Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista Peruana de Ginecología y Obstetricia
versión On-line ISSN 2304-5132
Rev. peru. ginecol. obstet. vol.64 no.4 Lima oct./dic. 2018
http://dx.doi.org/10.31403/rpgo.v64i2130
SIMPOSIO CIRUGÍA FETAL EN AMÉRICA LATINA
Fetoscopic laser surgery for twin to twin transfusion syndrome
Cirugía láser fetoscópica en el síndrome de transfusión feto fetal
Savino Gil Pugliese1,2 ORCID: 0000-0003-2725-971X
1 Fetal Medicine Team, Instituto de Maternidad y Ginecología Nuestra Señora de las Mercedes, Tucumán, Argentina
2 Fetal Medicine Team, Hospital Privado Universitario de Córdoba, Córdoba, Argentina
ABSTRACT
Twin to twin transfusion syndrome (TTTS) is a complication of circa 10% of all monochorionic pregnancies (MC). A predominantly unidirectional, net intertwin blood flow through placental vascular anastomoses triggers a variety of complex renal and cardiovascular disturbances in both fetuses. Left untreated, severe TTTS leads to a mortality rate above 80% and 15-50% morbidity in the survivors. Fetoscopic laser coagulation (FLC) of the placental anastomoses is the first line treatment for severe cases before 27 weeks and it has been shown to improve perinatal outcomes. The risk of preterm premature rupture of membranes (PPROM) and prematurity associated with FLC has to be balanced with the risk of adopting expectant management in every particular case. Since there is no accurate way to predict the evolution of the disease, and no effective method to prevent post-procedure PPROM has been described, the indication of FLC is as challenging as the procedure itself.
Key words: Fetal therapies, Feto-fetal transfusion, Fetoscopy, Laser coagulation, Twin pregnancy.
RESUMEN
El síndrome de transfusión de gemelo a gemelo (TTTS) complica alrededor del 10% de todos los embarazos monocoriónicos (MC). Un flujo sanguíneo intergemelar neto, predominantemente unidireccional, a través de anastomosis vasculares placentarias, desencadena una variedad de alteraciones renales y cardiovasculares complejas en ambos fetos. Sin tratamiento, el TTTS grave conduce a una mortalidad superior al 80% y una morbilidad del 15 a 50% en los sobrevivientes. La coagulación láser fetoscópica (CLF) de las anastomosis placentarias es el tratamiento de elección para casos graves antes de las 27 semanas y ha demostrado mejorar los resultados perinatales. El riesgo de rotura prematura de membranas (RPM) y de prematuridad asociada a CLF se debe sopesar con el riesgo de la conducta expectante en cada caso particular. Dado que no hay una manera precisa de predecir la evolución de la enfermedad, y no se ha descrito aun ningún método eficaz para prevenir la RPM posterior al procedimiento, la indicación de la CLF es tan desafiante como el procedimiento en sí mismo.
Palabras clave. Embarazo gemelar, Transfusión feto-fetal, Coagulación con láser, Terapias fetales.
Introduction
Monochorionic (MC) twin pregnancy occurs in 1 of every 250 pregnancies and represents 20% of all twin pregnancies(1). Perinatal mortality and morbidity are significantly higher in MC than in dichorionic twin pregnancies since the circulation of both fetuses is interconnected through vascular anastomoses in the common placenta. Therefore, the wellbeing of each fetus is both cause and consequence of that of the other(2,3). In the unfortunate event of the demise of one of the twins, there is a significant risk of death or handicap in the co-twin due to acute blood loss through the placental anastomoses towards the death fetus(4,5).
Establishing chorionicity in every twin pregnancy during early gestation or at the time of the 11 to 13 weeks scan is crucial. A close follow up every fortnight from 16 weeks onwards to detect early signs of complications is needed for MC twins(6).
One of the most common complications in MC twins is twin to twin transfusion syndrome (TTTS), a complex condition in which an unbalanced blood flow exchange through placental anastomoses causes hypovolemia in one twin (donor) and hypervolemia in the other (recipient). This triggers a cascade of hormone-mediated renal and cardiovascular disturbances in both twins. Left untreated, severe TTTS leads to 80-100% perinatal mortality and 15-50% neurological morbidity in the survivors(7).
Although vascular anastomoses are just the anatomical component of a complex condition, its elimination via fetoscopic laser ablation can resolve de syndrome and significantly improve survival(8). Considering the risk of premature rupture of membranes and preterm delivery added by an invasive procedure, the dilemma relies on the decision of the need and timing for the intervention.
Vascular anastomoses
In MC twin pregnancies, where both cords insert and branch over a common placenta, 3 types of anastomosis can connect the circulation of both fetuses: 1) arterio-arterial (AA), 2) veno-venous (VV), and 3) arterio-venous (AV) anastomoses.
AA and VV anastomoses are superficial, presenting on the placental surface, where macroscopic vessels from the two cords can fuse in a true endto-end connection. On the contrary, AV anastomosis are deep, presenting at capillary level, via a functional anastomosis in a shared cotyledon, arterially perfused by one of the twins, but drained into the venous circulation of the other. AV anastomosis have a constant unidirectional flow, whereas superficial anastomoses (AA or VV) can allow flow in both directions depending on the hemodynamic interaction between both circulations. In particular, AA anastomoses may act as protective compensatory shunts preventing the development of TTTS(9).
Postnatal placental studies confirm that almost all (95%) MC pregnancies have vascular interconnections at placental level. However, only 10 to 20% of monochorionic pregnancies will develop TTTS due to a predominantly unidirectional net blood flow from one twin to the other. This seems to be caused by the presence of at least one AV anastomosis (shared cotyledon) and the absence or scarcity of compensatory AA anastomoses. The role of VV anastomoses is controversial; they are rarer and there is evidence suggesting that its presence could contribute to the development of TTTS, especially in the absence of AA anastomosis(9,10).
Diagnosis of TTTS
TTTS is diagnosed by ultrasound by the presence of polyhydramnios in one twin (DVP >8 cm before 20 weeks and >10 after 20 weeks) and oligohydramnios in the other (DVP <2 cm).
A new cut-off of a DVP >6 cm to define polyhydramnios in the recipient before 18 weeks, has recently been proposed to account for the gestational age dependent difference of the 90th centile in the normal DVP distribution(11).
Concomitant selective fetal growth restriction (sFGR)
Placental anastomoses are not the only factor complicating MC pregnancies. The distribution of the placental mass is usually unequal in MC twins. When this discrepancy is excessive and one of the twins has a very small placental mass, its growth can be compromised.
sFGR is diagnosed when one of the fetuses has an abdominal circumference <5th centile before 22 weeks or an estimated fetal weight (EFW) <5th centile after 22 weeks together with a discrepancy in size with the cotwin >25%(1,12). This complication has its own classification and management depending on the characteristics of the umbilical artery Doppler of the restricted fetus(12). TTTS is superimposed in around 70% of MC twin pregnancies affected with sFGR(13).
Natural history of TTTS
The donor twin suffers from hypovolemia and oliguria with progressive decrease in the amount of amniotic fluid, leading to anhydramnios and a persistent empty bladder. In the most severe cases there is absent end diastolic flow in the umbilical artery. On the contrary, the recipient twin is hypervolemic and polyuric presenting a large polyhydramnios. Volume overload may cause absent or reversed wave in the ductus venosus, pulsatile flow in the umbilical vein, tricuspid regurgitation and fetal hydrops in the most severe cases(7). Hypertrophic cardiomyopathy suggesting systemic hypertension can also be seen in the recipient leading to right ventricular outflow tract obstruction (RVOTO) in 3% of the cases(14,15).
All these cardiovascular and renal disturbances described for both twins cannot be explained only by a blood volume depletion or overload. There is evidence to conclude that, in response to hypovolemia, activation of the renin-angiotensin system (RAS) in the donor and the placenta, aiming to increase vascular resistance, can have a negative paradoxical effect in both twins(16). Also, upregulation of endothelin and vasoactive peptides may be involved(17).
The course of the disease in severe cases can lead to intrauterine demise of either twin. Consequently, exsanguination of the surviving cotwin into the fetoplacental territory of the death fetus can cause double fetal demise in up to 50% of the cases(18). In those surviving after the demise of a co-twin, abnormal cranial imaging is found in up to 35% of the cases(19,20).
Evaluation of severity
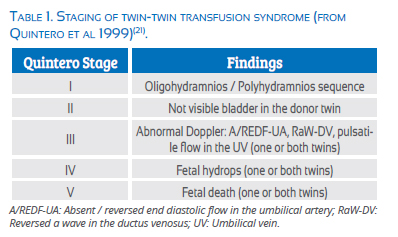
Some of the sonographic findings described have been used by Quintero et al in 1999 to stablish a staging system for TTTS (QS) [Table 1](21). This widespread classification is useful to describe, to some extent, the severity of the condition at the time of the examination, but its ability to predict the evolution of any particular case is limited. Some cases will progress, some will remain unchanged during the whole pregnancy and some others will even regress(22). So far, no effective method to predict evolution has been described.
Although important prognostic factors such as specific cardiac findings in recipients, cervical length, EFW discrepancy and gestational age have been introduced in an attempt to produce new staging systems, QS in its simplicity remains broadly used(23,24). This is probably because to some extent, in daily practice, MC twin pregnancies are being followed up in institutions were FLC is not available, therefore their duty is to differentiate mild from severe cases, or in other words, those cases needing expectant management from those needing urgent referral for active management and QS can do that.
Management
Mild cases (QS I) will be followed expectantly. Active management is reserved for severe cases (QS II-IV) where surgical treatment or elective delivery by caesarean section will be needed depending on gestational age.
Fetoscopic laser coagulation (FLC) of the placental anastomoses is the first line treatment between 16 and 27 weeks. It eliminates the etiological cause of TTTS resulting in double survival in 70% of the cases and a least one fetus surviving in 85% of the cases(25).
Performing any invasive procedure before 16 weeks carries an increased risk for adverse pregnancy outcomes. Although early severe cases are rare, FLC before 16 weeks should be considered specially for QS IV or when imminent fetal demise is suspected(26).
The upper limit of 27 weeks is related to the technical difficulties found during fetoscopy. Visibility and space for maneuvering with the fetoscope are reduced due to bigger fetal size. How far this limit can be pushed is debatable. Intrauterine treatment can significantly reduce neonatal morbidity and mortality by decreasing prematurity and allowing time for the fetuses to recover in-utero from the cardiac and renal insult. In general terms, if severe TTTS is diagnosed after 27-28 weeks, elective caesarean delivery after lung maturation may be preferred for QS III-IV weeks. Amnioreductions should be considered for QS II with elective caesarean section in case of progression(26,27).
Active management in mild cases
Short cervix
Considering that polyhydramnios is the first manifestation of the syndrome, cervical length becomes a very important prognostic factor for adverse outcome related to miscarriage, preterm delivery and prematurity in twin pregnancies affected with TTTS. Cervical cerclage for CL <15 mm, immediately after FLC with amniodrainage may improve gestational age at delivery and survival in severe cases(28). Therefore, the same management can be considered for QS I cases with a CL <15 mm, especially when they present early in gestation. FLC would eliminate the cause of the polyhydramnios and avoid repeated amniodrainage.
In cases of short cervix presenting after 28 weeks, serial amniodrainage could prolong the pregnancy and elective delivery would be indicated in case of progression of the syndrome.
Reversal of TTTS
A reversal of TTTS is a rare situation. Ultrasonographic findings suggest an inversion of the phenotype, with the donor becoming recipient and vice versa. Its diagnosis is challenging, and prognosis is very poor with double demise occurring before the criteria for TTTS is fulfilled. It is an indication for immediate active management according to gestational age(29).
Selective fetal growth restriction (sFGR)
A growth restricted MC twin has a higher risk for intrauterine demise (IUD) and therefore the risk of demise or handicap is increased for the cotwin. Mild TTTS in the context of sFGR has high incidence of adverse outcome and it may be considered an indication for active management (25,30).
Fetoscopic laser coagulation technique
FLC is a minimally invasive procedure performed usually under local or regional anesthesia. Using Seldinger technique, a 10 fr (3 mm) vascular introducer is placed into the amniotic cavity of the recipient through the maternal abdomen. A thorough ultrasonographic examination is needed to decide the best position for the trocar. While the recipient can move freely in the polyhydramnios cavity, the donor is pushed against the uterine wall and the placenta, trapped by its own amniotic sac and the intertwin membrane (stuck twin). The exact location and boundaries of the placenta and the stuck twin should be defined. Both cord insertions must be identified to estimate the vascular equator, were the anastomosis are expected to be present. Velamentous cord insertion is not uncommon specially when TTTS and sFGR coexist(31).
The trocar has to be placed perpendicular to the long axis of the stuck twin and away from it. Perforating the intertwin membrane would cause an accidental septostomy allowing transfer of the recipient´s amniotic fluid into the donor´s sac, adding technical difficulties to the procedure and risk of pseudo-amniotic bands syndrome(32). Ideally, the introduction of the trocar should also be away from the placental edge, aiming perpendicularly to the vascular equator. These basic principles are true regardless of placental position but entering away from the placental edge and perpendicular to the vascular equator in anterior placentas is not always possible, regardless of the shape of the fetoscope (straight or curved).
Once the trocar has been placed, the introduction of the fetoscope in the amniotic cavity will allow visualization of the placental vessels, identification of the vascular equator and localization of the anastomoses. A 400 or 600 µm laser fiber can be passed through the working channel of the fetoscope. A diode or NdYag laser machine with a wave length of 980 nm is used to produce a 40W laser beam to coagulate the vascular connections under direct vision.
Correct individualization of every anastomosis and selective coagulation of these specific areas, preserving the rest of the placenta, has been broadly used but it is being abandoned since it may miss small anastomoses leading to incomplete treatment. Coagulating the placental plate from edge to edge with a continuous line joining the vascular anastomoses (Solomon technique), has been proven to be superior in achieving successful dichorionization, with a fivefold reduction of the incidence of postoperative complications derived from incomplete lasering(33,34).
Coagulation in a sequential manner, depending on the type and flow direction of the anastomoses, could contribute to intraoperative balancing of the circulation and may improve double survival(35,36).
Once the laser coagulation has finished, amniodrainage is performed through the introducer aiming for a DVP of 5 or 6 cm.
Post-operative complications
Preterm premature rupture of membranes (PPROM)
Post procedure PPROM <34 weeks has an overall rate close to 40%. Median procedure-to-PPROM interval is 46 days. Thus, PPROM is particularly deleterious when it follows FLC performed before viability, leading to miscarriage or extreme preterm birth(37). Chorioamniotic separation and membrane tearing at the entry site may be responsible for these complications. Due to the inability of the amniotic membranes to heal, and since no effective method to seal the defect caused by the trocar has been found, fetoscopy is performed when the potential benefits of the surgery outweigh the risks related to PPROM (miscarriage or preterm delivery). Assuming these risks in severe cases (QS II-IV) is justified. Considering that survival is influenced directly by the severity of the condition at the time of the FLC(38), and since there is no accurate method for predicting progression, preventive FLC for QS I may be justified, but assuming the risk of PPROM, especially in pre-viable cases, is controversial.
Recurrent TTTS
Recurrent TTTS can be diagnosed in around 3% and 7% of the cases at any time following selective FLC or Solomon technique respectively(39). A persistent unidirectional blood flow from the donor to the recipient together with an upregulated RAS, could possibly explain why, even though most of the anastomosis were eliminated, the full spectrum of the syndrome can reappear. Early diagnosis is challenging specially if unintentional septostomy was caused by laser coagulation during the procedure, leading to a milder oligo-polyhydramnios sequence. Non-normalization of donor´s urination and/or Doppler parameters may help in the diagnosis.
Post-operative twin anemia-polycythaemia sequence (taps)
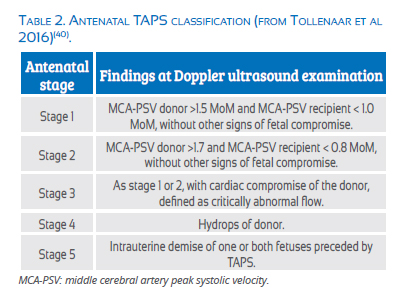
Primary TAPS affects up to 5% of MC twins. It is a complication derived from predominant unidirectional blood flow through small and scarce placental anastomoses. The amount of blood exchange is insufficient to produce a TTTS but, in the long run, this chronic transfusion will result in anemia in one twin and polycythemia in the other(9). Its classification and management depend on severity [Table 2](40).
An incomplete FLC can lead to postoperative TAPS in 1% and 16% of the cases treated with selective coagulation or Solomon technique respectively(39). Usually the ex-recipient twin is the one presenting anemia. This could indicate that in these cases the anastomoses left untreated allows mainly unidirectional blood flow from the ex-recipient to the ex-donor.
Pseudo amniotic band syndrome (pabs)
PABS can affect 2% of MC twin pregnancies treated with FLC. Unintentional septostomy during surgery, chorioamniotic membrane separation and PPROM following FLC are risk factors for PABS, and close surveillance of fetal limbs during follow up is needed. If any of the limbs is affected and its blood supply compromised, fetoscopic release of the amniotic band can be attempted(32,41).
Repeated fetal laser coagulation
Repeated FLC should be attempt if the recurrent TTTS or severe TAPS are seen before 28 weeks. A free-floating intertwin membrane due to unintentional septostomy during FLC usually adds technical difficulties in the identification of untreated anastomoses(32). In some cases, blood stained amniotic fluid can also affect visualization. Due to the complexity of the procedure and further iatrogenic damage to the amniotic membrane, the overall survival following repeated FLC is around 40%(42).
After 27 weeks, elective delivery after lung maturation is preferred for post-operative severe TTTS or TAPS. In order to prolong pregnancy, expectant management and amnioreduction or blood transfusion can be considered for mild TTTS and TAPS respectively.
Long term outcome following FLC
In general terms, the rate of severe neurodevelopmental impairment in twin pregnancies treated with FLC for TTTS is around 10%, with cerebral palsy as the most frequent condition (40%) and no difference between donors and recipients(43). Lower birth weight seems to be an independent factor associated with neurodevelopmental impairment (OR 1.33 for each week, 95% CI 1.05- 1.67, P=.02)(44).
Conclusion
The indication for FLC using Solomon technique for severe TTTS presenting before 28 weeks is clear. Mild cases in the context of sFGR, reversal of TTTS or short cervix are also candidates for surgery.
Cervical cerclage after FLC should be considered for a CL <15 mm at any stage. Survival and longterm outcomes are directly influenced by the severity of TTTS; therefore, FLC for mild cases has been proposed. If a method to significantly decrease the risk of postoperative PPROM and preterm delivery is found, every MC twin pregnancy presenting with the oligohydramnios / polyhydramnios sequence will be treated. Two relevant randomized control trial are being held: 1) Immediate treatment vs. delayed intervention upon progression for QS I cases (ClinicalTrials. gov Identifier: NCT01220011); and, 2) Prophylactic use of Arabin cervical pessary after FLC for TTTS vs. FLC alone (ClinicalTrials.gov Identifier: NCT01334489).
Conflictos de interés: Ninguno.
Financiamiento: Ninguno
Citar como: Gil Pugliese S. Fetoscopic laser surgery for twin to twin transfusion syndrome. Rev Peru Ginecol Obstet. 2018;64(4):607-614 DOI: https://doi.org/10.31403/rpgo.v64i2130
References
1. Gratacós E, Ortiz JU, Martinez JM. A systematic approach to the differential diagnosis and management of the complications of monochorionic twin pregnancies. Fetal Diagn Ther. 2012;32(3):145-55. doi: 10.1159/000342751. [ Links ]
2. Sebire NJ, Snijders RJ, Hughes K, Sepulveda W, Nicolaides KH. The hidden mortality of monochorionic twin pregnancies. Br J Obstet Gynaecol. 1997;104:1203-7. [ Links ]
3. Acosta-Rojas R, Becker J, Munoz-Abellana B, Ruiz C, Carreras E, Gratacos E; Catalunya and Balears Monochorionic Network. Twin chorionicity and the risk of adverse perinatal outcome. Int J Gynaecol Obstet. 2007;96:98-102.5. [ Links ]
4. Ong SS, Zamora J, Khan KS, Kilby MD. Prognosis for the co-twin following single-twin death: a systematic review. Br J Obstet Gynecol. 2006;113:992e8. [ Links ]
5. Fusi L, Gordon H. Twin pregnancy complicated by single intrauterine death. Problems and outcome with conservative management. Br J Obstet Gynaecol. 1990;97:511e6. [ Links ]
6. Kilby MD, Bricker L on behalf of the Royal College of Obstetricians and Gynaecologists. Management of monochorionic twin pregnancy. BJOG. 2016;124:e1– e45. [ Links ]
7. Djaafri F, Stirnemann J, Mediouni I, Colmant C, Ville Y. Twin-twin transfusion syndrome What we have learned from clinical trials. Semin Fetal Neonatal Med. 2017 Dec;22(6):367-75. doi: 10.1016/j.siny.2017.08.005. [ Links ]
8. Senat MV, Deprest J, Boulvain M, Paupe A, Winer N, Ville Y. Endoscopic laser surgery versus serial amnioreduction for severe twin-to-twin transfusion syndrome. N Engl J Med. 2004;351:136e44. [ Links ]
9. Couck I, Lewi L. The placenta in twin-to-twin transfusion syndrome and twin anemia polycythemia sequence. Twin Res Hum Genet. 2016 Jun;19(3):184-90. doi:10.1017/thg.2016.29. [ Links ]
10. Zhao DP, Cohen D, Middeldorp JM, Klumper FJ, Haak MC, Oepkes D, Lopriore E. The role of veno-venous anastomoses in twin-twin transfusion syndrome. Placenta. 2014 May;35(5):334-6. doi: 10.1016/j.placenta.2014.03.002. [ Links ]
11. Khalil A. Modified diagnostic criteria for twin-to-twin transfusion syndrome prior to 18 weeks’ gestation: time to change? Ultrasound Obstet Gynecol. 2017 Jun;49(6):804-5. doi:10.1002/uog.17443.
12. Gratacós E, Lewi L, Muñoz B, Acosta-Rojas R, Hernandez-Andrade E, Martinez JM, Carreras E, Deprest J. A classification system for selective intrauterine growth restriction in monochorionic pregnancies according to umbilical artery Doppler flow in the smaller twin. Ultrasound Obstet Gynecol. 2007 Jul;30(1):28-34. [ Links ]
13. Peeva G, Bower S, Orosz L, Chaveeva P, Akolekar R, Nicolaides KH. Endoscopic placental laser coagulation in monochorionic diamniotic twins with type II selective fetal growth restriction. Fetal Diagn Ther. 2015;38(2):86-93. doi:10.1159/000374109. [ Links ]
14. Mahieu-Caputo D, Salomon LJ, Le Bidois J, Fermont L, Brunhes A, Jouvet P, Dumez Y, Dommergues M. Fetal hypertension: an insight into the pathogenesis of the twin-twin transfusion syndrome. Prenat Diagn. 2003 Aug;23(8):640-5. [ Links ]
15. Eschbach SJ, Boons LSTM, Van Zwet E, Middeldorp JM, Klumper FJCM, Lopriore E, Teunissen AKK, Rijlaarsdam ME, Oepkes D, Ten Harkel ADJ, Haak MC. Right ventricular outflow tract obstruction in complicated monochorionic twin pregnancy. Ultrasound Obstet Gynecol. 2017 Jun;49(6):737-43. doi:10.1002/uog.16008. [ Links ]
16. Galea P, Barigye O, Wee L, Jain V, Sullivan M, Fisk NM. The placenta contributes to activation of the renin angiotensin system in twin-twin transfusion syndrome. Placenta. 2008 Aug;29(8):734-42. doi:10.1016/j.placenta.2008.04.010. [ Links ]
17. Van Mieghem T, Doné E, Gucciardo L, Klaritsch P, Allegaert K, Van Bree R, Lewi L, Deprest J. Amniotic fluid markers of fetal cardiac dysfunction in twin-to-twin transfusion syndrome. Am J Obstet Gynecol. 2010 Jan;202(1):48.e1-7. doi:10.1016/j.ajog.2009.08.013. [ Links ]
18. Ong SS, Zamora J, Khan KS, Kilby MD. Prognosis for the co-twin following single-twin death: a systematic review. Br J Obstet Gynecol. 2006;113:992e8. [ Links ]
19. Hillman SC, Morris RK, Kilby MD. Co-twin prognosis after single fetal death: a systematic review and meta-analysis. Obstet Gynecol. 2011 Oct;118(4):928-40. doi:10.1097/AOG.0b013e31822f129d. [ Links ]
20. Jelin AC, Norton ME, Bartha AI, Fick AL, Glenn OA. Intracranial magnetic resonance imaging findings in the surviving fetus after spontaneous monochorionic cotwin demise. Am J Obstet Gynecol. 2008 Oct;199(4):398.e1-5. doi:10.1016/j.ajog.2008.06.062. [ Links ]
21. Quintero RA, Morales WJ, Allen MH, Bornick PW, Johnson PK, Kruger M. Staging of twin–twin transfusion syndrome. J Perinatol. 1999;19:550–5. [ Links ]
22. Duryea EL, Happe SK, McIntire DD, Dashe JS. The natural history of twin-twin transfusion syndrome stratified by Quintero stage. J Matern Fetal Neonatal Med. 2016 Nov;29(21):3411-5. doi:10.3109/14767058.2015.11312 63. [ Links ]
23. Habli M, Michelfelder E, Cnota J, Wall D, Polzin W, Lewis D, Lim FY, Crombleholme TM. Prevalence and progression of recipienttwin cardiomyopathy in early-stage twin-twin transfusion syndrome. Ultrasound Obstet Gynecol. 2012 Jan;39(1):63-8. doi: 10.1002/uog.10117. [ Links ]
24. Stirnemann JJ, Nasr B, Proulx F, Essaoui M, Ville Y. Evaluation of the CHOP cardiovascular score as a prognostic predictor of outcome in twin-twin transfusion syndrome after laser coagulation of placental vessels in a prospective cohort. Ultrasound Obstet Gynecol. 2010;36:52e7. [ Links ]
25. Gil Guevara E, Pazos A, Gonzalez O, Carretero P, Molina FS. Doppler assessment of patients with twinto-twin transfusion syndrome and survival following fetoscopic laser surgery. Int J Gynaecol Obstet. 2017 Jun;137(3):241-5. doi:10.1002/ijgo.12143. [ Links ]
26. Baud D, Windrim R, Keunen J, Kelly EN, Shah P, van Mieghem T, Seaward PG, RyanG. Fetoscopic laser therapy for twin-twin transfusion syndrome before 17 and after 26 weeks’ gestation. Am J Obstet Gynecol. 2013 Mar;208(3):197.e1-7. doi:10.1016/j.ajog.2012.11.027.
27. Middeldorp JM, Lopriore E, Sueters M, Klumper FJ, Kanhai HH, Vandenbussche FP, Oepkes D. Twin-to-twin transfusion syndrome after 26 weeks of gestation: is there a role for fetoscopic laser surgery? BJOG. 2007 Jun;114(6):694-8. [ Links ]
28. Salomon LJ, Nasr B, Nizard J, Bernard JP, Essaoui M, Bussieres L, Ville Y. Emergency cerclage in cases of twin-to-twin transfusion syndrome with a short cervix at the time of surgery and relationship to perinatal outcome. Prenat Diagn. 2008 Dec;28(13):1256-61. doi: 10.1002/pd.2156. [ Links ]
29. Sumie M, Nakata M, Murata S, Miwa I, Sugino N. Two cases of reversal of twin-twin transfusion syndrome diagnosed by measuring hourly fetal urine production. J Obstet Gynaecol Res. 2009 Oct;35(5):983-6. doi:10.1111/j.1447-0756.2009.01042.x. [ Links ]
30. Huber A, Diehl W, Zikulnig L, Bregenzer T, Hackelöer BJ, Hecher K. Perinatal outcome in monochorionic twin pregnancies complicated by amniotic fluid discordance without severe twin-twin transfusion syndrome. Ultrasound Obstet Gynecol. 2006 Jan;27(1):48-52. [ Links ]
31. Costa-Castro T, De Villiers S, Montenegro N, Severo M, Oepkes D, Matias A, Lopriore E. Velamentous cord insertion in monochorionic twins with or without twin-twin transfusion syndrome: Does it matter? Placenta. 2013 Nov;34(11):1053-8. doi:10.1016/j.placenta.2013.08.009. [ Links ]
32. Cruz-Martinez R, Van Mieghem T, Lewi L, Eixarch E, Cobo T, Martinez JM,Deprest J, Gratacos E. Incidence and clinical implications of early inadvertent septostomy after laser therapy for twin-twin transfusion syndrome. Ultrasound Obstet Gynecol. 2011 Apr;37(4):458-62. doi: 10.1002/uog.8922. [ Links ]
33. Peralta CF, Molina FS, Gómez LF, Bennini JR, Gomes Neto O, Barini R. Endoscopic laser dichorionization of the placenta in the treatment of severe twin-twin transfusion syndrome. Fetal Diagn Ther. 2013;34(4):206-10. doi: 10.1159/000354898. [ Links ]
34. Slaghekke F, Lopriore E, Lewi L, Middeldorp JM, van Zwet EW, Weingertner AS,Klumper FJ, DeKoninck P, Devlieger R, Kilby MD, Rustico MA, Deprest J, Favre R, Oepkes D. Fetoscopic laser coagulation of the vascular equator versus selective coagulation for twin-to-twin transfusion syndrome: an open-label randomised controlled trial. Lancet. 2014 Jun 21;383(9935):2144-51. doi:10.1016/S0140-6736(13)62419-8. [ Links ]
35. Akkermans J, Peeters SH, Klumper FJ, Middeldorp JM, Lopriore E, Oepkes D. Is the sequential laser technique for twin-to-twin transfusion syndrome truly superior to the standard selective technique? A meta-analysis. Fetal Diagn Ther. 2015;37(4):251-8. doi: 10.1159/000365212. [ Links ]
36. Murata S, Takano M, Kagawa Y, Sumie M, Nakata M. The experience of modified sequential selective laser photocoagulation of communicating vessels technique for twin-twin transfusion syndrome. J Matern Fetal Neonatal Med. 2018 May;31(9):1137-1141. doi: 10.1080/14767058.2017.1311309. [ Links ]
37. Snowise S, Mann LK, Moise KJ Jr, Johnson A, Bebbington MW, Papanna R. Preterm prelabor rupture of membranes after fetoscopic laser surgery for twintwin transfusion syndrome. Ultrasound Obstet Gynecol. 2017 May;49(5):607-11. doi:10.1002/uog.15958. [ Links ]
38. Huber A, Diehl W, Bregenzer T, Hackeloer BJ, Hecher K. Stage-related outcome in twin352 twin transfusion syndrome treated by fetoscopic laser coagulation. Obstet Gynecol. 2006 Aug;108(2):333-7. [ Links ]
39. Donepudi R, Akkermans J, Mann L, Klumper FJ, Middeldorp JM, Lopriore E, Moise KJ Jr, Bebbington M, Johnson A, Oepkes D, Papanna R. Recurrent twin-twin transfusion syndrome (rTTTS) and twin anemia polycythemia sequence (TAPS) after fetoscopic laser surgery (FLS): size (of the cannula) does matter. Ultrasound Obstet Gynecol. 2017 Sep 19. doi: 10.1002/uog.18904. [ Links ]
40. Tollenaar LS, Slaghekke F, Middeldorp JM, Klumper FJ, Haak MC, Oepkes D,Lopriore E. Twin anemia polycythemia sequence: current views on pathogenesis,diagnostic criteria, perinatal management, and outcome. Twin Res Hum Genet. 2016 Jun;19(3):222-33. doi: 10.1017/thg.2016.18. [ Links ]
41. Ting YH, Lao TT, Law KM, Cheng YK, Lau TK, Leung TY. Pseudoamniotic band syndrome after in utero intervention for twin-to-twin transfusion syndrome: Case reports and literature review. Fetal Diagn Ther. 2016;40(1):67-72. doi:10.1159/000368601. [ Links ]
42. Robyr R, Lewi L, Salomon LJ, Yamamoto M, Bernard JP, Deprest J, Ville Y. Prevalence and management of late fetal complications following successful selective laser coagulation of chorionic plate anastomoses in twin-totwin transfusion syndrome. Am J Obstet Gynecol. 2006 Mar;194(3):796-803. [ Links ]
43. Rossi AC, Vanderbilt D, Chmait RH. Neurodevelopmental outcomes after laser therapy for twin-twin transfusion syndrome: a systematic review and meta-analysis. Obstet Gynecol. 2011 Nov;118(5):1145-50. doi: 10.1097/AOG.0b013e318231827f. [ Links ]
44. Lopriore E, Ortibus E, Acosta-Rojas R, Le Cessie S, Middeldorp JM, Oepkes D, et al. Risk factors for neurodevelopment impairment in twinetwin transfusion syndrome treated with fetoscopic laser surgery. Obstet Gynecol. 2009 Feb;113(2 Pt 1):361-6. doi: 10.1097/AOG.0b013e318195873e. [ Links ]
Correspondencia:
Recibido: 25 octubre 2018
Aceptado: 28 octubre 2018