INTRODUCTION
Placenta accreta is a rare1 and life-threatening obstetric occurrence of pregnancy2. This abnormality in placental adhesion is caused by the absence or deficiency in the formation of Nitabuch's membrane, which is a fibrinoid layer that separates the chorion frondosa from the basal decidua and at delivery forms the separation membrane between the placenta and the uterus, which prevents the placenta from infiltrating the myometrium. Irvin and Hertin postulated in 1973 that the placenta accreta is caused by a deficiency in the decidual layer3.
It occurs more frequently towards the end of pregnancy and not necessarily during labor4. In the first trimester of pregnancy, its occurrence has been reported in few cases5.
Current cesarean section rates and the association with placenta previa increase the risk of placenta accreta more than 10-fold2,4. Placenta percreta, a rarer and more severe clinical form of placenta accreta, accounts for 5% of all cases4.
Other factors, in addition to cesarean section and placenta previa, increase the risk of occurrence of abnormal placental adhesion and uterine rupture: multiparity, advanced reproductive age, endometriosis, uterine dilatation and curettage, conventional myomectomies and hysteroscopic resections of septa and myomas1-7. Recently, some studies have associated assisted reproduction (in vitro fertilization and cryopreserved embryo transfer) to the appearance of abnormal placental adhesion, particularly placenta percreta, with spontaneous uterine rupture in the first trimester4,7. In this article we present a case of placenta accreta in early pregnancy. The clinical history, results of imaging tests, histopathology and management are described.
CLINICAL CASE
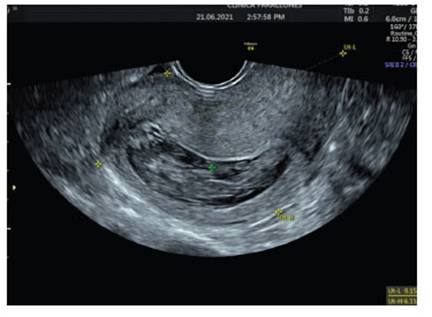
Patient 31 years old, G4 P2 A1, with two vaginal deliveries; the abortion occurred 2 years ago was managed with uterine curettage. Last menstrual period was on May 8, 2021; the planning method consisted of monthly injectable hormonal contraceptive injections (norethisterone (50 mg) and estradiol (5 mg)). She consulted a tertiary clinic for abnormal uterine bleeding. On first admission, incomplete abortion was diagnosed by ultrasound and positive β−hCG; the ultrasound showed a uterus with endometrium of 21 mm, heterogeneous, thickened, with mixed echoes and no other abnormal findings (Figure 1). She underwent curettage with Novak curette, with apparently normal discharge.
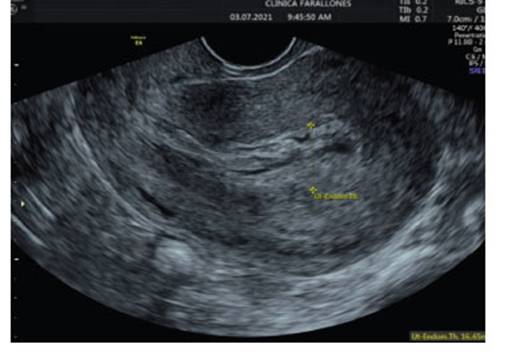
On second admission three days later, she continued with vaginal bleeding. Transvaginal ultrasound was reported with uterus in retroversoflexion (RVF), 39 mm thickened endometrium showing echo-mixed content and irregular gestational sac of 22 mm, for 6.6 weeks' gestation containing embryo and vitelline vesicle (Figure 2). A new uterine curettage was performed and the discharge was apparently normal.
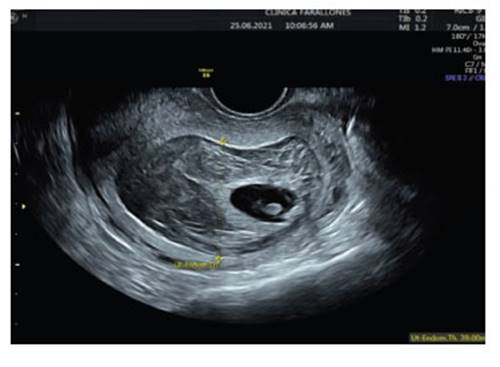
Figure 2 thickened endometrium, with ecomixed content and irregular gestational sac of 22 mm at 6.6 weeks' gestation.
After 8 days, the patient consulted again for pelvic pain and persistent heavy bleeding. A new ultrasound showed a 17 mm thickened endometrium (Figure 3). The pathology results of the previous curettage were reviewed and showed decidual fragments and placental debris, respectively. Medical management with misoprostol was decided and the patient was discharged.
She consulted again 37 days later, due to abundant vaginal bleeding of sudden onset, associated with lipotimia. She was admitted to the emergency room. Transvaginal ultrasound was reported with RVF uterus, 82 x 55 x 56 mm, homogeneous endometrium of 9.2 mm without evidence of intrauterine gestation, greater than 4 weeks. Towards the fundus and posterior uterine region, a tortuous vascularized image at the level of the myometrium was identified with Doppler type II. Findings were suggestive of uterine vascular malformation (Figure 4).
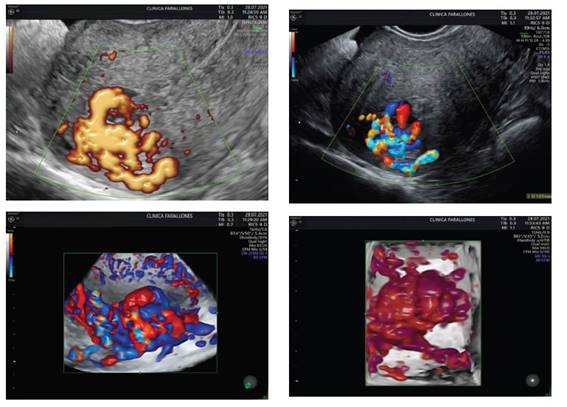
Figure 4 homogeneous endometrium without evidence of intrauterine gestation, with size more than 4 weeks. towards the fundus and posterior region, a tortuous vascularized image was identified with doppler type ii.
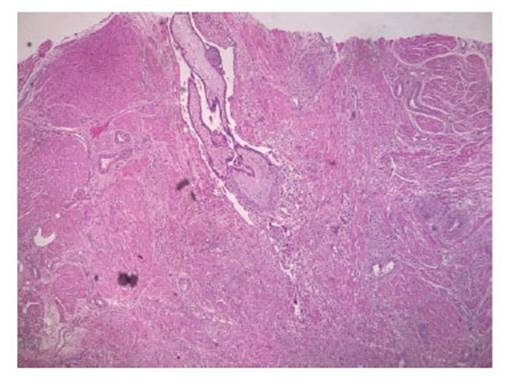
Due to persistent bleeding, decreased hemoglobin and satisfied parity, it was decided to perform a total abdominal hysterectomy. The specimen was sent for histopathological study. In the macroscopic description, an exophytic lesion was reported in the uterine fundus, irregular, hemorrhagic, with tortuous and congestive vessels, extending to the thickness of the underlying myometrium. In the microscopic description, the lesion described in the uterine fundus corresponded to numerous chorionic villi, some of them with hyaline changes, surrounded by trophoblastic cellular elements in direct contact with deep myometrial muscle fibers, without intervening decidual tissue. The final report concluded placental accreta (Figure 5). The patient had a favorable evolution and was discharged the day after surgery, clinically stable and asymptomatic. No wound healing problems were observed during the 3-month follow-up period.
DISCUSSION
The infiltration of the myometrium by chorionic villi is called placental accreta and, depending on the involvement of the myometrium, it is also described as the increta form that penetrates the myometrium and the percreta form, which penetrates the myometrium into the serosa or adjacent organs2,8. Placental accreta is a pathology mainly of last trimester gestations. However, its rare presentation has been shown in first trimester gestations. The frequency of presentation varies between 10 and 48 per 10,000 deliveries7,9.
Myometrial infiltration is associated with defects in early placentation due to infiltration of the invading chorionic villi (non-hairy trophoblast), in the presence of a deficiency of the fibrinoid glycoprotein of the decidua, which constitutes the immunological and physical barrier to the trophoblast. This phenomenon would be caused, among others, by previous endometrial injury, such as that occurring in cesarean section scars, uterine curettage, Asherman's syndrome, more than three deliveries, factors also associated with placenta previa, which are linked to placental accreta in up to 25% of cases and with abnormal development of the uterus-placental circulation. It manifests itself from the second trimester onwards, so its finding in the first trimester is a rare event, but not exempt from complications10,11.
Although the etiology of placental accreta is unknown, several risk factors have been identified, including a history of cesarean section, especially cases with repeated lower segment cesarean section which leaves scar tissue on the anterior uterine wall1,9,12.
The diagnosis of placental accreta is known to be difficult during the first trimester, with lower accuracy compared to those obtained in the second and third trimester1,2,6,9,12.
Several ultrasound markers have been described in the first trimester and their prevalence and type vary according to the weeks of the first trimester: early (6-9 weeks gestation) and late trimester (11-14 weeks gestation)6). These include low implantation of the gestational sac at 6-9 weeks, explained by the growth of the sac towards the fundic portion of the endometrium as pregnancy progresses; and a relationship with implantation in the lower uterine segment of patients with previous cesarean section, which markedly increases the risk of accretism6.
Other markers traditionally described in the second and third trimesters have also been identified in the late first trimester. Definitions of individual markers have been inconsistent, but include the presence of placental lacunae (sensitivity 80%), bladder with abnormal uterovesical interface (sensitivity 90%), uterovesical hypervascularity, and loss of the retroplacental clear zone (sensitivity 84.3%)6).
With color Doppler ultrasound and power Doppler imaging, evidence of first trimester placental accreta is similar to that seen in the second and third trimesters, including diffuse turbulent flow in the placental lacunae and increased peripheral vascularization. These findings can be seen at 8-9 weeks gestation6.
In addition, MRI with or without gadolinium can be used as a complementary diagnostic modality to improve prenatal diagnosis13. When comparing ultrasound and MRI with gadolinium, high sensitivity (77% and 88%, respectively) and high specificity (96% and 100%, respectively) are found for both modalities6,10,11.
Treatment of placenta accreta requires aggressive total surgical removal of trophoblastic material, good intravascular volume management and reconstructive surgery if necessary. The goal is hemorrhage control, for which total hysterectomy is indicated. However, there is little experience in the treatment of the disease during the first trimester. Therefore, the choice between hysterectomy or conservative therapy depends on the severity of the bleeding; but, in the cases found in the literature, hysterectomy is the standard treatment1,12.
Recent reports indicate that conservative invasive procedures to preserve fertility have also been described; these procedures included angiographic embolization of the uterine artery and laparoscopic or surgical resection of the affected area of the uterus. In some settings, preservation of the uterus, with the placenta in situ, may be considered. Adjuvant therapy with methotrexate has also been used to accelerate placental resorption1,12.