Dopamine effects accompany by second messenger system(3).
Some evidence represent that midbrain dopamine neurons are heterogeneous in their prediction targets, responses to environmental stimuli, pharmacology, and effects on motivated behavior.(4) Based on public beliefs about the Dopamine central role in causing “wanting” and “pleasure”, detailed studies demonstrated that Dopamine could not simply equated with liking and pleasure(5) but pleasure centers have been identified both with Dopamine system and outside Dopamine system(6).
Dopamine also used as manufactured medication that formly named Intropin, Dopastat, and Revimine(7). As drug it is mostly used in hilling of severe low blood pressure in infants(8), slow heart rate by both increasing sodium excretion by the kidney, increasing in urine output and increasing force on in low doses(9,10), and also it has main role in several significant medical conditions such as Parkinson's disease, an age- related movement disorder, in which the main symptoms accompany by the loss of dopamine-secreting cells in the substantia nigra(11), schizophrenia(12) and addiction(13,14,15).
Since the participation of serotonin and norepinephrine in pain spinal reduction is well known, Dopamine has a critical role in pain processing in different levels of central nervous system, including spinal cord, periaqueductal gray, thalamus, basal ganglia, and cingulate cortex(16). Other advantages of dopamine are in regulation of plasma glucose levels, endocrine function, and neuroimmune regulation(17).
Serotonin (5-hydroxytryptamine (5-HT)) with C10H12N2O molecular formula and 176.215 g/mol molecular mass, the monoamine neurotransmitter, has effective role in contributor to feelings of well-being and happiness although its actual biological function is complicated(18).
Biochemically, serotonin synthesized from amino acid tryptophan throughh the addition of a hydroxyl group and removal of the carboxyl group(19,20). In human body the most of total serotonin is located in enterochromaffin cells in the GI tract (gastrointestinal tract) and works as intestinal movements regulation(21,22) and it is also secreted luminally and basolaterally and leads to increased serotonin uptake by circulating platelets and activation after stimulation and eventually an increase in myenteric neurons and gastrointestinal motility Irritability(23).
Furthermore, serotonin also stored in blood platelets and released during vasoconstriction, which acts as an agonist to other platelets(24). Serotonin has numerous effects including, effects on mood, anxiety, sleep, appetite, temperature, eating behavior, sexual behavior, movements and gastrointestinal motility(25).
Drugs that selectively target specific serotonin receptor subtypes therapeutically used for anti-depression effects and called selective serotonin re-uptake inhibitors, their functions are dependent on serotonin availability in the synapse(26).
There are many electrochemical techniques for the detection of biomolecules however the expansion of sentiment platform with high sensitivity and selectivity is kinda challengeable. Nanomaterials-based sensor platforms are fascinating for researchers for their capability to carry out the electrochemical analysis of neurotransmitters and they have been extensively reported on for their sensitive detection of some biochemical molecules such as epinephrine, dopamine, serotonin, glutamate, acetylcholine, nitric oxide, and purines(27).
Since dopamine andserotonin are important neurotransmitters that have dealings in the brain, dopamine could easily detected with electrochemical sensors but the detection of serotonin has more difficulty because the observation of serotonin oxide can reduce sensitivity and by using carbon nanotubes (CNTs) treatments , the sensitivity of system will be increase and promote electron transfer, and reduce sedimentation(28).
In addition, carbon-based nanomaterials such as single- walled carbon nanotubes (SWCNTs), multi-walled carbon nanotube (MWCTs) and graphene due to their biocompatibility for instant, non-toxic properties, and low costs can be used in such biosensing and electrochemical sensing applications(29,30).
Among the carbon-based nanomaterials, CNTs widely explored in electrochemical sensors studies due to their fantastic structure, high surface area, rapid electron kinetic, high thermal conductivity, stability and electronic properties(31,32,33).
MATERIALS AND METHODS
The molecular structure, quantum mechanics and Theoretical computations, charges distribution for Serotonin and Dopamine binding to SWCNTs with four different diameters such as 7.0 , 7.5 , 7.7 ,10.0 (nm) calculated using standard GIAO and B3LYP level of theory with 6-31 G(d) basis set with the gaussian 09 program to performed to study chemical and physical properties of nuclei and NMR chemical shift data(34). Gaussian 09 is a computational chemistry software package appropriate to demonstration interaction of electrons in atoms and molecules. Molecular orbital energies, bond energies, molecular geometries and energies, and vibrational frequencies are the other features can be found in this program. The chemical shift refers to aspect dependent on the secondary magnetic field created by the induced motions of the electrons surrounding nucleus(35). The NMR analysis have been fulfilled with four parameters including, magnetic isotropic (σiso) and magnetic anisotropic (σaniso) shielding,σ11,σ22,σ33 as shown in the following result for its fundamental importance in chemistry and biochemistry studies ,in which the σ defined as magnetic shielding tensor(ppm) and (η) defined as shielding asymmetry .σ also refers to the differential resonance shift due to the induced motion od the electrons(36). Kinetic and thermodynamic investigations, geometry optimization, Monte Carlo and vibrational analysis done by using HyperChem 8.0.8 software which is a sophisticated molecular modeling environment that is familiar for its quality, Flexibility, and ease of use. 3D embodiment with quantum calculations, molecular mechanics, and dynamics are other capability of this tool(31). For Monte Carlo in molecular mechanic method we optimized Potential, kinetic and total energy with 10 steps in 310 k degree (the most stable and important temperature) and 298 k degree (environment temperature) and for Semi empirical the Am1 method with all parameters (Total Energy, Binding Energy, Isolated Atomic Energy, Electronic Energy, Core-Core Interaction and Heat of Formation) is the best vibration analysis of molecules using a quantum mechanical approach that was obtained Semi empirical(36).
RESULTS
In this work we have studied theoretically the magnetic and kinetic properties of atoms and vibrational analysis to specified chemical and physical properties of atomic nuclei. According to NMR quantum mechanics-based ,our results were summarized to calculation of magnetic shielding tensor (σ, ppm),shielding asymmetry(η),magnetic shielding anisotropy (σaniso,ppm),isotropic shielding value (σiso) ,the skew of a tensor (Κ), chemical shift anisotropy (Δσ) and chemical shift (δ) . these results are shown in figures 1,2,3 and are listed below in Tables 1 2,3,4,5.
Tabla 1 Calculated optimized energy parameter (kcal/mol) of Dopamine binding to nanotubes in temperature of 298 K and 310 K by Monte Carlo method(opls).
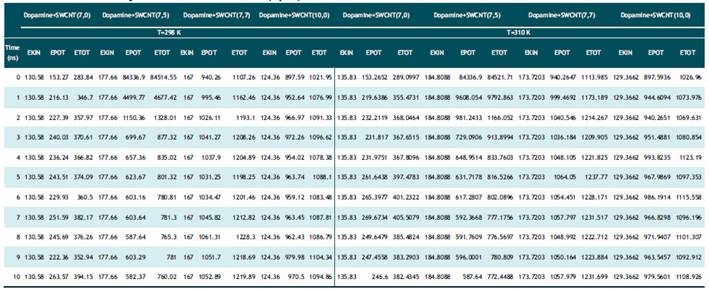
Tabla 2 Calculated optimized energy parameter (kcal/mol) of Serotonin binding to nanotubes in temperature of 298 K and 310 K by Monte Carlo method (opls).
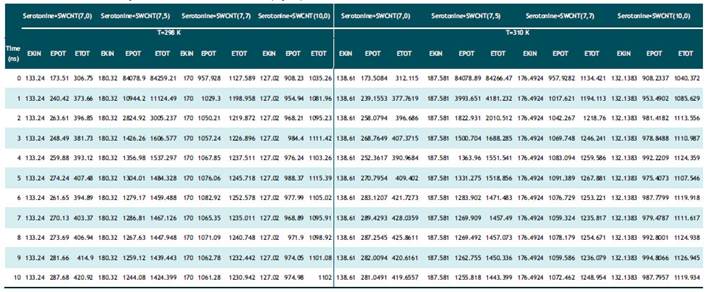
Tabla 3 Optimized parameters of total energy, binding energy, isolated atomic energy, electronic energy, core-core interaction and heat of formation(cal/mol) for Dopamine + SWCNTs and Serotonin + SWCNTs by AM1 calculations.
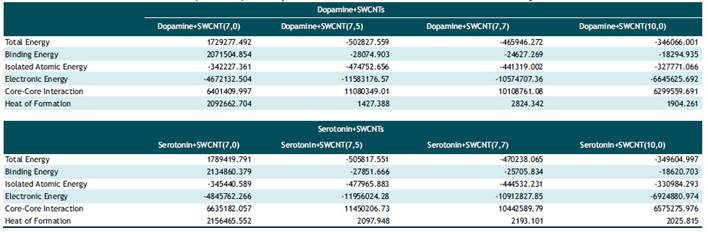
Tabla 4 Comparison of NMR chemical shielding tensors data calculated by B3LYP models with 6-31G(d) basis set for N, O atoms in Dopamine with 7.0,7.5,7.7,10.0 (nm)diameters SWCNT.
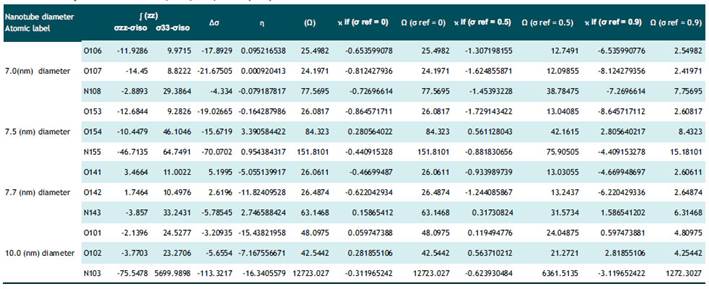
Tabla 5 Comparison of NMR chemical shielding tensors data calculated by B3LYP models with 6-31G(d) basis set for N, O atoms in Serotonin with 7.0,7.5,7.7,10.0 (nm) diameters SWCNT.
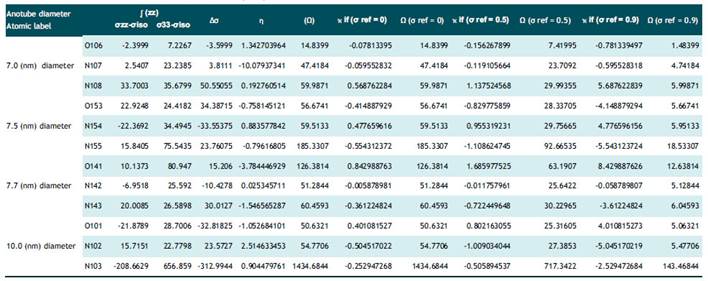
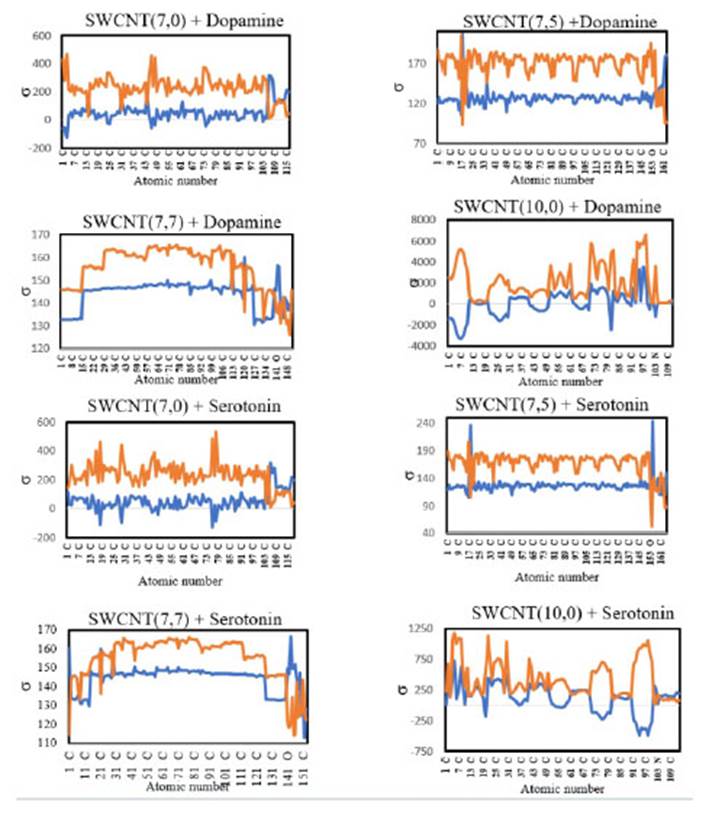
Figura 1 NMR parameters of isotropic and anisotropic shielding for SWCNT+Dopamine and SWCNT+ Serotonin in gas phases at the B3LYP/6-31G (blue line is related to σiso and red line is related to σaniso)
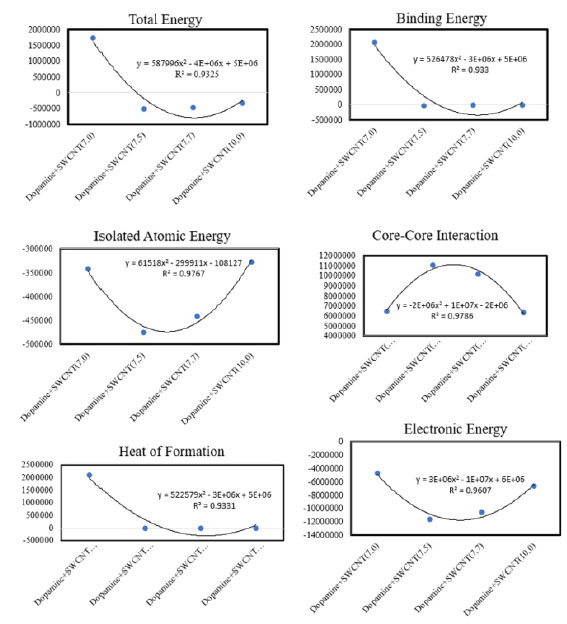
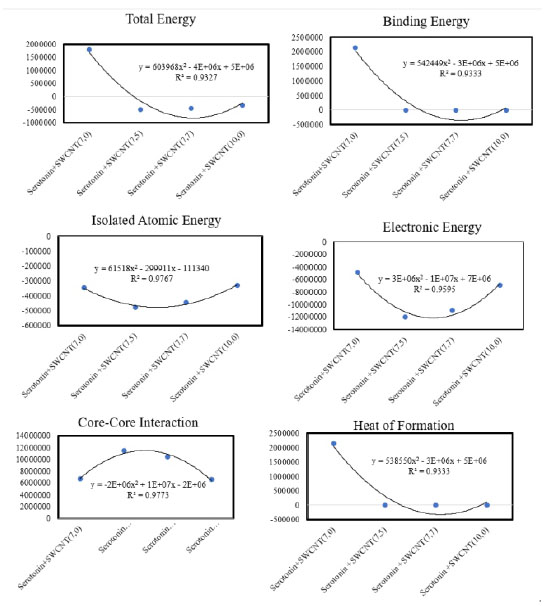
Figura 2 Optimized parameters of total energy, binding energy, isolated atomic energy, electronic core-core interaction and heat of formation(cal/mol) for Dopamine + SWCNTs by AM1 calculations.energy
DISCUSSION
Based on table1-2 in Dopamine-SWCNT combination at 298ºK the optimized potential energy is in 10th step 263.5 (kcal/mol) and at body temperature (310K) is 246.6 kcal/mol in Dopamine+SWCNT (7.0) and in Serotonin-SWCNT combination is respectively 287.6 kcal/mol and 281.04 kcal/mol. As it shown in the table 1 and 2, the stability of potential energy in 10th step in the drug-SWCNT combination except SWCNT(7.0) become minimized by increasing compound diameter at 310ºk . The optimum Kinetic energy for Dopamine compound in both 298 and 310 ºk belongs to Dopamine+SWCNT10.0 complex and respectively are equal to 124.3, 129.3 kcal/mol. Serotonin +SWCNT10.0 nm also has the same behavior and the kinetic energy parameter at 298 ºk is 127.0 kcal/mol and at 310K is 132.1 kcal/mol. What is revealed from table5, the minimum total energy belongs to Dopamine +SWCNT 7.5 complex (-502827.559 cal/mol). Binding energy is another parameter checked in semi empirical methods that is much more in Dopamine+SWCNT 7.0 as a result of more stable structure (2071504.8 cal/mol) and the third parameter investigated is Core-Core interaction which in Dopamine+SWCNT 7.5 is the strongest one (11080349.01 cal/mol. Connection of Dopamine with SWCNT 7.5 release minimum amount of heat of formation (1427.3 cal/mol) and connection with SWCNT 7.0 frees up 2092662.7 cal/mol.
Semi empirical results of Dopamine gathered in Fig 2 observed R2=0.93 for total and binding energy and heat of formation, R2=0.96 for electronic energy, R2=0.97 for isolating energy and Core-Core interaction.For Serotonin based on table 3, Serotonin+SWCNT 7.5 has minimum total and binding energy (-505817.551 cal/mol) (-27851.666 cal/mol) and also has the maximum value of Core-Core interaction (11450206.733 cal/mol). Serotonin+SWCNT 7.0 Complex has the maximum value of heat of formation (2156465.552 cal/mol). As it comes out from Fig3, results obtained R2=0.93 for total and binding energy and also for heat of formation, R2=0.97 for isolating atomic energy and Core-Core interaction and for electronic energy R2 =0.95.
The second level of the results specified to NMR chemical shielding tensors data for the complex of Dopamine and Serotonin+SWCNT have been recorded in table 4-5. According to table 4, Δσ for all Nitrogen atoms in the dopamine-SWCNT complex has the negative value and the minimum value belongs to N103 in Dopamine+ SWCNT10.0 (-113.32) and among oxygen atoms,O141 has the maximum amount in the Dopamine + SWCNT 7.7 complex (5.19) .N103 in Dopamine complex also has the minimum value of Etta (η) (-16.34) although it has maximum amount of Ω (12723.027 ). The maximum level of η (3.39) belongs to O154 in this complex by 7.5 nm diameter. As it comes from table10, the maximum value of Δσ belongs to N108 in the Serotonin-SWCNT complex with 7.0 nm diameter nanotube (50.55), maximum amount of η belongs to N102 (in 10.0 nm diameter nanotube) about 2.51. The nitrogen with atomic label 103 in this combination has both the lowest value of chemical shift anisotropy (Δσ) about -312.9 and the most and positive value of Ω (1434.6).
In this work we study theoretically the structure features of Dopamine and Serotonin in the complex of single-walled carbon nanotube as a neurotransmitter. molecular mechanic and quantum mechanics data such as potential ,total and kinetic energy , geometrical optimization and vibrational analysis have been investigated by using Mont Carlo and Semi empirical methods .Chemical shift anisotropy asymmetry (η), isotropy (σ iso), anisotropy (σ aniso), Δσ, skew of a tensor (Κ) and chemical shift tensor (δ) were calculated based on theoretical data obtained from NMR and BL3Y/6-31G(d) levels of theory. It is figured out in Mont Carlo method our two specific drug and its nanotube with small diameter are the most stable one than the others. The larger diameter leads the combination stability into lower value