INTRODUCCIÓN
Cacao (Theobroma cacao L.) is a crop of great economic importance in Peru and the world. Peru is one of the main producers and suppliers of fine aroma cacao and the second producer of organic cocoa worldwide, and the crop that is the main source of income to 90000 farming families in the Peruvian Amazon (MINAGRI, 2018). The Amazonas department is the main producer of this crop, and in the provinces of Bagua and Utcubamba a native fine aroma cacao known as “Cacao Amazonas Peru” is produced (Díaz-Valderrama et al., 2020). Therefore, it is very important to know the beneficial microbial communities found in the soil and their relationship with the plant.
Specific groups of microorganisms establish beneficial relationships with plants in the soil-root interface that can improve plant growth and development, such as the arbuscular mycorrhizal fungi (AMF). This plant-microorganism relationship is essential for the development of both organisms that form a mutualistic symbiotic association (mycorrhiza) with approximately 90% of terrestrial plants (Camarena-Gutierrez et al., 2012; Camargo-Ricalde et al., 2012; Tedersoo et al., 2020). The AMF produces an extensive extra-radical mycelium that penetrates the root of plants, connecting the plant and the soil. This association benefits the plant in several ways: increases the absorption of low-mobility nutrients such as phosphorus, enhances the production of growth hormones, and AMF secret antibiotics, protects the plant against several soilborne pathogens, and aggregates soil particles for a better root performance (Klironomos, 2003; Neuenkamp et al., 2019). Therefore, this mycorrhizal association plays an important role in the growth and nutrition of higher plants.
Cacao is highly colonized by AMF that influences nutrient absorption, tolerance to water stress and improvements to adverse biotic and abiotic conditions (Rojas-Mego et al 2014; Rincón et al., 2021; Flores de Valgaz et al., 2022). The objective of this study is to identify the AMF in native cacao plantations in the provinces of Bagua and Utcubamba, Amazonas.
MATERIAL Y MÉTODOS
Sampling
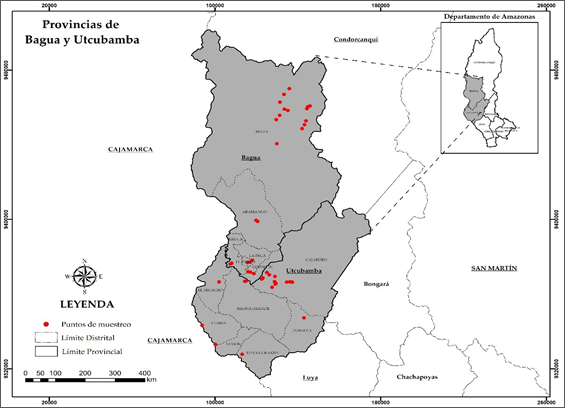
We collected rhizospheric soil samples in forty-four native cacao plots in the provinces of Bagua (24 plots) and Utcubamba (20 plots), Amazonas department (Figure 2). Sampling was performed following previously described protocols by Bernier (Bernier, 1999; Mendoza & Espinoza, 2017; Murrieta & Palma, 2018) with some modifications. Five cacao plants per plot were randomly selected. In each plant, we obtained three subsamples at 0-20 cm depth and one meter apart from the base of each plant. Subsamples were homogenized, taking a single composite sample, of approximately 1 kg (Figure 1). The samples were coded with the following information: altitude, latitude, name of the producer, GPS coordinates of the plot, age of the plantation and the cultivated area. Subsequently, samples were stored in thermal boxes for transport to the laboratory, and once there, samples were stored in large trays and at room temperature for drying until further processing.
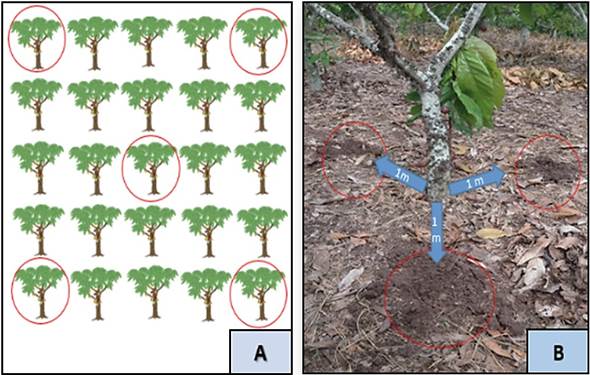
Figure 1. Sampling strategy. (A) Random selection of cacao plants in each plot (B) Collection of three rhizospheric soil subsamples per plant.
AMF isolation
For the extraction and isolation of AMF spores, a sieving and decanting methodology, and a centrifugation protocol in sucrose (Daniels & Skipper, 1982) previously described was performed with some modifications. Sucrose was diluted at 20% and 60% w/v under heat and preserved in glass containers. We weighed 100g of soil, previously dried and ground, and dissolved in two liters of water. This suspension was shaken for 10 seconds and left to stand for 20 seconds to eliminate large particles by sedimentation. Then the suspension was passed through 250- and 38-µm sieves, consecutively, repeating this operation four times. Subsequently, the content retained in the 38-µm sieve was transferred to a 50 ml falcon tube with sucrose at 20% and 60% (15 ml each) and centrifuged at 3500 rpm for 4 minutes. The supernatant was decanted, and the samples washed with a 38-micron sieve until the sucrose was removed. Finally, the content was placed in a Petri dish to be observed under a stereoscope (Zeiss brand, Discovery V8 model).
AMF identification
We used the taxonomic keys described by the International Culture Collection of Vesicular Arbuscular Mycorrhizal Fungi (INVAM) (Shenck & Perez, 1990). Spores were grouped according to their size, color and shape (morphotypes), 5-10 spores were mounted in polyvinyl alcohol plus lactoglycerol (PVLG). By gently pressure applied with a coverslip, the spore hatching took place. Then, we let the sample dry at room temperature, and fixed them. Spores were observed and photographed under a Leica optical microscope at 20x magnification and measured using the Image Tool software (Krita Manual v.4.3.0). The morphological characteristics allowed us to make comparisons with taxonomic keys (Shenck & Perez, 1990). The morphological characteristics considered were size, (mean spore diameter), color (hyaline, yellow, red, black, honey, pink, etc.) and shape (round, spherical, oval, irregular, ellipsoid, subglobose, etc.).
Soil physicochemical analysis
Twelve rhizospheric soil samples were analyzed, distributed by districts, thus having six samples per province. The determination of pH and electrical conductivity (EC) was determined by the methodology of 1:1 ratio (weight/volume), where 20 g of soil is diluted in 20 ml of distilled water, measuring the electrical conductivity with a conductivity meter (White, 1969). Soil organic matter (OM) was quantified by the procedure of Walkley and Black (1934). Phosphorus (P) was extracted with Sodium Bicarbonate NaHCO3 (pH = 8.5) and determined by the Modified Olsen methodology and finally the determination of soil texture was determined with the Bouyoucos hydrometer (Bouyoucos, 1962).
RESULTS AND DISCUSSION
We identified 57 different morphotypes, classified into 9 genera (Table 1): Glomus, the most abundant genus with 30 different morphotypes (Figure 3 and 4), followed by Acaulospora with 14 morphotypes (Figure 5), Gigaspora with 3 morphotypes (Figure 6). The genera Funneliformis (Figure 7), Rhizophagus (Figure 8), Scutellospora (Figure 9), Sclerocystis (Figure 10) all were represented by two morphotypes each. Finally, the genera Diversispora (Figure 11) and Rhizoglomus (Figure 12) had one morphotype. The most frequent genera were Glomus and Acaulospora with 53% and 25%, respectively, followed by Gigaspora with 5%. All the other genera found had less than 5% frequency (Table 1). These results agree with a previous study (Rojas, 2010), that identified 21 morphotypes of AMF in the rhizosphere of cacao under two production systems in the department of San Martín, Peru. Rojas (2010) found Glomus was the most predominant AMF genera with 17 morphotypes and Acaulospora with 4 morphotypes.
Similarly, in Yurimaguas, department of Loreto, another study (Ruiz, 1992) identified spores of Glomus, Acaulospora, Gigaspora, Scutellospora and Entrophospora, being the most predominant Glomus and Scutellospora. The dominance of the Glomus genus in the mycorrhizal composition of the Amazon trapezium and the presence of Acaulospora associated with more acidic soils was also reported (Arcos, 2003). Similarly, another study reported the AMF in five cacao agroforestry systems from the Ecuadorian humid tropics, finding four genera: Glomus, Acaulospora, Gigaspora and Scutellospora, with the Glomus genus having greater representation and number of spores in all the sampled sites, followed by Gigaspora (Prieto-Benavides et al., 2012). Additionally, twelve genera of AMF (Glomus, Acaulospora, Ambispora, Archeospora, Cetraspora, Clareideoglomus, Diversispora, Fuscutata, Kuklospora, Pacispora, Paraglomus, and Sclerocystis) were identified in three cacao agroecosystems in the department of Ucayali, in the Peruvian Amazon (Rojas-Mego et al, 2014). Four of these genera were obtained in this study. This speaks of their wide distribution in the Amazonian trapezium and their relationship with the cultivation of native cacao. However, another study analyzed the AMF of wild cacao in the departments of Ucayali and Madre de Dios, Peru, and identified only seven morphotypes of the genus Glomus (Arévalo-Hernández, 2016), much less than the number of morphotypes identified in this study. This could have been due to the sampling was carried out in a dry season, causing the spores to remain dormant until they find adequate humidity conditions to sporulate, which made it difficult to identify more genera in this highly diverse ecosystem. Finally, Hernandez & Monroy (2017) identified eleven morphotypes AMF associated with three clones of cacao in Yopal, Casanare, Colombia, which were classified in the genera Glomus and Acaulospora, being Glomus the most abundant. The higher number of morphotypes found in our study could have been due to the agroecosystem and the variety of the cacao plantations.
The concentration of the physicochemical properties of the soil oscillates in average ranges of concentration (Table 2), with the following data: (pH) Hydrogen Potential (minimum range 5.40%, maximum range 8.29, average 7.66%); (E.C) Electrical Conductivity (minimum range 0. 25 dS/m, maximum range 0.56 dS/m, average 0.41 dS/m); (P) Phosphorus (minimum range 4.56 ppm, maximum range 20.62 ppm, average 11.44 ppm); (M.O) Organic Matter (minimum range 1.72%, maximum range 6.47%, average 4.29%); (N) Nitrogen (minimum range 0.09%, maximum range 0.32%, average 0.21%). The texture of the soils is distributed as Sandy Clay loam (Fr.Ar.A), Clay loam (Fr.Ar), Sandy loam (Fr.A) and Clay loam (Ar). The physicochemical analysis showed average concentration ranges, which did not affect the presence and abundance of AMF, since the physicochemical characteristics of the soil and the edaphoclimatic factors have a direct relationship with the presence, distribution and abundance of AMF (Entry et al., 2002; Sieverding, 2005; Khana et al., 2006).
Table 1. Number of AMF morphotypes by genera found in the provinces of Utcubamba and Bagua
| Genera | Province | Number of Morphotypes | Frequency | ||
| Utcubamba | Bagua | ||||
| Glomus | 23 | 23 | 30 | 53% | |
| Acaulospora | 6 | 8 | 14 | 25% | |
| Gigaspora | 1 | 3 | 3 | 5% | |
| Funneliformis | 2 | 2 | 2 | 4% | |
| Rhizophagus | 1 | 2 | 2 | 4% | |
| Scutellospora | 1 | 1 | 2 | 4% | |
| Sclerocystis | 1 | 1 | 2 | 4% | |
| Diversispora | 1 | 0 | 1 | 2% | |
| Rhizoglomus | 0 | 1 | 1 | 2% | |
| TOTAL | 57 | 100% | |||
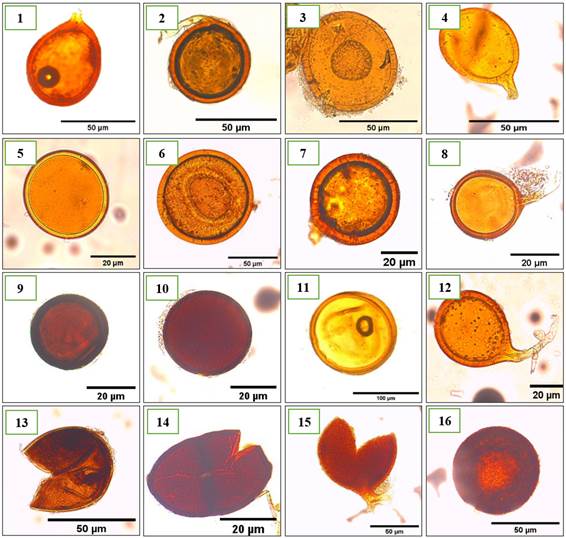
Figure 3. First sixteen morphotypes of AMF of the genus Glomus identified in the provinces of Utcubamba and Bagua. 1: Glomus sp. m1; 2: Glomus sp. m2; 3: Glomus sp. m3; 4: Glomus sp. m4; 5: Glomus sp. m5; 6: Glomus sp. m6; 7: Glomus sp. m7; 8: Glomus sp. m8; 9: Glomus sp. m9; 10: Glomus sp. m10; 11: Glomus sp. m11; 12: Glomus sp. m12; 13: Glomus sp. m13; 14: Glomus sp. m14; 15: Glomus sp. m15; 16: Glomus sp. m16. *m= morphotype.
Table 2. Physicochemical analysis of soil, distributed by districts within the provinces of Utcubamba and Bagua
| PROVINCES | DISTRICTS | pH | E.C | P | M.O | N | Textural class | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Sand | Silt | Clay | Texture | |||||||
| (1:1) | dS/m | ppm | % | % | % | % | % | |||
| UTCUBAMBA | Cajaruro | 7.15 | 0.56 | 15.91 | 4.31 | 0.22 | 48 | 20 | 32 | Fr.Ar.A. |
| Bagua Grande | 8.28 | 0.49 | 7.06 | 2.59 | 0.13 | 28 | 26 | 46 | Ar. | |
| Yamón | 7.88 | 0.39 | 7.45 | 3.45 | 0.17 | 66 | 16 | 18 | Fr.A. | |
| Cumba | 8.27 | 0.42 | 14.08 | 3.88 | 0.19 | 36 | 27.3 | 36.7 | Fr.Ar. | |
| El Milagro | 8.29 | 0.32 | 10.62 | 1.72 | 0.09 | 30 | 16 | 54 | Ar. | |
| Jamalca | 7.81 | 0.35 | 12.45 | 6.38 | 0.32 | 60 | 12 | 28 | Fr.Ar.A. | |
| BAGUA | Copallín | 8.11 | 0.4 | 10.91 | 3.45 | 0.17 | 44 | 18 | 38 | Fr.Ar. |
| Imaza | 5.40 | 0.25 | 4.56 | 4.31 | 0.22 | 72 | 15.3 | 12.7 | Fr.A. | |
| Aramango | 6.78 | 0.40 | 20.62 | 6.47 | 0.32 | 82 | 7.3 | 10.7 | Fr.A | |
| Bagua Capital | 7.66 | 0.51 | 7.45 | 5.17 | 0.26 | 52 | 16 | 32 | Fr.Ar.A. | |
| El Parco | 7.97 | 0.51 | 13.6 | 6.29 | 0.31 | 56 | 13.3 | 30.7 | Fr.Ar.A. | |
| La Peca | 8.28 | 0.33 | 12.74 | 3.45 | 0.17 | 72 | 11.3 | 16.7 | Fr.A. | |
*pH = Hydrogen Potential; *E.C = Electrical Conductivity; *P = Phosphorus; *O.M = Organic Matter; *N = Nitrogen. *Textural Class = (Sand, Silt, Clay); *Fr.Ar.A. = Sandy clay loam, *Fr.Ar. = Clay loam, *Fr.A. = Sandy loam, *Ar. = Clayey.
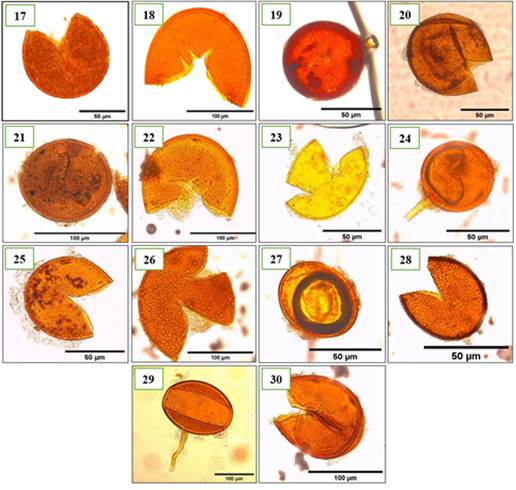
Figure 4. Fourteen other morphotypes of AMF of the genus Glomus identified in the provinces of Utcubamba and Bagua. 17: Glomus sp. m17; 18: Glomus sp. m18; 19: Glomus sp. m19; 20: Glomus sp. m20; 21: Glomus sp. m21; 22: Glomus sp. m22; 23: Glomus sp. m23; 24: Glomus sp. m24; 25: Glomus sp. m25; 26: Glomus sp. m26; 27: Glomus sp. m27; 28: Glomus sp. m28; 29: Glomus sp. m29; 30: Glomus sp. m30. *m= morphotype.
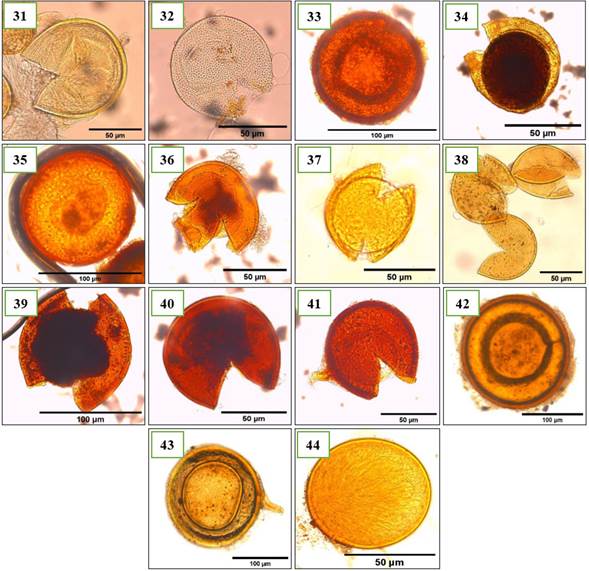
Figure 5. Morphotypes of AMF of the genus Acaulospora identified in the provinces of Utcubamba and Bagua. 31: Acaulospora sp. m1; 32: Acaulospora sp. m2; 33: Acaulospora sp. m3; 34: Acaulospora sp. m4; 35: Acaulospora sp. m5; 36: Acaulospora sp. m6; 37: Acaulospora sp. m7; 38: Acaulospora sp. m8; 49: Acaulospora sp. m9; 40: Acaulospora sp. m10; 41: Acaulospora sp. m11; 42: Acaulospora sp. m12; 43: Acaulospora sp. m13; 44: Acaulospora sp. m14. *m= morphotype.
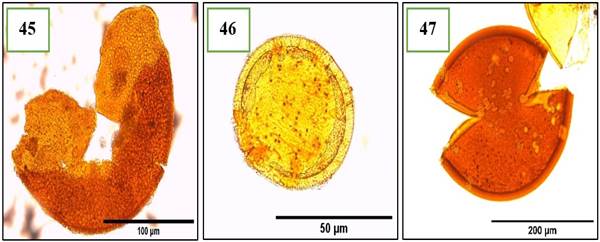
Figure 6. Morphotypes of AMF of the genus Gigaspora identified in the provinces of Utcubamba and Bagua. 45: Gigaspora sp. m1; 46: Gigaspora sp. m2; 47: Gigaspora sp. m3. *m= morphotype.
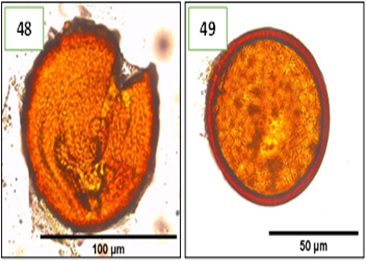
Figure 7. Morphotypes of AMF of the genus Funneliformis identified in the provinces of Utcubamba and Bagua. 48: Funneliformis sp. m1; 49: Funneliformis sp. m2. *m= morphotype.
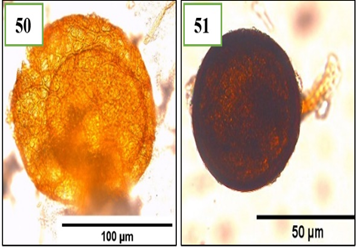
Figure 8. Morphotypes of AMF of the genus Rhizophagus identified in the provinces of Utcubamba and Bagua. 50: Rhizophagus sp. m1; 51: Rhizophagus sp.m2. *m= morphotype.
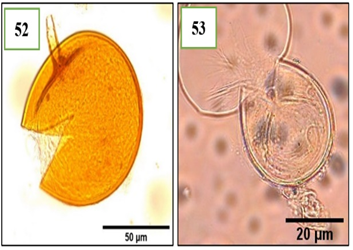
Figure 9. Morphotypes of AMF of the genus Scutellospora identified in the provinces of Utcubamba and Bagua. 52: Scutellospora sp. m1; 53: Scutellospora sp. m2. *m= morphotype.

Figure 10. Morphotypes of AMF of the genus Sclerocystis identified in the provinces of Utcubamba and Bagua. 54: Sclerocystis sp. m1; 55: Sclerocystis sp. m2. *m= morphotype.
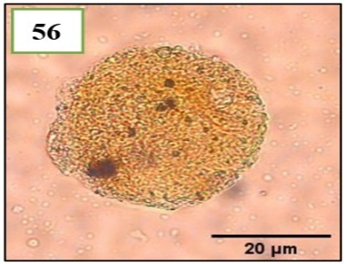
Figure 11. Morphotypes of AMF of the genus Diversispora identified in the provinces of Utcubamba and Bagua. 56: Diversispora sp. m1. *m= morphotype.
CONCLUSIONES
The following study presents the morphological identification of fungi that form arbuscular mycorrhizae associated with cocoa cultivation. 57 different morphotypes were identified, classified into 9 genera such as: Glomus, Acaulospora, Gigaspora, Funneliformis, Rhizophagus, Scutellospora, Sclerocystis, Diversispora and Rhizoglomus, with Glomus and Acaulospora being the genera with the highest representativeness index with 53% and 25% respectively. Managing to report its wide distribution in native cocoa ecosystems in the Bagua and Utcubamba provinces of the Amazon region. The results obtained provide important information that contributes to the knowledge of mycorrhizal diversity and fungus-plant symbiotic dynamics in cocoa crops.












 uBio
uBio