INTRODUCTION
Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) are characterized by the separation of the dermoepidermal junctions with subsequent necrosis and epidermal detachment of extensive mucocutaneous areas 1. These two entities are variants of the same process that differ in the extent of affected body surface. They occur due to delayed hypersensitivity reaction 2,3. We describe a case of TEN that occurred after concomitant treatment with valproate, lamotrigine, and phenobarbital. Based on causality assessment, this case was “very likely” caused by lamotrigine and phenobarbital. Although, it is possible that the interaction between the three drugs has increased the risk of TEN 1,2,3. The aim of this report is to present this rare but potentially fatal adverse reaction; and to emphasize the importance of its early recognition, the suspension of the causative agent, and to install an early and aggressive supportive treatment that is critical for survival.
CASE PRESENTATION
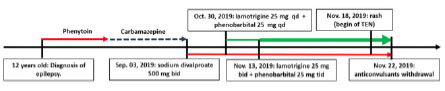
The Sixteen-year-old girl, who presented epilepsy from 12 years old, treated with phenytoin, and afterwards carbamazepine. Because of persistent seizures, on September 03, 2019 she was turned to sodium divalproate 500 mg 1 tablet every 12 hours PO. On October 30, 2019 lamotrigine 50 mg ½ tablet every 24 hours and phenobarbital 100 mg ¼ de tablet every 24 hours PO were added. Two weeks afterwards, doses were increased: lamotrigine 50 mg ½ tablet every 12 hours and phenobarbital 100 mg ¼ tablet every 8 hours. On November 18, 2019 she began with an erythematous itchy rash on face, then it spread to the entire surface area. On November 22, she was admitted in a private hospital, where betamethasone 7mg every 24 hours EV and cetirizine/pseudoephedrine 10/120 mg every 24 hours PO was administered, and anticonvulsants were withdrawn. In the day following, mucocutaneus lesions got worse. Then this patient was transferred to our hospital, being admitted on November 28, 2019. Figure 1 shows a time line of the drugs taken by the patient.
Physical Exam. Patient in poor general health, puffy face with expression of severe pain; BP 90/60 mmHg., RR 26 bpm., HR 134 bpm., temperature: 37.8 °C, oxygen saturation 95% on room air. Skin and mucous membranes: bilateral conjunctival injection with eyelid desquamation. Dry mucous membranes, erythematous fissured and bleeding lips; limitation of mouth opening due to pain, erythematous oropharynx. Erythematous rash morbiliforme involving over than 90% of body surface area with areas of sloughing of lips, nostrils, (Figure 2) and genitals. Soft pitting edema of the face, trunk, abdomen, and extremities. Respiratory, cardiovascular and abdominal system: without alterations, except for tachypnea and tachycardia. Neurological: sleepy, but oriented.
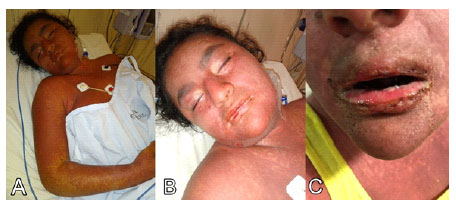
Figure 2 Day of admission (November 28, 2019). Morbilliform and erythematous rash involving more than 90% of the body surface with sloughing of the lips and nostrils (A-C). Bloated face; dry mucous membranes, erythematous, fissured and bleeding lips; and limitation of mouth opening (A-C).
Diagnosis. TEN.
Treatment. Physiological saline 150 ml/hour, oxygen therapy, dexamethasone 4 mg every 8 hours EV, chlorphenamine 10 mg every 8 hours EV, ranitidine 50 mg every 8 hours, fentanyl 50 μg/hour.
Evolution. Two days after admission, the patient presented fever, hypotension, and an episode of generalized tonic-clonic seizures. Mucocutaneous sloughing and multiple blisters with positive Nikolsky and Asboe-Hansen sign were evidenced. (Figure 3), and methylprednisolone 1 gram every 24 hours IV, piperacillin/tazobactam 4.5 grams every 6 hours IV, vancomycin 1 gram every 12 hours IV, and norepinephrine 0.1 μg/kg/min were started. Skin detachment was maximum on the third day after admission (Figure 4). On the fifth day after admission, improvement of the lesions was evidenced (Figure 5), she tolerated withdrawal of vasopressor and oxygen. The result of the skin biopsy showed lichenoid-interface dermatitis, apoptotic keratinocytes, subepidermal blisters, spongiosis, basal vacuolation and perifollicular lymphocytic infiltrate, compatible with TEN. Based on the causal assessment of TEN, 500 mg valproate was reinitiated every 12 hours orally. The patient evolved favorably and was discharged ten days after admission.
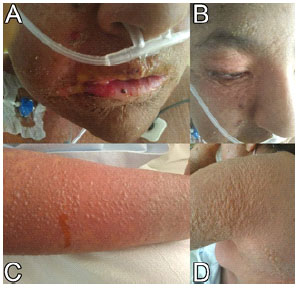
Figure 3 Two days after admission (November 30, 2019). There is dry oral muco-sa, erythematous lips, fissured with limitation of the mouth opening (A). Fine peeling of skin and mucous membranes, multiple blisters (A-B). Sloughing and formation of blisters in extremities and body (C-D).
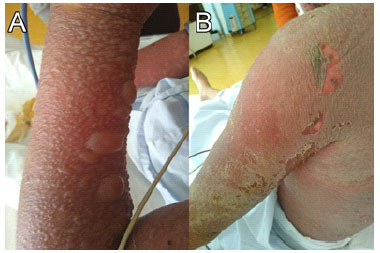
Figure 4 Three days after admission (December 01, 2019). Large mucocutaneous detachment with formation of vesicles and bullae (A-B). Pressure on these blisters causes shedding of the skin (positive Nikolsky and Asboe-Hansen signs) (B).
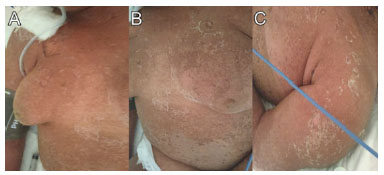
Figure 5 Fifth day after admission (December 03, 2019). Notable improvement of mucocutaneous lesions with fine desquamation and re-epithelialization (A-C).
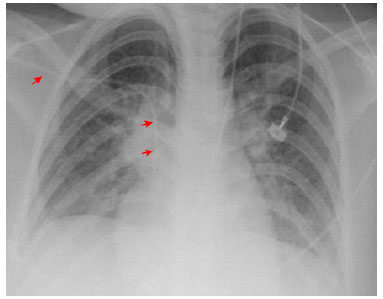
Routine tests were normal. The chest radiograph showed a discrete bilateral interstitial-alveolar infiltrate with a predominance of bases (Figure 6).
DISCUSSION
SJS/TEN appears mainly due to an idiosyncratic reaction to drugs (> 80% of cases). Anticonvulsants account for 50% of cases; non-steroidal anti-inflammatory drugs 33%; antibiotics 34-80% (sulfa, beta-lactams and quinolones); antiretrovirals (especially nevirapine) and allopurinol are also important causes 1,2,3,4,5.
The latency period between initial exposure and drug reaction is 4 days to 4 weeks 4,5. The risk of SJS/TEN is higher after recent initiation of the drug and appears to be limited to the first 8 weeks of treatment 6. Drugs used longer are unlikely causes of SJS/TEN 5,6. Other etiologies of SJS/TEN are infections (her-pes simplex virus, Mycoplasma pneumoniae), and vaccines 1,2,3,4.
Up to 20-33% of SJS/TEN cases remain idiopathic 1,2,3,4,5. SJS/TEN typically appears 3 weeks after the start of the drug, with prodrome of fever and catarrhal symptoms, that begin 1-3 days after the appearance of mucocutaneous lesions. Photophobia, conjunctival itching or pain, and odynophagia are early symptoms of mucosal involvement 1,4. Fever, mucositis, disproportionate skin hypersensitivity, and the appearance of blisters should alert early to the possibility of SJS/TEN 1,2,4. Our patient presented these last four characteristics. Mucosal involvement occurs in 90% of SJS / TEN cases, and includes conjunctival, nasal, oropharyngeal, tracheobronchial, esophageal, vulvovaginal, genitourinary, and anorectal lesions 1,2,3,4,5. By consensus, if the epidermal detachment is less than 10%, it is considered SJS; between 10-30%, SJS/TEN overlap; and TEN, when it is greater than 30% 3,4. Dermal lesions start on the face and chest, then spread symmetrically to other areas. Generally, the scalp, palms, and soles are rarely affected. As the disease progresses, vesicles and blisters appear, the skin sloughs, and Nikolsky and Asboe-Hansen signs appear. The acute phase of SJS/TEN lasts 8-12 days. Re-epithelialization begins after several days and usually lasts 2-4 weeks 1,2,4.
There are no universally accepted diagnostic criteria for SJS / TEN. Histopathology is neither diagnostic nor specific, but it serves to exclude other entities that can mimic SJS/TEN 1,4. It is possible to diagnose SJS / TEN based on the following clinical characteristics: 1) previous exposure to drugs (1-4 weeks, mean 14 days); re-exposure can result in onset of symptoms within 48 hours; 2) prodrome and acute-onset febrile illness; 3) rapidly progressing painful rash; 4) erythematous macules, target lesions, or diffuse erythema progressing to vesicles and bullae; 5) Nikolsky and / or Asboe-Hansen signs; 6) mucositis; 7) epidermal necrosis and sloughing 1,2,3,4.
Identification of the offending drug is essential for two reasons: 1) early dis-continuation improves prognosis; and 2) to prevent re-exposure. The evaluation of causality is based on a detailed history, clinical judgment, and knowledge of the drugs most frequently associated with SJS/TEN. The ALDEN algorithm is a tool to evaluate the causality of SJS/TEN, particularly useful in cases with exposure to various drugs, such as our patient. This score categorizes as “very probable” (≥6), “probable” (4-5), “possible” (2,3), “unlikely” (0-1), and “very unlikely” (≤0) (5). Among the drugs with probable or definitive causation, antiepileptics are the most frequent (53%) of SJS/TEN 6,7,8,9,10,11.
According to RegiSCAR, lamotrigine and phenobarbital are “strongly associated” with SJS/TEN, although with different frequencies (lamotrigine 2.5-10% and phenobarbital 2-8%) 11,12. There are some reported cases of SJS associated with valproate; but, TEN secondary to valproate monotherapy appears to be an extremely rare event 4,7,8,9,10,11,12. The causality evaluation identified lamotrigine and phenobarbital as “very probable” causes (ALDEN score 6) and valproate as “very unlikely” cause (ALDEN score 0) of TEN, and allowed valproate to be reinitiated early and definitively discontinued lamotrigine and phenobarbital.
The SCORTEN (SCORe of Toxic Epidermal Necrosis) score estimates the prognosis and mortality of a patient with TEN during the first 5 days of hospitalization. Consequently, it allows deciding the best management scenario (intensive care, burn unit, or common room) and evaluating the efficacy of therapeutic interventions 4,13,14,15,16,17. Our patient had a SCORTEN of 2, which corresponds to a low risk of mortality (12.1%). Mortality for SJS is 10-40% and up to 50-70% for TEN 1,2,3. Although the SCORTEN score is a prognostic scale developed to be used in adults, SCORTEN has been evaluated and validated for its application in pediatric patients 18,19 and its use is recommended for the management of pediatric TEN 20.
The available evidence on the optimal treatment of SJS / TEN is scarce 16,17. Corticosteroid treatment can give good results, and some authors recommend cyclosporine A 1,4,7,8,9,10,11,16,17. In this case, we administered corticosteroids, anti-histamines, and support measures (isolation, oxygen therapy, vasopressors, hydration, and analgesia), moisturizing and abrasive creams, and bandages.
CONCLUSIONS
This case is interesting for some reasons: the infrequency of this type of serious reaction to anticonvulsants; the favorable final result in spite of the severity of the clinical presentation; and because it highlights the importance of installing an early and aggressive support treatment that can save the patient's life