Introduction
Gerbode defect (GD) is a communication between the left ventricle and the right atrium and was initially described before the development of echocardiography, as an extremely rare congenital heart disease (<0.08%) 1. Thurman described the first case in 1938, but it was not until 1958 that Frank Gerbode published the first series of successfully repaired cases and coined his name 1,2. He defined this pathology as a high ventricular septal defect associated with a tricuspid septal valve defect allowing blood from the left ventricle to enter the right atrium 2,3.
This condition has an unknown etiology 4. The membranous septum is 5-10 mm apical the mitral leaflet on the left side. It is divided by the tricuspid septal leaflet into an atrioventricular (right atrium separated from the left ventricle), and an interventricular (between both ventricles) portion 4. Riemenschneider and Moss classified it as: Supravalvular, which occurs on the atrioventricular membranous septum (true GD); Infravalvular, which is on the interventricular membranous septum (always associated with an anomaly of the tricuspid septal leaflet, causing a flow directly to the right atrium); and finally, the Combined variant, which unifies previous characteristics 3,4.
Case report
A 43-year-old woman with no relevant medical history was admitted to the emergency service due to intense abdominal pain in the right iliac fossa for the past 24 hours. The clinical picture and laboratory findings were consistent with acute appendicitis. There were abnormal findings in the physical examination and cardiac auscultation. We identified a holosystolic murmur (III/VI) in the aortic focus with the splitting of the second heart sound. The electrocardiogram revealed a right bundle branch block with signs of hypertrophy on the left ventricle; chest tomography showed moderate dilatation of the right heart chambers (mainly the right atrium) and the pulmonary arterial trunk.
These incidental findings did not delay the emergency surgical indication, and we performed an appendectomy. In the postoperative period, there were no complications related to the surgery, and the patient underwent a transthoracic echocardiogram (TTE), that revealed a 4 mm defect in the high membranous septum, which seemed to connect the left ventricle and the right atrium with a high-speed holosystolic shunt (pressure gradient, 109 mmHg), left-right flow directed to the thickened and curved septal leaflet of the tricuspid valve. The Qp/Qs was 1.1, and we also found an image suggestive of a subaortic fibromuscular ring or membrane that did not produce left ventricular outflow tract obstruction (Figure 1).

Figure 1 Transthoracic echocardiogram findings. A-B. Anterior 4-chamber view (A4V) shows a GD in the membranous septum (white arrow), color Doppler shows left to right shunt and aliasing due to flow acceleration (white arrow), continuous Doppler shows velocities greater than 5 m/s. C-F. Parasternal short axis view (PSAV) in the aortic plane in systole (CD) and diastole (E-F) shows the GD (white arrows), subaortic membrane (arrowhead), tricuspid septal leaflet (asterisk). The color Doppler shows the shunt directed towards the base of the tricuspid septal leaflet, and the drop in flow velocity allows us to appreciate its course. G. Parasternal long axis view (PLA) in the aortic plane in systole shows the subaortic membrane (arrowhead).
Initially, the first diagnosis was a ruptured sinus of valsalva with right aorto-atrial fistula (AAF); however, the transesophageal echocardiogram (TEE-2D/3D) showed that the defect communicated the left ventricle and the right atrium; and confirmed the subaortic fibromuscular ring (Figure 2). These incidental findings were consistent with a supravalvular GD and a subaortic membrane that did not require surgical repair. Subsequently, the patient was discharged from the hospital with an indication for outpatient cardiology follow up.
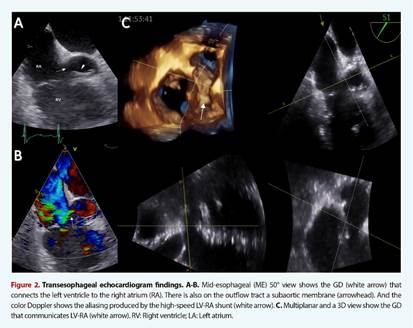
Figure 2 Transesophageal echocardiogram findings. A-B. Mid-esophageal (ME) 50° view shows the GD (white arrow) that connects the left ventricle to the right atrium (RA). There is also on the outflow tract a subaortic membrane (arrowhead). And the color Doppler shows the aliasing produced by the high-speed LV-RA shunt (white arrow). C. Multiplanar and a 3D view show the GD that communicates LV-RA (white arrow). RV: Right ventricle; LA: Left atrium.
Discussion
With the evolution and improvement of cardiac imaging, there are more and more cases that report acquired atrioventricular defects around the world, which far exceed congenital defects (74% vs. 26%) 4-6. This finding is more frequent in younger subjects that may present in isolation or associated with other cardiac malformations 3,7,8.
Cases of acquired GD are frequently associated with a previous history of cardiac valve surgery (70%). So, the risk of developing this condition increases with the number of cardiac procedures 6. Other etiologies that may cause GD are infective endocarditis, inferior myocardial infarction, iatrogenic punctures of the high membranous septum, and an unusual case report due to extensive calcification of the membranous septum secondary to mediastinal radiotherapy 4,5,8.
Timely diagnosis includes a thorough physical examination. Symptoms, the dissociation between the murmur (similar to ventricular septal defect), and the dilatation of right cavities suggest the presence of GD 4,9. It is important to highlight that the TTE plays a role as the first test that shows the membranous septal defect, with a diagnostic accuracy of 66.9% 4,8. The presence of high-velocity systolic flow (>4 m/s) from the left ventricle to the right atrium, associated with dilatation of the latter, guarantees the diagnosis in most patients 3-5. In our case, diagnostic doubt initially existed because the images obtained from the four- and five-chamber views did not clearly show whether the defect was between the left ventricle and the right atrium or interventricular. Some authors describe that the parasternal short axis can determine better the origin of this defect compared to the apical view; however, our findings in the parasternal axis also gave us the differential diagnostic suspicion of rupture of the sinus of Valsalva and right AAF 3,5,6.
Some reports described that AAF constitutes the first differential diagnosis of GD; however, there is a clear difference in its correct identification. GD presents a left to right shunt in systole while the AAF is continuous (systole and diastole) 4. On the other hand, the flow of the GD could be confused with that of the tricuspid regurgitation, causing the excess of pulmonary pressures. Some indirect findings in the echo that favors the diagnosis of GD are the presence of an atypical high-velocity jet with absence of indirect signs of pulmonary hypertension (right ventricular hypertrophy and septal flattening), and an estimated normal pulmonary diastolic pressure from pulmonary regurgitation 5,8,10.
The presence of two distinguishable trans-tricuspid flows during systole and of different velocities is an important finding that could help to diagnose infravalvular GD. Likewise, the three-dimensional images obtained by TTE could define and support the diagnosis 7. TEE/2D-3D is the diagnostic method of choice for GD because it provides details of the location, size, and direction of the shunt, which allows differentiation from tricuspid regurgitation flow and rules out alternative diagnoses. Currently, there are other imaging techniques like tomography and cardiac resonance that we can use. Even some reports highlight the superiority of computed tomography angiography even over transesophageal echocardiography under specific clinical scenarios (sensitivity, 85% vs. 68%) 4,9. However, all these recommendations have to be under the center expertise and local resources.
There are currently no specific indications for the closure of the GD. Despite this, the management we use is the same as in other septal defects. The severity of the symptoms, the dilation of the right cavities, the Qp/Qs ratio, and pulmonary hypertension are important for decision-making 3,5. Although cardiac surgery and closure with a pericardial patch is the most widely used technique, there are more and more reports on percutaneous closure in patients with high surgical risk 5,8,9. The technical success of surgical repair is excellent, and it has low mortality rates (<3%); however, surgery in the context of acquired GD due to endocarditis or myocardial infarction could be associated with higher mortality (15-65%) 4,6.
In conclusion, despite the lack of comparative studies, patients with GD will require close clinical and echocardiographic follow-up in shorter intervals than for other septal defects. We have to bear in mind that the right atrium is the chamber with the lowest pressure, and having high pressure from the left ventricle, will cause enlargement and then increase pulmonary blood flow, which ends in pulmonary hypertension. Therefore, outpatient imaging with echocardiographic plays an important role in the close follow-up of patients with this rare condition.















