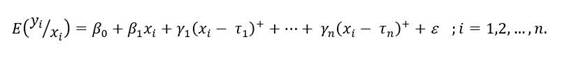Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de la Facultad de Medicina Humana
Print version ISSN 1814-5469On-line version ISSN 2308-0531
Rev. Fac. Med. Hum. vol.21 no.1 Lima Jan-Mar 2021
http://dx.doi.org/10.25176/rfmh.v21i1.3592
Original article
Gastric cancer mortality rate trend in Peru: Segmented regression model from 1995 to 2013
1Facultad de Ciencias y Filosofía de la Universidad Peruana Cayetano Heredia, Lima-Perú.
2Laboratorio de Series de Tiempo, Facultad de Ciencias Matemáticas, Universidad Nacional Mayor de San Marcos, Lima-Perú.
Objective:
To analyze the trends in gastric cancer mortality in Peru from 1995 to 2013 and their differences by gender, age groups, geographical and political regions.
Methods:
Ecological study of time series based on 49,690 deaths recorded from the Ministry of Health, from 1995 to 2013. Crude, specific, and standardized mortality rates were calculated by year, according to gender, age group, political and geographic regions. It analyzed trends by estimating joinpoints and annual percentage changes (CPA, for its initials in Spanish), using segmented regression models, adjusted using the software Joinpoint Regression Desktop version 4.5.0.0.
Results:
The standardized mortality rate (SMR) trend due to gastric cancer in Peru was decreasing, falling from 16.1 per 100,000 inhabitants. in 1995 to 11.4 per 100,000 inhabitants in 2013 (CPA: -2.3), observing decreasing trends by age groups, gender, and 17 of 25 political regions. Differences were found when comparing by subgroups: a more accelerated decrease in women (CPA -2.5) versus men (-2.0) and age groups (CPA for 75 - 79 years: -2.57 versus CPA for 40 - 44: -1.39); Three geographic areas with high mortality was identified: Central Andes (Huancavelica, Huánuco, Ayacucho, Pasco, and Junín), northern area (Lambayeque, La Libertad and Cajamarca) and central coast (Ica, Callao, Ancash, and Lima), also trends were observed growing in Huancavelica, Ayacucho and Pasco. On the coast, mortality has decreased since 1998. However, it decreases significantly in the Peruvian Highlands and Jungle as of 2009.
Conclusions:
The trend of SMR due to gastric cancer was decreasing for the period 1995 - 2013 with disparities by sex, age groups, political and geographic regions
Keywords: Trends; mortality rate; regression analysis; gastric cancer; Peru. (fuente: MeSH NLM).
INTRODUCTION
Worldwide, the incidence and mortality of gastric cancer have been drastically reduced in the last 70 years1,2,3. Despite this, it is the fifth most common cancer and the third leading cause of death due to cancer. According to the International Agency for Research on Cancer (IARC), there were 1,033,701 new cases of gastric cancer (incidence rate 11.1 per 100,000 inhabitants) and 782,685 deaths from this disease (mortality rate of 8 , 2 per 100,000 pop) in 20184,5. About 3 out of 4 new cases and deaths from gastric cancer occur in developing countries. (6.
In Peru, cancer ranks second in mortality since the 1990s7. The incidence of gastric cancer is 16.1 per 100,000 inhabitants, one of the highest in the world, surpassed by Asian countries such as South Korea (39,6), Mongolia (33,1), Japan (27,5), China (20,7), Bhutan (19,4) Kyrgyzstan (18,6) and other countries such as Chile (17,8) and Belarus (16,5) (4. Furthermore, the incidence rate is similar to the mortality rate in Peru due to patients’ poor survival after they are diagnosed8
Regional variations reflect differences in eating habits, food storage, and availability of fresh produce, as well as the prevalence ofHelicobacter pyloriinfection. (9,10ChronicH. pyloriinfection is the strongest identified risk factor for stomach cancer. (11
Some publications descriptively show a decreasing trend in gastric cancer mortality in Peru, although in a shorter period, such as 10 years (2005 to 2014) (12)and 8 years (2008 to 2015) (13, showing a reduction in the standardized rate. There are differences within Peru for gastric cancer mortality. The highest rates are found in the central highlands and the coast. The political regions with the highest mortality rate are Huánuco, Huancavelica, and Junín14, which are also regions with higher poverty indicators, less access to health services, less availability of drinking water15, In rural areas, the population drains its waste into the local river. This water is usually the same one treated for human consumption, finding a low concentration of chlorine. All of these factors favor the spread ofH. pyloriinfection. (8. Although it is also followed by more urbanized regions such as La Libertad and Callao12
Cancer is a disease of high cost and social impact on the population and implies a greater deployment of economic, human, and technological resources that must be optimally provided by health managers. Thus far, the reports of trends in mortality rates have been descriptive. They have used non-parametric trend analysis or linear log models. Therefore, the importance of improving epidemiological knowledge when performing the analysis of trends under a segmented regression model over long period (19 years). In this way, when estimating change points or joinpoints, the influence of variables that change over time can be hypothesized, either by a planned intervention or unintentional phenomena that alter the environment. (16,17
This research aimed to analyze the trends in gastric cancer mortality in Peru applying segmented regression models, during the period 1995 - 2013 and their differences by age groups, gender, political region, and geographic region.
METHODS
Type of study
An ecological time series study was carried out based on standardized mortality rates from stomach cancer.
Population and sample
The population was the set of all the executions of the stochastic process (ensemble) constituted by the standardized annual death rates for gastric cancer in Peru, from 1995 to 2013, the unit of analysis was the population of the country in each period. The sample consisted of the standardized annual death rates for the mentioned period.
Ethical aspects
The data used for this research are from a secondary source and were provided anonymously by the General Office of Statistics and Informatics of the Ministry of Health. (18
Data collection and processing
Records with an underlying cause of death coded according to the International Classification of Diseases (ICD) were selected, using the C152 code ninth version (ICD-9) from 1995 to 1999 and the C16 code of the tenth edition (ICD-10), from the year 2000 onwards. 97.9% of the records were filled out by health professionals.
The annual country population was obtained from the census projections of the National Institute of Statistics and Informatics19. The gross and specific rates expressed per 100,000 inhabitants were calculated with this data by gender, five-year age groups (except those under 40 and 80 and over), and geographic and political regions. The population estimated by the World Health Organization (WHO) was used as the reference population to calculate the age-standardized rates20
Statistical analysis
The temporal trend of the standardized mortality rates was analyzed using a segmented regression model, gender, age, political regions of origin (departments), and geographic (coast, mountains and jungle) were incorporated as independent variables, by considering the belonging of the majority of the population of a political region to a certain geographic area.
A joinpoint segmented regression model was fitted in order to identify the periods between the years 1995 and 2013 in which there were significant changes in the standardized mortality rate.
The model was adjusted under the hypothesis of the existence of a linear evolution of the natural logarithm of the expected standardized mortality rates and unrelated errors.
The method was selected“Grid search”, considering between 0 and 3 joinpoints to locate the joinpoints. As a summary measure, the annual percentage change (CPA, for its initials in Spanish) and the Average Annual Percentage Change (CPAP, for its initials in Spanish) were used. Unlike other trend studies conducted in Peru12,13, this model made it possible to identify the moment in which significant changes occur in the observed trend.
The evaluation of the goodness of fit of the model and the significance of the changes in the trend was carried out by using the classic Bayesian Information Criterion (BIC) and the parametric Student's t-test for a significance level of 5%.
To adjust the model, theJoinpoint Desktop Softwarev.4.5.0.0(Division of Cancer Control and Population Sciences, National Cancer Institute)was used. (21
A map was prepared from the standardized mortality rates (SMD) due to gastric cancer in Peru for 2013 with the rates estimated using the adjusted segmented regression model and they were ordered from highest to lowest in 4 groups, taking as reference the national standardized mortality rate (MNR) of 11.9 per 100,000 pop.
- Very high: when the TME > 150% TMEN,
- High: when the TME: 100 - 150% TMEN,
- Medium: when the TME: 50- 100% TMEN,
- Low: when the TME< 50% TMEN.
The map prepared from the TMEN softens the standardized mortality rates and allows better identification of vulnerable territories.
RESULTS
A total of 49,690 death records with basic cause of death from gastric cancer were analyzed in Peru from 1995 to 2013. In 1995, the standardized mortality rate was 16.1 per 100,000 inhabitants. In 2013 it decreased to 11.4 per 100,000 inhabitants, observing a statistically significant decreasing trend (p <0.05) and without change points or joinpoints, with a reduction of 2.3% per year (seeTable 1).
Table 1. Trends in age-standardized mortality rate due to gastric cancer by gender in Peru from 1995 to 2013. Segmented regression analysis.
| Group | TMEE ( x 100 000 inhab.) | CPA | CPA | Trend | Period | ||
| INITIAL | FINAL | 95% CI | t-test | p-value | |||
| Men | 18,22 | 20,79 | 6.8 | (-8,4; 24,6) | 1 | 0.355 | 1995-1997 |
| 20,79 | 14,82 | -5.1* | (-8,1; -1,9) | -3.7 | 0.002 | 1997-2003 | |
| 14,82 | 16,25 | 0.2 | (-2,1; 2,5) | 0.2 | 0.851 | 2003-2010 | |
| 16,25 | 13,25 | -6.2 | (-12,2; 0,3) | -2.2 | 0.1 | 2010-2013 | |
| Women | 0,41 | 0,32 | -2.5* | (-3,1;-1,9) | -8.3 | < 0,001 | 1995-2013 |
| Total | 16,09 | 11,41 | -2,3* | (-2,8; -1,8) | -8,9 | < 0,001 | 1995-2013 |
TMEE, for its initials in Spanish: Age-standardized mortality rate, estimated in the model. CPA: Annual percentage change. *Statistically significant trend
Mortality trends standardized by gender
In men the standardized mortality trend was changing: the optimal segmented regression model showed three joinpoints (BIC: 1,72) which occur in: 〖〖〖(x〗_i-τ〗_k)〗^+=3 ; τ_1=1997, τ_2=2003 y 〖 τ〗_3=2010;, and determine 4 segments: a) period 1995-1997: increasing trend (CPA 6.8%), not statistically significant b) period 1997-2003, decreasing trend (CPA -5.1%), statistically significant (p value <0 , 05), c) period 2003-2010, slightly increasing trend (CPA 0.2%) and not statistically significant and d) period 2010-2013, decreasing trend (CPA -6.2%), but not statistically significant (seeTable 1,Figure 1).

Figure 1. Comparative gastric cancer mortality trend by gender in Peru 1995-2013, segmented regression models.
The optimal segmented regression model for the standardized mortality trend in women was decreasing and statistically significant, with a decrease of 2.5% per year, and does not contain joinpoints (BIC = 2.05). There is no parallelism between men and women (p-value = 0.05). (SeeFigure 1).
Standardized mortality trends by age group
When standardized mortality trends are compared according to age groups and a statistically significant decreasing trend observed, three joinpoints are detected: for the group of 60 to 64 years in the years 1997, 2007 and 2010 and another three; for the 65 to 69 age group in the years 1997, 2003 and 2009. It is observed that the CPA decreases as the age group increases. For example, for the 40-44-year-old group, it was -1.39% annually, while for the 75-79-year-old age group, it was -2.57% annually (seeTable 2).
Table 2. Trends in TMEE due to gastric cancer by age group in Peru 1995 - 2013. Segmented regression analysis.
| Age group | TMEE ( x 100 000 inhab.) | CPA | CPA | Period | ||
| INITIAL | FINAL | 95% IC | ||||
| < 40 years | 0,61 | 0,42 | -2,07* | (-3,1 -1,0) | 1995-2013 | |
| 40 - 44 years | 0,41 | 0,32 | -1,39* | (-2,3-0,4) | 1995-2013 | |
| 45 - 49 years | 0,62 | 0,43 | -2,00* | (-2,9-1,1) | 1995-2013 | |
| 50 - 54 years | 0,94 | 0,65 | -2,06* | (-3,0-1,1) | 1995-2013 | |
| 55 - 59 years | 1,22 | 0,82 | -2,19* | (-2,8-1,6) | 1995-2013 | |
| 60 - 64 years | 1,47 | 1,71 | 8,9 | (-17,2-43,2) | 1995-1997 | |
| 1,71 | 1,15 | -3,7* | (-6,0-1,4) | 1997-2007 | ||
| 1,15 | 1,41 | 5,1 | (-18,5-35,6) | 2007-2010 | ||
| 1,41 | 0,89 | -13,1* | (-24,1-0,4) | 2010-2013 | ||
| 65 - 69 years | 1,88 | 2,45 | 13,98 | ( 1,3- 0,2) | 1995-1997 | |
| 2,45 | 1,52 | -7,60* | (-3,6-0,0) | 1997-2003 | ||
| 1,52 | 1,72 | 2,01 | ( 0,9- 0,4) | 2003-2009 | ||
| 1,72 | 1,26 | -7,45* | (-2,6- 0,0) | 2009-2013 | ||
| 70 - 74 years | 2,52 | 1,63 | -2,39* | (-3,1-1,7) | 1995-2013 | |
| 75 - 79 years | 2,67 | 1,67 | -2,57* | (-3,3-1,9) | 1995-2013 | |
| > 80 years | 4,98 | 3,72 | -1,60* | (-2,2-1,0) | 1995-2013 | |
| Total | 18,06 | 11,88 | -2,3* | (-2,8 -1,8) | 1995-2013 | |
TMEE: Age-standardized mortality rate, estimated in the model CPA: Annual Percentage Change *Statistically significant trend
Mortality rates standardized by regions
The highest mortality from gastric cancer in 2013 is concentrated in the central Andes, where the regions of Huancavelica (3.5 times the TMEN), Huánuco (2.5 times the TMEN), Ayacucho and Pasco have the highest mortality, and Junín tops the higher mortality regions. Also, two other high mortality zones are identified: the first in the north of the country (Lambayeque, La Libertad, and Cajamarca) and the second in the central coast (Ica, Callao, Ancash) (seeTable 3,Figure 2).
Table 3. Trends standardized mortality rates for gastric cancer by political regions in Peru from 1995 to 2013. Analysis of segmented regress.
| Region | TMEE ( per 100 000 inhab.) | CPA | CPA | Period | CPA | ||||
| INITIAL | FINAL | 95% IC | |||||||
| Amazonas | 10,85 | 21,74 | 8,0* | ( 0,0, 16,7) | 1995-2004 | -0,9 | |||
| 21,74 | 5,47 | -29,2 | (-54,3, 9,7) | 2004-2008 | |||||
| 5,47 | 9,22 | 11 | (-10,4, 37,4) | 2008-2013 | |||||
| Ancash | 15,4 | 11,98 | -1,4 | (-3,8, -1,1) | 1995-2013 | -1,4 | |||
| Apurímac | 7,72 | 17,24 | 9,3 | (-1,5, 21,3) | 1995-2004 | 0,6 | |||
| 17,24 | 8,64 | -7,4 | (-16,1, 2,3) | 2004-2013 | |||||
| Arequipa | 12,38 | 8,74 | -4,8 | (-10,1, 0,7) | 1995-2002 | -3,8 | |||
| 8,74 | 11,35 | 5,4 | (-7,9, 20,5) | 2002-2007 | |||||
| 11,35 | 6,16 | -9,7* | (-15,4, -3,5) | 2007-2013 | |||||
| Ayacucho | 10,17 | 19,74 | 3,8* | (1,3, -6,3) | 1995-2013 | 3,8* | |||
| Cajamarca | 15,43 | 22,06 | 4,1 | (-1,6, 10,0) | 1995-2004 | -1,7 | |||
| 22,06 | 12,12 | -7,2* | (-11,9, -2,3) | 2004-2013 | |||||
| Callao | 18,05 | 12,46 | -2,0* | (-3,5, -0,6) | 1995-2013 | -2,0* | |||
| Cusco | 14,42 | 5,97 | -4,8 | (-7,1, -2,4) | 1995-2013 | -4,8 | |||
| Huancavelica | 7,91 | 37,26 | 24,8* | (9,8, 41,8) | 1995-2002 | 9,7* | |||
| 37,26 | 41,65 | 1 | (-3,1, 5,3) | 2002-2013 | |||||
| Huánuco | 33,13 | 44,37 | 2,5 | (-0,6, 5,6) | 1995-2007 | -0,7 | |||
| 44,37 | 29,35 | -6,7 | (-13,4,0,7) | 2007-2013 | |||||
| Ica | 22,09 | 13,88 | -2,6* | (-4,0, -1,0) | 1995-2013 | -2,6* | |||
| Junín | 24,19 | 29,05 | 3,7 | (-7,9, 16,9) | 1995-2000 | -2,9 | |||
| 29,05 | 18,43 | -8,7 | (-22,5,7,6) | 2000-2005 | |||||
| 18,43 | 27,73 | 8,5 | (-6,7,26,2) | 2005-2010 | |||||
| 27,73 | 14,21 | -20 | (-38,2,3,6) | 2010-2013 | |||||
| La Libertad | 18,41 | 23,13 | 7,9 | (-9,1, 28,0) | 1995-1998 | -1,6 | |||
| 23,13 | 13,8 | -3,4* | (-4,6,-2,2) | 1998-2013 | |||||
| Lambayeque | 16,04 | 18,57 | 5 | (-27,4, 51,9) | 1995-1998 | -0,7 | |||
| 18,57 | 2,92 | -18,6* | (-26,8,-9,5) | 1998-2007 | |||||
| 2,92 | 13,17 | 65,1 | (-44,8,393,9) | 2007-2010 | |||||
| 13,17 | 14,18 | 2,5 | (-23,7,37,7) | 2010-2013 | |||||
| Lima | 21,54 | 12,96 | -4,1* | (-5,5, -2,7) | 1995-2007 | -3,3 | |||
| 12,96 | 15,29 | 5,7 | (-15,2,31,7) | 2007-2010 | |||||
| 15,29 | 11,81 | -8,2 | (-17,7,2,3) | 2010-2013 | |||||
| Loreto | 13,77 | 3,28 | -7,7* | (-10,7, -4,5) | 1995-2013 | -7,7* | |||
| Madre de Dios | 23,22 | 7,84 | -5,9* | (-11,5, 0,1) | 1995-2013 | -5,9* | |||
| Moquegua | 15,35 | 5,69 | -5,4* | (-8,4, -2,3) | 1995-2013 | -5,4* | |||
| Pasco | 24,55 | 53,97 | 48,3 | (-17,3,165,8) | 1995-1997 | -1,3 | |||
| 53,97 | 26,43 | -21,2 | (-55,6,39,9) | 1997-2000 | |||||
| 26,43 | 19,23 | -2,4 | (-5,3,0,5) | 2000-2013 | |||||
| Piura | 14,87 | 19,32 | 14 | (-31,7,90,1) | 1995-1997 | -3,3 | |||
| 19,32 | 7,26 | -17,8* | (-31,9,-0,8) | 1997-2002 | |||||
| 7,26 | 8,09 | 1 | (-2,8,5,0) | 2002-2013 | |||||
| Puno | 10,2 | 4,96 | -3,9* | (-5,7, -2,1) | 1995-2013 | -3,9* | |||
| San Martín | 17,46 | 16,81 | -0,3 | (-3,3,2,7) | 1995-2006 | -3,4* | |||
| 16,81 | 9,42 | -7,9 | (-13,0,-2,6) | 2006-2013 | |||||
| Tacna | 10,61 | 11,19 | 0,4 | (-2,9,3,8) | 1995-2009 | -4,9 | |||
| 11,19 | 4,29 | -21,3 | (-39,1,1,8) | 2009-2013 | |||||
| Tumbes | 18,45 | 10 | -3,4* | (-6,3, -0,3) | 1995-2013 | -3,4* | |||
| Ucayali | 18,22 | 21,84 | 1,3 | (-2,1, 4,8) | 1995-2009 | -5,4* | |||
| 21,84 | 6,73 | -25,51 | (-41,2, 5,7) | 2009-2013 | |||||
| Total | 18,06 | 11,88 | -2,3* | (-2,8, -1,8) | 1995-2013 | -2,3* | |||
TMEE: Age-standardized mortality rate, estimated in the model CPA: Annual percentage CPAP: Average annual percentage change *Statistically significant trend
Mortality trends standardized by political regions
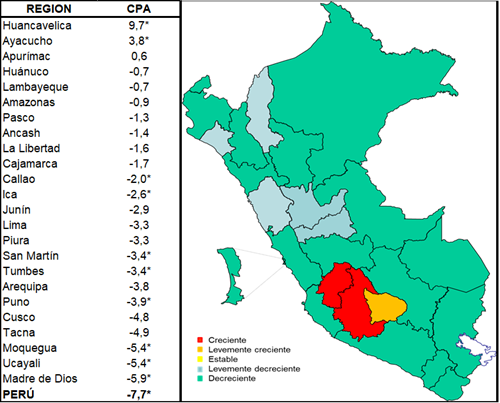
Using the interpretation of the annual percentage change proposed by SERGAS22, the regions were classified according to their estimated standardized mortality trend (using the adjusted segmented regression model) as increasing (CPA ≥1.5%), slightly increasing (0, 5% ≤ CPA <1.5%), stable (0.5% < CPA < 0.5%), slightly decreasing (-1.5% < CPA ≤ -0.5%) and decreasing (CPA ≤ -1, 5%). The case of the regions where no joinpoint(s) were found, the CPA value was used. The Average Annual Percentage Change (CPAP) was used in the regions that had one or more joinpoint.
The map shows a growing trend in 2 out of 25 regions, Huancavelica and Ayacucho (CPA: 9.7% and 3.8% respectively). In the Apurímac region a slightly increasing trend is observed (CPA: 0.6%). In 5 regions, the trend is slightly decreasing: Huánuco (CPA: -0.7%), Lambayeque (CPA: -0.7%), Amazonas (CPA: -0.9%), Pasco (CPA: -1.3 %) and Ancash (CPA: -1.4%). in the rest of the 17 out of 25 regions, a decreasing trend is observed (CPA≤1.5%) (seeTable 3,Figure 3).
In general, in the central Andes area, made up of the Huancavelica, Ayacucho and Apurímac regions, gastric cancer is growing and constitutes the area of greatest risk to die from this disease. The regions with slow decrease are observed in the corridor constituted by the Ancash, Huánuco and Pasco regions, and are also observed in two focal regions with slow decline: Lambayeque and Amazonas (seeFigure 3).
Table 3shows the results of the fit of the segmented regression model with the standardized mortality rates estimated at the beginning and at the end of each period of time determined by thejoinpoint (s). CPAs are presented for each time segment estimated by the model and for the entire period (CPAP). Are joinpoint (s) to the trend of gastric cancer standardized mortality in the period 1995 to 2013 in 15 of the 25 regions.
Mortality trends standardized by natural regions
It is observed that the Coast has a growing trend until 1998 (CPA: 1%); then a decreasing one until 2003 (CPA: -6.4%) and then slightly decreasing (CPA: -0.9%). The Peruvian Highlands showed a growing trend until 1997 (CPA: 12.9%). Then it remains stationary until 2009 (CPA: 0%) when it decreases (CPA: -8.3%). The Peruvian Jungle has a decreasing trend until 2009 (CPA: -2.5%) when it becomes more marked (CPA: -12%).
In 1996, the Highlands went from third to second place and it ranks first in mortality from gastric cancer since 1999; the coast that occupied the first place, occupies the second place since 1999, and the Jungle occupies the third place since 1996.
DISCUSSION
For the year 2012, the standardized gastric cancer mortality rate in developed countries was 9.2 per 100,000 inhabitants and for developing countries, it was 14.4 per 100,000 inhabitants.6. Peru has experienced in recent years a reduction of 2.3% annually. Under the model studied, it comes closer to developed countries (11.6 per 100,000 inhabitants), occupying in the region of the Americas the 6th place, surpassed by Guatemala, Ecuador, Honduras, Chile and El Salvador. (23
An investigation of the standardized gastric cancer mortality rates in Peru from 2005 to 2014 carried out by Hernández-Vásquez et al., shows a reduction from 15.49 to 12.13 per 100 000 inhabitants. (12). The disadvantage of this graphic analysis is the insufficiency to study the temporal evolution of numerical series since it does not allow characterizing the phenomenon due to the distortions presented by the annualized data, which is eliminated by using generalized linear models that allow a more objective analysis. A longer series from 1986 to 2015 on gastric cancer mortality in Peru shows a reduction of 40%, with a constant rate of 2% per year24. However, this trend was calculated by adjusting log-linear models with the Poisson distribution. In these models a canonical link function is used, and the data is aggregated, stratified by age groups and year of death. Unlike these studies, our work analyzes the logarithm of the standardized rates using linear regression models by segments that have the advantage of conducting a study stratified by subgroups and identifying sudden changes in the trend.
In Latin America, a decreasing trend is observed in gastric cancer mortality25. In Peru this decreasing trend is similar to the ones observed in Asian or Eastern European countries26. Decreasing trends in stomach cancer mortality have also been documented in regions with historically high rates, including countries in Asia (Japan, China, and Korea)3, Latin America25and Europe (Ukraine) (3. The factors that have contributed to this decline are attributed to the increased availability of fresh produce, fruits and vegetables, less dependence of canned food on salinity, and the reduction in chronic H. pylori infection due to better sanitation and antibiotics10.
In Peru, the decrease in mortality from this cancer, according to various authors, could be attributed to improvements in the population's living conditions, such as the use of safe water, provision of adequate sanitation or sewerage systems, and cooling of foods15, factors that contribute to reduce the transmissibility ofHelicobacter pylori, more than the medical progress aimed at treating gastric cancer cases, since there is a huge gap in cancer services in the country8.
Since 2001, Kaneko and Yoshimura postulate that the decrease in intestinal-type gastric cancer incidence is the factor that most contributes to the decrease in the incidence of gastric cancer worldwide27. However, Henson et al. (2004), point out that the incidence of diffuse-type gastric carcinoma, particularly the signet ring type, has been increasing28.
ChronicH. pyloriinfection is the identified risk factor with strongest association to stomach cancer, with approximately 90% of new cases of non-cardiac gastric cancer worldwide cases11. Ramirez Ramos et al. (2003) conducted a study of variation in the prevalence of infectionH. pyloriin Peru between 1985 and 2002, with reference to the population of a clinic in Lima Metropolitana which was considered medium and high socioeconomic. It was found that in 1,260 patients with active chronic gastritis a prevalence of H. pylori that decreased from 83,3% to 58,7% (p-value <0,001), being more significant in those under 30 years of age. In 178 patients with duodenal ulcer it decreased from 89,5% to 71,9% (p value = 0,004), but there were no statistically significant differences in patients with gastric ulcer or normal mucosa29. In general, is observed a decrease in the prevalence of H. pylori in the different Peruvian studies carried out30.
52.1% of deaths from gastric cancer in Peru occurred in men, which is lower than that reported worldwide. In 2012, it was estimated that 64.9% of deaths from gastric cancer occurred in men and developing countries reported 66,1%6. A recent study on the trend in gastric cancer mortality in Peru from 2003 to 2016, which uses the classic non-parametric Mann-Kendall trend test to check whether there is a trend in the time series, shows a decrease in the proportion of Deaths attributed to gastric cancer in those over 50 years statistically significant for men from 25.7% to 21.8% and for women from 24,4% to 16,44%.31. Another study analyzed the histological type of 3568 patients with gastric cancer registered in the National System of Epidemiological Surveillance of cancer of the Ministry of Health between the years 2009-2010 and 43.2% were of unspecified type, 33.6% of intestinal type (the most related to Helicobacter pylori) and 18,7% of diffuse type. A higher prevalence of the diffuse type is observed in women, 21.6% versus 16% in men, and in general, a higher percentage compared to that reported in the literature, which could explain why gastric cancer in Peru occurs in a almost 1:1 ratio of men to women. (14
This study finds an increase in the gastric cancer mortality rate adjusted by age group as it increases (seeTable 1), as reported in the literature. In the age segment of 40-44 years for the Americas region in 2012 the rate was 3.9 per 100,000 inhabitants. and it increases to 64.9 per 100,000 inhabitants. for the age group from 70 to 74 years. (23
Higher gastric cancer mortality in Peru is concentrated in the central Andes, this scenario is constituted by the regions of Huancavelica, Huánuco, Ayacucho, Pasco and Junín. The second high mortality scenario is found in the north of the country (Lambayeque, La Libertad and Cajamarca), the third in the central coast (Ica, Callao, Ancash and Lima) (seeFigure 2). Piñeros et al. (2017) have also noted this pattern that attests a positive correlation between altitude and gastric cancer. However, it has been suggested that altitude is a proxy for factors that could be grouped into mountainous regions- for example, host genetic, bacterial, dietary, and environmental factors32. Local studies in Lima reported a positive association between ethnic descent and stomach cancer, and a predominant role for socioeconomic factors associated with ethnicity and disparities in access to health services33.
The United Nations Development Programme (2013) reports shows that Peru reached an HDI of 0,74 and regions with the lowest HDI were Huancavelica with 0,297; then Ayacucho with 0.334, Apurimac with 0.344 and Huánuco with 0,37434. This finding postulates the hypothesis that poor conditions of poverty, education, and sanitation would contribute to higher mortality from gastric cancer, possibly due to greater transmissibility and chronicity ofH. pyloriinfection; as well as, less access to health services. Bray also points out this in the publication of global cancer trends for the period 2008-203035.
Regarding the socio-economic level and after reviewing studies in various care centers for patients of socioeconomic level medium and high Ramírez Ramos et al. (2006) points out that there is a significant decrease in the prevalence ofH. Pylori(45%) compared to 80% observed a decade ago29.
One of the most important factors for the decreasing trend in gastric cancer mortality in Peru could be the substantive improvement in living conditions that reduces the transmissibility of infection by helical pylori: a) between 2001 and 2014, the percentage of rural households with drinking water in their home from 35,1 to 61,8%.15, b) A reduction of 12.4 percentage points in the number of homes with poor excreta disposal from 2001 to 2014 is documented, being more substantive at the rural level. It was significantly reduced from 49.8 to 24% 15.
However, from 1995 to 2015 the standardized mortality rate reported by the Ministry of Health of Peru increased from 18.4 per 100,000 inhabitants at the rural level to 30.9 per 100,000 inhab. While in urban areas it was reduced from 32.3 per 100,000 inhabitants to 17.7 per 100,000 inhab. (24. This migration of mortality from gastric cancer could be due to problems of timely access to health services, since cancer treatment centers are all located in urban areas. As well as access to diagnosis by endoscopy, although it is not very widespread, it is carried out almost entirely in urban areas. (8In accordance with what was reported by Pereira et al., Who pointed out that the rural area encompasses socioeconomic factors associated with ethnicity and disparities in access to health services33.
In the present study it is also observed changes in gastric cancer mortality trends by natural regions that are in accordance with rurality, since the mountains and jungle concentrate the regions with the highest rurality with respect to the coast. This explains why in 1995 the Coast had the highest mortality and in 2013 the Highlands was the one with the highest mortality, it is important to propose targeted socio-sanitary studies to test the hypothesis regarding whether the lower migration of the inhabitants of rural areas of the sierra, genetic and environmental conditions and inequities in access to health services explain the higher mortality from gastric cancer.
LIMITATIONS
One of the main limitations of the research is the under-reporting of the
of deaths of vital statistics of the Ministry of Health. Another limitation is the period analyzed between 1995 to 2013, since it is possible that there are changes in the trends of the successive years, however, a great amount of years is required to have the consistent final data and to be incorporated into the segmented regression model.
CONCLUSION
The trend in stomach cancer mortality in Peru is decreasing and statistically significant for the period 1995 - 2013 (-2.3% per year). Different behavior due to sex, age groups, political and geographical groups are observed. The departments with an increasing mortality trend are Huancavelica, Ayacucho and Pasco in the central Andes. A greater reduction in SMD is observed in women compared to men.
REFERENCES
1. Crew K, Neugut A. Epidemiology of gastric cancer. World Journal of Gastroenterology. 2006 January; 12(3): p. 354-362. [ Links ]
2. Malvezzi M, Bonifazi M, Bertuccio P, Levi F, La Vecchia C, Decarli A, et al. An age-period-cohort analysis of gastric cancer mortality from 1950 to 2007 in Europe. Annals of Epidemiology. 2010 December; 20(12): p. 898 - 905. [ Links ]
3. Bertuccio P, Chatenoud L, Levi F, Praud D, Ferlay J, Negri E, et al. Recent patterns in gastric cancer: a global overview. International Journal of Cancer. 2009 August 1; 125(3): p. 666 - 673. [ Links ]
4. Ferlay J, Colombet M, Seorjomataram I, Mathers C, Parkin DM, Piñeros M, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2019; 144(8): p. 1941 - 1953. [ Links ]
5. IARC. GLOBOCAN 2018. (Online).; 2020. Available from: https://gco.iarc.fr/today [ Links ]
6. Torre L, Bray F, Siegel R, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA: A Cancer Journal for Clinicians. 2015 march/april; 65(2): p. 87-108. [ Links ]
7. Oficina General de Epidemiología. La priorización del cáncer en el Perú. Documento de trabajo. Lima: Ministerio de Salud; 2001. [ Links ]
8. Ramos W, Venegas D, Medina J, Guerrero P, Cruz A. Análisis de la Situación de Cáncer en el Perú 2013 Epidemiología DGd, editor. Lima: Ministerio de Salud; 2013. [ Links ]
9. Correa P, Houghton JM. Carcinogenesis of Helicobacter pylori. Gastroenterology. 2007 August; 133(2): p. 659-672. [ Links ]
10. Parkin D. The global health burden of infection-associated cancers in the year 2002. International Journal of Cancer. 2006 June 15; 118(12): p. 3030 - 3044. [ Links ]
11. Plummer M, Franceschi S, Vignat J, Forman D, De Martel C. Global burden of gastric cancer attributable to pylori. International Journal of Cancer. 2015 January 15; 136(2): p. 487 - 490. [ Links ]
12. Hernández-Vásquez A, Bendezú-Quispe G, Azañedo D, Huarez B, Rodríguez-Lema B. Temporal trends and regional variations in gastrointestinal cancer mortality in Peru, 2005-2014. Rev Gastroenterol Peru. 2016; 36(4): p. 320-9. [ Links ]
13. Ruiz EF, Torres-Roman S, Servan SA, Martínez-Herrera JF, Arce-Huamani MA, Carioli G, et al. Trends and geographic pattern of stomach cancer mortality in Peru. Cancer Epidemiology. 2018. [ Links ]
14. Alvarado C, Venegas D. Survival rate of patients with stomach cancer in Peru, 2009-2010. Pan American Journal of Public Health. 2015 March; 37(3): p. 133-139. [ Links ]
15. Instituto Nacional de Estadística e Informática. Perú: Encuesta Nacional de Hogares Lima: INEI; 2010. [ Links ]
16. Kafle RC. Trend Analysis and Modeling of Health and Environmental Data: Joinpoint and Functional Approach Florida: University of South Florida; 2014. [ Links ]
17. Jiang Z, Qiu Z, Hatcher J. Joinpoint trend analysis of cancer incidence and mortality using Aberta data. Canada:; 2010. [ Links ]
18. Organización Panamericana de la Salud. Lineamientos básicos para el análisis de la mortalidad Washington, DC: OPS; 2017. [ Links ]
19. Instituto Nacional de Estadística e informática. Boletín Especial N° 17. Perú: Estimaciones y Proyecciones de Población Total, por Años calendario y Edades Simples 1950 - 2050 INEI , editor. Lima: INEI; 2009. [ Links ]
20. Ahmad OB, Boschi-Pinto C, Lopez AD, Murray CJ, Lozano R, Inoue M. Age standardization of rates: a new WHO standard. DPE Discuccion paper series: N°31. (Online).: World Health Organization; 2001. Available from: http://www.who.int/healthinfo/paper31.pdf [ Links ]
21. National Cancer Institute. (Joinpoint Help Manual 4.5.0.0.).; 2017 (cited 2017 may 16. Available from: https://surveillance.cancer.gov/help/joinpoint/ [ Links ]
22. SERGAS. La mortalidad por cáncer de esófago en galicia, de 1980 a 2001. Boletin epidemiológico de Galicia. 2005 abril; XVII(6). [ Links ]
23. GLOBOCAN. GLOBOCAN 2012, IARC. (Online).; 2012 (cited 2016 10 15. Available from: http://globocan.iarc.fr/old/age-specific_table_r.asp?selection=219992&selection=224900&title=WHO+Americas+region+%28PAHO%29%2C+World&sex=0&type=1&stat=0&window=1&sort=1&submit=%C2%A0Execute [ Links ]
24. Ministerio de Salud del Perú. Análisis de las Causas de Mortalidad en el Perú, 1986 - 2015 LIma: Centro Nacional de Epidemiología, Prevención y Control de Enfermedades; 2018. [ Links ]
25. Carioli G, La Vecchia C, Bertuccio P, Rodríguez T, Levi F, Boffetta P, et al. Cancer mortality predictions for 2017 in Latin America. Annals of Oncology. 2017; 28: p. 2286-2297. [ Links ]
26. Ang TL, Fock KM. Clinical epidemiology of gastric cáncer. Singapore Med J 2014. 2014; 55(12): p. 621-628. [ Links ]
27. Kaneko S, Yoshimura T. Time trend analysis of gastric cancer incidence in Japan by histological types, 1975-1989. British Journal of Cancer. 2001; 84(3): p. 400 - 405. [ Links ]
28. Henson D, Dittus C, Younes M, Nguyen H, Albores-Saavedra J. Differential trends in the intestinal and diffuse types of gastric carcinoma in the United States, 1973-2000: increase in the signet ring cell type. Archives of Pathology & Laboratory Medicine. 2004 July; 128: p. 765 - 770. [ Links ]
29. Ramírez-Ramos A, Sánchez-Sánchez R. Contribución de Latinoamérica al estudio del Helicobacter pylori. Acta Gastroenterol Latinoam. 2009; 39(3): p. 197-218. [ Links ]
30. Prochazka Zarate R, Salazar Muente F, Barriga Calle E, Salazar Cabrera F. Prevalencia de Helicobacter pylori en una clínica privada de Lima: sensibilidad de las biopsias del antro y el cuerpo, y la prueba rápida de la ureasa. Rev. Gastroenterol. Perú. 2010; 30(1): p. 33-39. [ Links ]
31. Zafra-Tanaka JH, Tenorio-Mucha J, Villarreal-Zegarra D, Carrillo-Larco R, Bernabe-Ortiz A. Cancer-related mortality in Peru: Trends from 2003 to 2016. PLoS ONE. 2020; 2(e0228867). [ Links ]
32. Piñeros M, Ramos W, Antoni S, Abriata G, Medina L, Miranda J, et al. Cancer patterns, trends, and transitions in Peru: a regional perspective. Lancet Oncol. 2017; 18: p. e573-86. [ Links ]
33. Pereira L, Zamudio R, Soares-Souza G, Herrera P, Cabrera L, Hooper C, et al. Socioeconomic and nutritional factors account for the association of gastric cancer with Amerindian ancestry in a Latin American admixed population. PLoS One. 2012; 7(8): p. e41200. [ Links ]
34. Naciones Unidas. Los datos demográficos. Alcances, limitaciones y métodos de evaluación. Serie Manuales - CEPAL N° 82 Santiago de Chile: Naciones Unidas; 2014. [ Links ]
35. Bray F, Jemal A, Grey N, Ferlay J, Forman D. Global cancer transitions according to the Human Development Index (2008-2030): a population-based study. Lancet Oncol. 2012; 13(8): p. 790-801. [ Links ]
36. Murray Cea. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. The Lancet. 2012 december; 380(9859): p. 2197-2223. [ Links ]
37. Howson C, Hiyama T, Wynder E. The decline in gastric cancer: epidemiology of an unplanned triumph. Epidemiologic Reviews. 1986;: p. 1-27. [ Links ]
38. Ramírez Ramos A. Helicobacter pylori. In Interna SPdM. Topicos Selectos en Medicina Interna: Gastroenterología. LIma: Sociedad Peruana de Medicina Interna; 2006. p. 177-195. [ Links ]
Received: November 15, 2020; Accepted: December 30, 2020











 text in
text in