Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Ginecología y Obstetricia
On-line version ISSN 2304-5132
Rev. peru. ginecol. obstet. vol.66 no.3 Lima Jul-Sep 2020
http://dx.doi.org/10.31403/rpgo.v66i2275
Original Articles
Human papillomavirus and associated factors in patients with unknown cytology treated in northern Peru
1 Faculty of Biological Sciences , Universidad Nacional Pedro Ruiz Gallo, Lambayeque, Perú.
2 Laboratory of Molecular Biology , Hospital Regional Lambayeque, Chiclayo, Perú.
Background: Human papillomavirus is cause of cervical cancer, one of the most common cancers among women. Objective: To determine the prevalence of human papillomavirus and associated factors in patients with unknown cytology. Methods: In gynecology patients with unknown cytology attended at Lambayeque Regional Hospital, at the northern coast of Peru, from April through June 2019, DNA extraction for human papillomavirus identification performed on cervical samples was based on the salting out method. Samples were processed by polymerase chain reaction. All samples were amplified for MY09 and MY11 primers, and PC04 / GH20 primers. Bivariate analysis used the chi-square and t-student tests. Results: 29.9% of the patients studied were infected with human papillomavirus. No statistically significant difference was found between human papillomavirus infection and age, age at first sexual intercourse, promiscuity, number of vaginal deliveries, cervical lesion, history of sexually transmitted infections, use of hormonal contraceptive or condoms, and smoking.
Key words: Human papillomavirus; Polymerase chain reaction; Clinical laboratory techniques
Introduction
Cervical cancer is one of the most common cancers in women in the world1. 585 278 new cases and 327 899 attribuTable deaths are predicted in 2010, with more than 80% of cases occurring in developing countries. Invasive cervical cancer accounts for 15% of cancers in women and ranks first or second among cancers in women in 13 of the 23 regions of the world2. Cervical cancer is caused by human papillomavirus (HPV) and epidemiological knowledge of the distribution of this virus infection in the general population is essential. Human papillomaviruses represent a heterogeneous family of doubly distributed DNA viruses of the taxonomic family Papillomaviridae3. Some types of human papilloma have a positive tropism for the skin and play a role in cancer4. The technique with the most sensitivity and specificity for HPV detection, and considered the gold standard for many years, is the polymerase chain reaction (PCR) using the MY09 and MY11 consensus primers, followed by genetic sequencing5. Molecular assays, specifically PCR, are very effective in the diagnosis of HPV6.
The objective of this study is to determine the prevalence of human papillomavirus and associated factors in obstetric-gynecological patients with unknown cytology in northern Peru.
Methods
This is a descriptive cross-sectional study. Consecutive non-probabilistic sampling was applied for convenience, which consisted of recruiting all the patients who met the selection criteria during the period April to June 2019. The patients filled out a questionnaire with closed questions with reproductive, demographic and sexual health data. Cervical samples. were collected from the OB / GYN service during the period, which were processed in the molecular biology laboratory of the hospital's research area.
The protocol and the research work were approved by the Ethics Committee of the Lambayeque Regional Hospital, Peru. All participating women signed an individual informed consent, prior explanation and information to carry out the study.
In the molecular characterization of samples, for the previous treatment, the tube containing the cytobrush was vortexed in order to detach all the cells that could remain in it. The cytobrush was carefully removed and the remaining solution was transferred to a 1.5 mL tube. The sample was concentrated by centrifugation at 12 000 rpm for 5 minutes, then all the supernatant was removed and the blank extraction tube was added.
For extraction and purification, 360 μL of lysis buffer and 3 μL of β-mercaptoethanol were added to the sample. It was incubated for 1 hour in a water bath at 65 ° C, vortexing every 10 min. 90 µL of 8M potassium acetate was added and it was centrifuged at 12 000 rpm for 5 minutes; then, the supernatant was recovered in another 1.5 mL tube (this step was repeated twice).
In the precipitation, 450 µL of isopropanol was added and centrifuged at 12 000 rpm for 5 minutes; the supernatant was removed. It was washed with 450 µL of 70% ethanol and centrifuged at 12 000 rpm for 5 minutes. Then it was allowed to dry until the humidity was eliminated. It was resuspended in 25 µL of H2O PCR.
The quality and integrity evaluation of the genomic DNA was carried out by agarose gel electrophoresis, whose concentration was 1% prepared with tris-acetate-EDTA buffer (TAE). For this, the agarose was dissolved in tris-acetateEDTA buffer and heated in a microwave until boiling. Then it was poured into a holder with the comb, waiting for it to gel. The gel ready was placed in the electrophoresis chamber containing 1X tris-acetate-EDTA buffer. In the wells of the agarose gel, 1 μL of sample previously mixed with 1 μL of loading buffer and 8 μL of H2O PCR was loaded. With the power source, current was applied at 30V for 10 minutes, 70V for 30 minutes and 30V for 10 minutes. After that time, the gel was stained in a solution with ethidium bromide at a concentration of 0.5 mg / mL. The DNA image was then visualized and captured with the help of the Pharos Fx Plus photo-documentator. The quality and quantity of DNA was determined by electrophoresis in 1% agarose gels. Quantification was carried out using a marker with a known concentration.
The extracted DNA was amplified by the polymerase chain reaction technique. All samples were amplified to primers MY09 and MY11, and an internal quality control was done to verify if it was amplifiable DNA (human betaglobin) primers PCo4 / GH20 and to observe its components. All samples were amplified for the 450 bp consensus restriction enzymes of the HPV L1 gene, MY09: (5'-CGTCCMARRGGAWACTGATC-3 ') and MY11: (5'-GCMCAGGGWCATAAYAATGG -3 ') (7-10), and beta subunit hemoglobin (HBB) GH20: (5′-GAAGAGCCAAGGACAGGTAC-3 ′) and PC04: (5′-CAACTTCATCCACGTTCACC-3 ′) of 268 bp were used(11). If a sample contained detecTable β-globin DNA, then the HPV DNA was intact and amplifiable during the polymerase chain reaction(12).
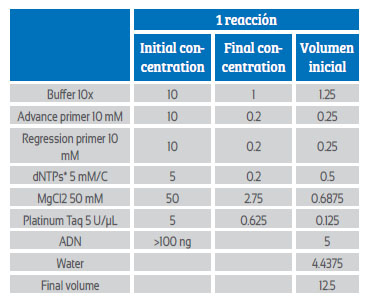
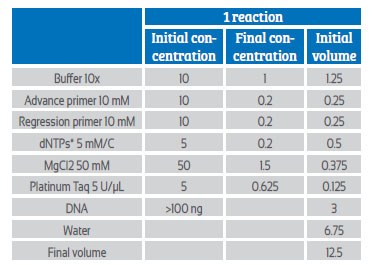
The preparation of the mix for the β-globin polymerase chain reaction and human papillomavirus was carried out according to Tables 1 and 2 at a final volume of 12.5 μL in each reaction.
Table 1 Bgh Taq platinium-invitrogen concentrations and volumes.
*dntps=deoxynucleotide triphosphates
The amplification reactions for the HPV polymerase chain reaction were carried out in an Eppendorf thermocycler and the oligonucleotide sequence MY09 / MY11 with the sequence MY09 (CGTCCMARRGGAWACTGATC), MY11 (GCMCAGGGWCATAAYAATGG) and PC04 / GH20. The optimal thermodynamic conditions consisted of the following steps: pre-naturalization 3 minutes at 94 ° C; annealing 35 cycles for 45 seconds at 94 ° C, 1 minute at 55 ° C and 1 minute at 72 ° C; extension at 72 ° C for 7 minutes.
Regarding electrophoresis and band visualization, the resuspended samples were examined by electrophoresis on a 1.5% agarose gel, using a 100 base pair molecular weight marker. Subsequently, it was taken to electrophoretic run at 70 V for 10 minutes and then at 120 V for 45 minutes. Then the gels were stained for 10 minutes in a 0.5 mg / mL concentration solution of ethidium bromide. For its visualization, a Pharos FX plus brand molecular scanner was used. To identify the association between human papillomavirus infection and demographic characteristics (age, educational level, marital status, number of sexual partners), the t-student and Mann-Whitney tests were performed, using the Stata version 15 program.
Results
186 patient samples were analyzed; of them, 53 had a positive result for human papillomavirus (29.9%) (Table 3) and 124 were negative (70.1%).
Table 3 General characteristics of patients.
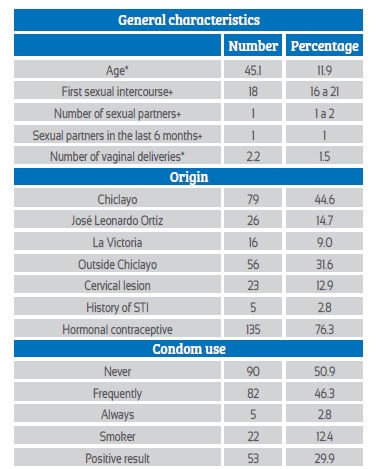
*DNTPS=deoxynucleotide triphosphates *Mean and standard deviation were used. +Mean and interquartile range were used. STI=sexually transmitted infections.
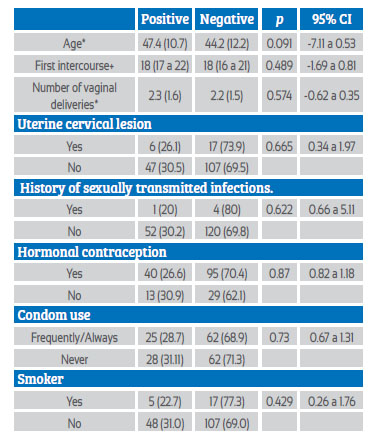
No statistically significant difference was found between the factors age, first sexual intercourse, number of vaginal deliveries, cervical injury, history of sexually transmitted infection (STI), use of hormonal contraceptives and condoms, promiscuity, smoker, and a positive result (Table 4).
Discussion
A small group of papillomaviruses are the etiologic agents of several types of human cancers, including carcinomas of the anogenital tract13. In the study in Chiclayo, a city in the north coast of Peru, in patients with unknown cytol ogy, prevalence of human papilloma virus in the uterine cervix was 29.9%, that is, in almost one third of the participants. A similar study with the same methodological characteristics performed by Serquén-López in the same city of Chiclayo in sex workers, a similar prevalence was found14. Studies carried out in other cities of Peru, such as Valderrama et al. (2007) in students from Lima, Peru, the prevalence was 8.4%15. SullcahuamanAllende et al., in a study at the National Institute of Neoplastic Diseases, Lima, Peru, found that 32.5% tested positive16. Manrique-Hinojosa et al. found high prevalence in Lima students (43.4%)17 and Iwasaki et al., 34.5% in Lima urban population18. Santos et al. detected HPV DNA in 95.3% of women with squamous cell carcinoma and in 92.0% of women with adenocarcinoma / adenosquamous carcinoma, compared to 17.7% in control women19. These results are similar to those of Mendoza et al.8, in 2012, with a prevalence of 21% in women with negative cytology in a Paraguayan locality, and by Winer et al., in 603 university students in the state of Washington, USA, at 4-month intervals between 1990 and 2000. At 24 months, the cumulative incidence of first-time infection was 3.3%20. The prevalence in the study by Brot et al. was 63% in a Brazilian population21, and Ingabire et al. found that 8.6% were positive for HPV at the beginning of the study in 200722. These differences are possibly due to the types of study and the region of research. In Peru, the prevalence of the reviewed studies is varied.
Sociodemographic factors such as age and marital status are frequently risk markers for exposure to HPV and other sexually transmitted infections16. Results such as age, beginning of active sexual life, number of sexual partners, promiscuity, number of vaginal deliveries, condom use, smokers, cervical injury, history of sexually transmitted infections, use of hormonal contraceptives were not associated with infection of human papillomavirus in our study. Other studies have found an association between HPV and these factors23-29. Sathian et al. and Bosch et al. mention that promiscuity and the number of sexual partners are factors of HPV infection30,31 and the study by Francheschi et al. mentions that there is a relationship between HPV infection and the age of first sexual intercourse32.
In our study, no statistical significance was found between human papillomavirus infection and the number of vaginal deliveries, similar to that found by Rajkumar et al.(33). Statistical significance was also not found between having had a cervical lesion and HPV infection, which differs from studies such as Bosch31, Oliveira34, Motoyama35, Naqvi, Wajid and Mitra36 and Harden and Munger13. The history of STIs, use of hormonal contraceptives and / or condoms and smoking did not show statistical significance with the positive result for HPV; This differs from the studies by Hellberg and Vaccarella, who find a relationship between these factors and HPV infection23,37,38.
The prevalence found in our study coincides with that of other studies. If we refer to associated factors, the results differ from those found in other continents, but are related to studies carried out in Peru. According to our results, the number of sexual partners and HPV infection are unrelated. This may be due to the fact that the study was done in a population with a low number of sexual partners, a conservative society, important factor in the contagion and persistence of the human papilloma virus in the body. A significant percentage of the patients were raped, thus beginning their sexual life. In addition, the patients tended to decrease the number of sexual partners during the interview, and there was a great lack of knowledge between the types of contraceptive methods.
Only a small part of this population smoked, so this carcinogenic factor would not be reflected in the results and would require the study of a larger population.
In studies similar to this one, there is lack of information in the patients' medical records. A strength of our study was the repetition of the test in case of indeterminate result. The hospital gynecological and oncological services can improve screening programs by performing molecular tests in case of negative and highly suspicious cytology39. The present study is one of the first accomplished in the Lambayeque region, Peru.
The World Health Organization has reported that cervical cancer is the second leading cause of malignancy and death in women worldwide40. Epidemiological studies have shown that highrisk human papillomavirus (HR-HPV) genotypes are the main cause of this disease41. It is necessary to use complementary diagnostic tools that allow detection of the HR-HPV genome, in order to increase the performance of conventional morphological diagnostic methods generally used to detect cervical cancer42. One weakness of the study is not having genotyped patients positive for human papillomavirus or having included patients with epithelial carcinoma in any of its stages in another group, which would give a better view of the types circulating in the region. Subsequent studies are recommended to increase the number of samples, better stratifying the patients and extending the test to men, since they are the reservoirs of the disease. Progress has been made with vaccinating girls in Peru, but this should soon cover boys and better focus on the types circulating in the region and in the country. Research on this topic should be strengthened, as it represents an important cause of female mortality.
We conclude that a high prevalence of patients with human papillomavirus was found. No statistically significant difference was found between human papillomavirus infection and age, age of first sexual intercourse, promiscuity, number of vaginal deliveries, cervical lesion, history of STIs, use of hormonal contraceptives, use of condoms, smoking.
REFERENCES
1. Ault KA. Epidemiology and natural history of human papillomavirus infections in the female genital tract. Infect Dis Obstet Gynecol [Internet]. 2006 [cited 16 June 2018];2006 Suppl:40470. http://www.ncbi.nlm.nih.gov/pubmed/16967912 [ Links ]
2. Ferlay J, Shin H-R, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer [Internet]. 15 December 2010 [cited 14 de agosto de 2019];127(12):2893-917. http://www.ncbi.nlm.nih.gov/pubmed/21351269 [ Links ]
3. Segondy M. Classification of papillomaviruses (HPV) [Internet]. Vol. 38, Revue Francophone des Laboratoires. Academic Press; 2008 [cited 14 August 2019]. p. 23-5. http://www.ncbi.nlm.nih.gov/pubmed/15183049 [ Links ]
4. de Sanjose S, Quint WG, Alemany L, Geraets DT, Klaustermeier JE, Lloveras B, et al. Human papillomavirus genotype attribution in invasive cervical cancer: a retrospective cross-sectional worldwide study. Lancet Oncol [Internet]. November 2010 [cited 14 August 2019];11(11):1048-56. http://www.ncbi.nlm.nih.gov/pubmed/20952254 [ Links ]
5. Gravitt PE, Manos MM. Polymerase chain reaction-based methods for the detection of human papillomavirus DNA. IARC Sci Publ [Internet]. 1992 [cited 14 August 2019];(119):121-33. http://www.ncbi.nlm.nih.gov/pubmed/1330905 [ Links ]
6. Rymsza T, Ribeiro EA, Das Chagas e Silva de Carvalho LF, Bhattacharjee T, de Azevedo Canevari R. Human papillomavirus detection using PCR and ATR-FTIR for cervical cancer screening. Spectrochim Acta Part A Mol Biomol Spectrosc. 2018 May 5;196:238-46. DOI: 10.1016/j.saa.2018.02.004 [ Links ]
7. Gravitt PE, Peyton CL, Alessi TQ, Wheeler CM, Coutlée F, Hildesheim A, et al. Improved amplification of genital human papillomaviruses. J Clin Microbiol [Internet]. 2000 [cited 16 June 2018];38(1):357-61. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC88724/pdf/jm000357.pdf [ Links ]
8. Mendoza L, Arbiza J, Páez M, Kasamatsu E, Castro A, Giménez G, et al. Características clínico-demográficas y tipificación del virus de papiloma humano en mujeres paraguayas con citologías negativas para lesión escamosa intraepitelial. Mem Inst Investig Cienc Salud Junio [Internet]. 2012 [citado 7 de octubre de 2017];10(1):46-55. http://scielo.iics.una.py/pdf/iics/v10n1/v10n1a06.pdf [ Links ]
9. Stamenkovic M, Knezevic A, Knezevic I, Kuzmanovic I, Karalic D, Milenkovic S, et al. High-risk human papilloma virus genotypes in cervical carcinoma of Serbian women: Distribution and association with pathohistological findings. Biologicals [Internet]. September 2016 [cited 5 October 2017];44(5):412-6. http://linkinghub.elsevier.com/retrieve/pii/S1045105616300264 [ Links ]
10. Mudini W, Palefsky JM, Hale MJ, Chirenje MZ, Makunike-Mutasa R, Mutisi F, et al. Human papillomavirus genotypes in invasive cervical carcinoma in HIV-seropositive and HIV-seronegative women in Zimbabwe. J Acquir Immune Defic Syndr [Internet]. 1 September 2018 [cited 30 September 2019];79(1):E1-6. http://www.ncbi.nlm.nih.gov/pubmed/29781877 [ Links ]
11. Reyna López L, Gonzalez Cabeza J, Araujo Jiménez A, Avila Vereau E, Gómez Castro K, Terán Rojas Y. Detección y genotipificación de papiloma virus humano por PCR-RFLP en pacientes atendidas en el hospital distrital Walter Cruz Vilca La Libertad-2015. Pueblo Cont [Internet]. 16 de febrero de 2016 [citado 7 de octubre de 2017];27(2):331-42. http://journal.upao.edu.pe/PuebloContinente/article/view/687 [ Links ]
12. Kleter B, Van Doorn LJ, Ter Schegget J, Schrauwen L, Van Krimpen K, Burger M, et al. Novel short-fragment PCR assay for highly sensitive broad-spectrum detection of anogenital human papillomaviruses. Am J Pathol [Internet]. December 1998 [cited 30 September 2019];153(6):1731-9. http://www.ncbi.nlm.nih.gov/pubmed/9846964 [ Links ]
13. Harden ME, Munger K. Human papillomavirus molecular biology. Mutat Res Mutat Res [Internet]. 2016 [cited 3 December de 2016];772. http://www.ncbi.nlm.nih.gov/pubmed/28528688 [ Links ]
14. Serquén-López LM, Iglesias-Osores SA, Arce-Gil ZL. Prevalencia de Papilomavirus Humano en trabajadoras sexuales atendidas en dos centros de salud de Chiclayo. Rev Del Cuerpo Médico Del HNAAA [Internet]. 2018;10(4):6-9. https://cmhnaaa.org.pe/ojs/index.php/rcmhnaaa/article/view/21/21 [ Links ]
15. Valderrama MC, Campos FE, Cárcamo CP, García PJ. Factores asociados a lesiones cervicales o presencia del virus del papiloma humano en dos poblaciones de estudiantes de Lima. Rev Peru Med Exp Salud Publica [Internet]. 2007 [citado 21 de octubre de 2017];24(3):234-9. http://www.scielo.org.pe/scielo.php?pid=S1726-46342007000300006&script=sci_arttext [ Links ]
16. Sullcahuaman-Allende Y, Castro-Mujica M del carmen, Mejía-Farro R, Castaneda CA, Castillo M, Dolores-Cerna K, et al. Características sociodemográficas de mujeres peruanas con virus papiloma humano detectado por PCR-RFLP. Rev Peru Med Exp Salud Publica [Internet]. 2015 [citado 5 de octubre de 2017];32(3):509-14. http://www.scielosp.org/scielo.php?script=sci_arttext&pid=S1726-46342015000300015&lng=en&nrm=iso&tlng=en [ Links ]
17. Manrique-Hinojosa J, Núñez-Teran MDC, Pretel-Ydrogo L, Sullcahuaman-Allende Y, Roa-Meggo Y, Juárez-Coello P, et al. Detection of the human papillomavirus in samples obtained by self-collection technique in a group of peruvian college students. Rev Peru Med Exp Salud Publica [Internet]. 2018 [citado 25 de octubre de 2019];35(4):642-6. http://www.ncbi.nlm.nih.gov/pubmed/30726431 [ Links ]
18. Iwasaki R, Galvez-Philpott F, Arias-Stella J, Arias-Stella J. Prevalence of high-risk human papillomavirus by cobas 4800 HPV test in urban Peru. September 2014;18(5):469-72. doi: 10.1016/j.bjid.2014.01.010 [ Links ]
19. Santos C, Muñoz N, Klug S, Almonte M, Guerrero I, Alvarez M, et al. HPV types and cofactors causing cervical cancer in Peru. Br J Cancer [Internet]. 28 September 2001 [cited 7 October 2017];85(7):966-71. http://www.ncbi.nlm.nih.gov/pubmed/11592767 [ Links ]
20. Winer RL, Lee S-K, Hughes JP, Adam DE, Kiviat NB, Koutsky LA. Genital human papillomavirus infection: incidence and risk factors in a cohort of female university students. Am J Epidemiol [Internet]. 1 February 2003 [cited 8 March 2019];157(3):218-26. http://www.ncbi.nlm.nih.gov/pubmed/12543621 [ Links ]
21. De Brot L, Pellegrini B, Moretti ST, Carraro DM, Soares FA, Rocha RM, et al. Infections with multiple high-risk HPV types are associated with high-grade and persistent low-grade intraepithelial lesions of the cervix. Cancer Cytopathol [Internet]. 1 February 2017 [cited 5 October 2017];125(2):138-43. http://doi.wiley.com/10.1002/cncy.21789 [ Links ]
22. Ingabire C, Lim MK, Won Y-J, Oh J-K. HPV genotype-specific persistence and potential risk factors among Korean women: results from a 2-year follow-up study. Cancer Res Treat [Internet]. 17 August 2017 [cited 4 October 2017];1-26. http://www.e-crt.org/journal/view.php?doi=10.4143/crt.2017.340 [ Links ]
23. Vaccarella S, Herrero R, Snijders PJF, Dai M, Thomas JO, Hieu NT, et al. Smoking and human papillomavirus infection: pooled analysis of the International Agency for Research on Cancer HPV Prevalence Surveys. Int J Epidemiol [Internet]. 1 June 2008 [cited 18 October 2017];37(3):536-46. https://academic.oup.com/ije/article-lookup/doi/10.1093/ije/dyn033 [ Links ]
24. Vieira RC, Valente S, Manso EP, Renata M, Tsutsumi MY, Aoba E, et al. Prevalence of type-specific HPV among female university students from northern Brazil. Infect Agent Cancer [Internet]. 2015 [cited 5 October 2017];10:1-5. http://www.ncbi.nlm.nih.gov/pubmed/26203300 [ Links ]
25. Müller EE, Rebe K, Chirwa TF, Struthers H, McIntyre J, Lewis DA. The prevalence of human papillomavirus infections and associated risk factors in men-who-have-sex-with-men in Cape Town, South Africa. BMC Infect Dis [Internet]. 22 August 2016 [cited 7 October 2017];16(1):440. http://www.ncbi.nlm.nih.gov/pubmed/27549219 [ Links ]
26. Baloch Z, Yasmeen N, Li Y, Ma K, Wu X, Yang S, et al. Prevalence and risk factors for human papillomavirus infection among Chinese ethnic women in southern of Yunnan, China. Brazilian J Infect Dis [Internet]. 1 May 2017 [cited 16 June 2018];21(3):325-32. https://www.sciencedirect.com/science/article/pii/S141386701630633X?via%3Dihub [ Links ]
27. Smith JS, Green J, Berrington De Gonzalez A, Appleby P, Peto J, Plummer M, et al. Cervical cancer and use of hormonal contraceptives: A systematic review. Lancet [Internet]. 5 April 2003 [cited 9 October 2017];361(9364):1159-67. https://www.ncbi.nlm.nih.gov/pubmed/12686037 [ Links ]
28. Green J, Berrington de Gonzalez A, Smith JS, Franceschi S, Appleby P, Plummer M, et al. Human papillomavirus infection and use of oral contraceptives. Br J Cancer [Internet]. 2003 [cited 4 October 2017];88(11):1713-20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2377143/pd-f/88-6600971a.pdf [ Links ]
29. Jemal A, Center MM, DeSantis C, Ward EM. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol biomarkers Prev [Internet]. 1 August 2010 [cited 19 October 2017];19(8):1893-907. http://www.ncbi.nlm.nih.gov/pubmed/20647400 [ Links ]
30. Sathian B, Babu MR, Van Teijlingen ER, Banerjee I, Roy B, Subramanya SH, et al. Ethnic variations in perception of human papillomavirus and its vaccination among young women in Nepal. Nepal J Epidemiol [Internet]. 13 July 2017 [cited 4 October 2017];7(1):647. www.nepjol.info/index.php/NJE [ Links ]
31. Bosch FX, Lorincz A, Munoz N, Meijer CJLM, Shah K V. The causal relation between human papillomavirus and cervical cancer. J Clin Pathol [Internet]. April 2002 [cited 7 October 2017];55(4):244-65. http://www.ncbi.nlm.nih.gov/pubmed/11919208 [ Links ]
32. Franceschi S, Plummer M, Clifford G, de Sanjose S, Bosch X, Herrero R, et al. Differences in the risk of cervical cancer and human papillomavirus infection by education level. Br J Cancer [Internet]. 1 September 2009 [cited 7 October 2017];101(5):865-70. http://www.ncbi.nlm.nih.gov/pubmed/19654578 [ Links ]
33. Rajkumar T, Cuzick J, Appleby P, Barnabas R, Beral V, Berrington De González A, et al. Cervical carcinoma and reproductive factors: Collaborative reanalysis of individual data on 16,563 women with cervical carcinoma and 33,542 women without cervical carcinoma from 25 epidemiological studies. Int J Cancer [Internet]. 1 September 2006 [cited 9 Octo-ber 2017];119(5):1108-24. http://doi.wiley.com/10.1002/ijc.21953 [ Links ]
34. Oliveira GR de, Siqueira JD, Finger-Jardim F, Vieira VC, Silva RL, Gonçalves C V, et al. Characterisation of complete highand low-risk human papillomavirus genomes isolated from cervical specimens in southern Brazil. Mem Inst Oswaldo Cruz [Internet]. October 2017 [cited 16 June 2018];112(10):728-31. http://www.ncbi.nlm.nih.gov/pubmed/28954002 [ Links ]
35. Motoyama S, Ladines-Llave CA, Luis Villanueva S, Maruo T. The role of human papilloma virus in the molecular biology of cervical carcinogenesis. Kobe J Med Sci [Internet]. 2004 [cited 21 October 2017];50(1):9-19. http://www.med.kobe-u.ac.jp/journal/contents/50/9.pdf [ Links ]
36. Naqvi SH, Wajid S, Mitra AB. Restriction fragment length polymorphism of L1 amplicon using Rsa 1 detects five different human papillomavirus types and their co-infections among women attending a gynaecological outpatient department. J Virol Methods [Internet]. April 2004 [cited 6 October 2017];117(1):91-5. http://linkinghub.elsevier.com/retrieve/pii/S0166093403004002 [ Links ]
37. Hellberg D, Stendahl U. The biological role of smoking, oral contraceptive use and endogenous sexual steroid hormones in invasive squamous epithelial cervical cancer. Anticancer Res. 2005;25(4):3041-6. [ Links ]
38. Kjellberg L, Hallmans G, Ahren AM, Johansson R, Bergman F, Wadell G, et al. Smoking, diet, pregnancy and oral contraceptive use as risk factors for cervical intra-epithelial neoplasia in relation to human papillomavirus infection. Br J Cancer [Internet]. April 2000 [cited 4 October 2017];82(7):1332-8. http://www.ncbi.nlm.nih.gov/pubmed/10755410 [ Links ]
39. Iglesias-Osores SA, Pando-Sánchez H. Tamizaje en cáncer de cérvix como prevención. Rev Exp en Med del Hosp Reg Lambayeque [Internet]. 2017;3(2):82. http://rem.hrlamb.gob.pe/index.php/REM/article/view/91/82 [ Links ]
40. Carestiato FN, Silva KC, Dimetz T, Oliveira LHS, Cavalcanti SMB. Prevalence of human papillomavirus infection in the genital tract determined by hybrid capture assay. Braz J Infect Dis. October 2006;10(5):331-6. [ Links ]
41. Coico-Vega MM, Iglesias-Osores S, Aguilar-Gamboa FR. Detección de oncoproteínas e6/e7: una alternativa para el tamizaje de cáncer de cérvix. Rev Exp en Med del Hosp Reg Lambayeque [Internet]. 2018;4(3). http://rem.hrlamb.gob.pe/index.php/REM/article/view/245 [ Links ]
42. Munoz N, Bosch FX, de Sanjosé S, Herrero R, Castellsagué X, Shah K V., et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med. February 2003;348(6):518-27. [ Links ]
Received: June 04, 2020; Accepted: August 17, 2020











 text in
text in