Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Peruana de Medicina Experimental y Salud Publica
Print version ISSN 1726-4634
Rev. perú. med. exp. salud publica vol.37 no.4 Lima Oct-Dec 2020 Epub Nov 30, 2020
http://dx.doi.org/10.17843/rpmesp.2020.374.5064
Original articles
Development of a model for predicting major infection following pediatric heart surgery
1 Facultad de Medicina, Universidad Peruana de Ciencias Aplicadas (UPC), Lima, Perú.
2 ProEd Communications, Inc., Cleveland, Ohio, Estados Unidos.
3 Departamento de Salud Pública, Erasmus MC, Rotterdam, Países Bajos.
4 Departamento de Estadística Médica, Leiden University Medical Center, Leiden, Países Bajos.
5 Escuela de Farmacia, University of Connecticut, Storrs, Connecticut, Estados Unidos.
6 Departamento de Cirugía, Unidad Postoperatoria Cardiovascular, Instituto Nacional de Salud del Niño (INSN), Lima, Perú.
INTRODUCTION
The aim of the prediction models is to help care personnel during the decision-making process by identifying patients with a higher risk of presenting a certain outcome 1. Postoperative complications occur in 43% of pediatric patients undergoing heart surgery with extracorporeal circulation 2 , 3. Postoperative infections are associated with pediatric heart surgery, with a reported incidence of 11% to 38% 1 - 10. In this context, there are three particularly important infectious complications: sepsis, mediastinitis, and endocarditis, which together form an entity called major postoperative infection (MPI), which is defined as one or more of the three infections during the established postoperative period from 48 hours after the operation to the date of discharge. The incidence of MPI worldwide varies between 2.8% and 3.5% in the pediatric and adult population 11 - 19. Although rare, MPI is a very severe complication due to the impact on mortality (22% to 25% in patients with MPI vs. 3% to 3.9% in patients without MPI), on length of hospital stay and on institutional costs 11 - 13 , 15 - 20.
Several factors are associated with the occurrence of MPI and the probability is higher in those procedures assisted by cardiopulmonary bypass or extracorporeal circulation (ECC) 1 , 12. Associated preoperative factors include younger age, low weight, previous long hospital stay, previous admission to an intensive care unit (ICU), higher functional class of heart failure, higher level of surgery complexity (Risk Adjustment for Congenital Heart Surgery score [RACHS-1]), respiratory support, cyanotic heart disease, genetic disorders, and other comorbidities 1 , 7 , 9 , 10 - 12 , 17 , 18. MPI has also been associated with intraoperative factors, such as prolonged surgery time and ECC, volume and number of blood transfusions, intraoperative hypothermia, use of intracardiac prosthesis, and open chest condition upon leaving the operating room (OR) 1 , 7 , 10 , 17 , 18.
Different predictive models have been proposed to calculate the MPI risk after heart surgery. Among the most relevant factors were the following: age under six months, time with open chest, and postoperative stay for more than two days 3. There is a predictive model of MPI from the Society of Thoracic Surgeons (STS) that includes preoperative variables, comorbidities, and cardiac surgical procedures 11; the model showed good performance and good discriminatory performance and was later validated by Kansy in Poland 12.
The aim of the research was to develop a risk prediction model for pediatric MPIs and to validate the one of the STS.
KEY MESSAGES
Motivation for the study: Postoperative complications after heart surgery, especially sepsis, have a big impact on the patient mortality rate, hospital stay and associated costs.
Main findings: A prediction model was designed using preoperative information to estimate the risk of major postoperative infection in pediatric patients undergoing cardiac surgery with extracorporeal circulation. The internally validated model performed well.
Implications: With this easy-to-use predictive model, children at high risk of developing a major postoperative infection after pediatric cardiac surgery can be identified. This will help to develop prevention strategies, healthcare protocols, and further research.
MATERIALS AND METHODS
Type of study
A retrospective cohort study was carried out. The target population was all patients under 18 who underwent heart surgery with ECC admitted between 2000 and 2010 to the Post-Operative Cardiovascular Unit (UPOCV) of the Instituto Nacional de Salud del Niño (INSN) in Lima, Peru. Patients who did not use ECC were excluded.
The UPOCV team of intensivists and an INSN pediatric infectious disease specialist made the diagnosis of infection. The diagnostic criteria and definitions established by STS for sepsis, mediastinitis and endocarditis were used 21.
Procedure
Independent variables
Risk factors for postoperative pediatric cardiac infections were formally sought in the literature 1 , 2 , 7 , 9 - 13 , 17 - 22. The search was conducted on PubMed and Scopus through October 2018. Preoperative variables studied included age, weight, sex, provenance, prematurity, acute malnutrition, length of preoperative stay, previous cardiac surgery, type of heart disease, Risk Adjustment for Congenital Heart Surgery score (RACHS-1), previous mechanical ventilation, pneumonia, pulmonary tuberculosis, tracheostomy, congenital malformations, genetic disorders, cardiac and non-cardiac malformations, hepatitis, sepsis and endocarditis, renal failure, dialysis, cardiogenic shock, arrhythmias, pulmonary hypertension, cyanotic heart disease, Ross-modified functional class, hypothyroidism, history of epilepsy and neurological damage 7 , 11 - 13 , 17 , 21. The intraoperative variables studied were non-elective surgery (versus elective surgery), intraoperative diagnosis, type of surgery, time of ECC, circulatory arrest, ultrafiltration during ECC, use of corticosteroid, deep hypothermia, and open chest.
Data source
The sources of information were the patient’s medical records and medical history. The data contained the same information indicated in the STS file for congenital heart surgery database. Data collection was performed by one of the researchers (EWS). The information collected was exported to the SPSS version 22 program (IBM Corp., Armonk, NY, USA) for statistical analysis.
Development of the model
We used The Chi-square test and Fisher’s exact test to determine differences in population distribution between patients with and without MPIs. Subsequently, we carried out a logistic and binary regression model and selected those with a p < 0.200 23. The association of each variable with the primary outcome was reported using odds ratio (OR) and a 95% confidence interval (95% CI).
Internal validation of the prediction model
The model was validated internally using the bootstrapping technique with 200 repetitions. Each repetition created models in samples identical in size to the study sample size, using random sampling with replacement of individuals. The internal validation provided a correction factor to adjust the model coefficients, assuming that the initial model coefficients were inflated for predictions in the new groups.
Evaluation of model performance
The predictive model was evaluated for discriminatory performance and clinical utility. Discrimination refers to how useful the model is in differentiating between patients with presence and absence of MPI. Discrimination is quantified with the c-statistic, which is equivalent to the area under the ROC curve. The c-statistic has values ranging from 0.5 to 1, and the closer it is to 1, the better the model. Clinical utility was evaluated with benefit analysis 24; this method evaluates the benefit of correctly identifying a patient with a high risk of MPIs (true positives) versus mislabeling them as probable MPIs (false positives) in a range of reasonable MPI risk thresholds where the physician feels comfortable deciding (for example, to administer antibiotics for an MPI). A threshold of 20% was considered reasonable, with a range of 5% to 40%.
Finally, the calibration of an internally validated model was evaluated with a calibration graph describing the relationship between the expected and observed MPI risks. Restricted cubic splines were used to visualize the relationship. The calibration of the model was also evaluated using the Hosmer-Lemeshow test.
Score and nomogram
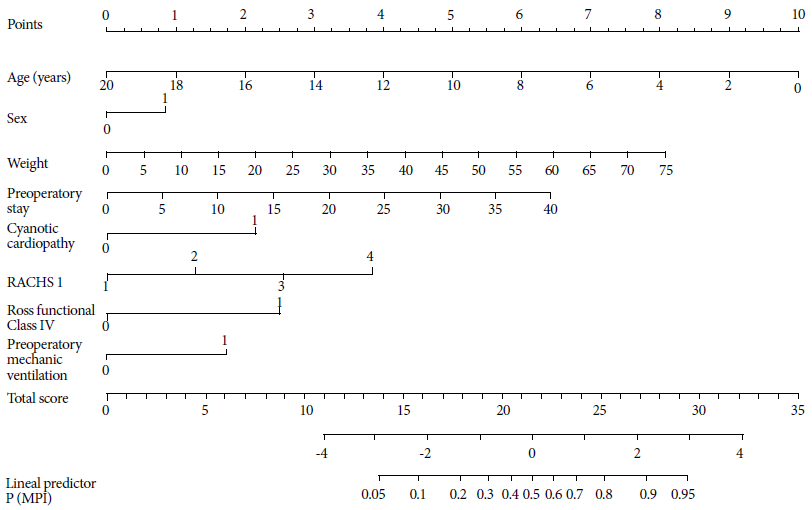
We presented a model as scores for clinical use. For each variable we assigned a score that was proportional to the strength of association with the primary outcome of MPI. Therefore, the variables of the final prediction model with higher OR had higher scores. The final score was obtained by the sum of the scores of each variable in the final model. The scores were created by multiplying the variable’s coefficient β by 10. We also created A nomogram, which allows the direct calculation of the probability of the primary outcome. In the nomogram, each variable has a different scale that is proportional to the weight of its coefficients 20 , 24 - 27. The equivalence of MPI scores and risks with preoperative variables was also calculated. We carried out all these analyses with the R 3.3.2 program (www.r-project.org).
Validation of STS model
Besides discrimination and internal calibration, the comparison of infection rates was also used for model validation. Our model and STS model were compared using the MPI rates of six heart surgeries comparable to our study (atrial septal defect, ventricular septal defect, atrial ventricular canal, tetralogy of Fallot, Glenn, and double outlet right ventricle) through Fisher’s exact test.
RESULTS
Demographic characteristics
A total of 1,025 patients were analyzed, 59 (5.8%) of them had at least one episode of MPI (4.8% sepsis, 1.0% mediastinitis, 0% endocarditis). Three patients had more than one type of infection. Ninety-five percent of the operated patients had postoperative sepsis. There were only ten cases of mediastinitis.
Patients with MPI were younger (2.95 vs. 5.42 years; p < 0.001), and 62.7% were men (Table 1). The average weight of patients with MPI was significantly lower than the rest of the patients (10.9 kg vs. 16.8 kg; p < 0.001) (Table 2). The length of preoperative stay was longer in patients with MPI (median of 40 vs. 15 days; p < 0.001). Most patients with MPI were diagnosed with cyanotic heart disease (69.5%; p < 0.001). In addition, patients with MPI had a higher percentage on the RACHS-1 3-4 scale (54% vs. 27%; p < .001). A similar result was found for Ross-modified functional class IV (higher in patients with MPI, 37% vs. 15% in the total population, p < 0.001). On the other hand, more patients with MPI had previous mechanical ventilation (11.9% vs 0.9%; p < 0.001). There was a greater number of patients with a history of previous infection resolved at the time of surgery in the cases with MPI (7.5% vs 32%; p < 0.001). There were no differences in congenital anomalies, malformations, and malnutrition status.
Table 1 Clinical demographics of pediatric patients who had cardiac surgery with extracorporeal circulation at Instituto Nacional de Salud del Niño, between 2000 and 2010
| Variables | Total | Yes (n = 59) | No (n = 966) | p value | OR (95% CI) | p value |
|---|---|---|---|---|---|---|
| Age (years) (mean, SD) | 5.27 (4.8) | 2.95 (4.6) | 5.42 (4.8) | <0.001 | 0.86 (0.79-0.93) | <0.001 |
| Age group | ||||||
| 0-6 m | 120 (11.7) | 28 (47.5) | 92 (9.5) | <0.001 | Ref. | |
| 7-11 m | 54 (5.3) | 5 (8.5) | 49 (5.1) | 7.30 (3.20-16.65) | 0.010 | |
| 12 m - 5 y | 502 (49) | 17 (28.8) | 485 (50.2) | 2.45 (0.77-7.82) | 0.130 | |
| 6-9 y | 149 (14.5) | 1 (1.7) | 148 (15.3) | 0.84 (0.36-1.98) | 0.690 | |
| 10 y or more | 200 (19.5) | 8 (13.6) | 192 (19.9) | 0.16 (0.02-1.31) | 0.090 | |
| Sex (n, %) | ||||||
| Male | 523 (51.0) | 37 (62.7) | 486 (50.3) | 0.040 | 1.66 (0.96-2.86) | 0.067 |
| Female | 502 (49.0) | 22 (37.3) | 480 (49.7) | Ref. | ||
| Provenance (n, %) | ||||||
| Lima | 533 (52.0) | 33 (55.9) | 500 (51.8) | 0.310 | 0.85 (0.49-1.43) | 0.530 |
| Provinces | 492 (48.0) | 26 (44.1) | 466 (48.2) | Ref. | ||
| Year of surgery (n, %) | ||||||
| 2001-2005 | 439 (42.8) | 20 (33.9) | 419 (43.4) | 0.176 | 0.67 (0.39-1.17) | 0.160 |
| 2006-2010 | 586 (57.2) | 39 (66.1) | 547 (56.6) | Ref. | ||
| Prematurity (n, %) | ||||||
| No | 1,013 (98.8) | 58 (98.3) | 955 (98.9) | 0.093 | 1.50 (0.19-11.79) * | 0.700 |
| 32-35 weeks | 9 (0.9) | 0 (0.0) | 9 (0.9) | |||
| 36 weeks | 3 (0.3) | 1 (1.7) | 2 (0.2) |
* Compared to prematurity.
OR: odds ratio; SD: standard deviation; m: months; y: years, Ref: reference.
Table 2 Preoperative and intraoperative clinical characteristics of pediatric patients who underwent extracorporeal circulation heart surgery at Instituto Nacional de Salud del Niño, between 2000 and 2010.
| Variables | Total | Yes (n = 59) | No (n = 966) | p value | OR (95% CI) | p value |
|---|---|---|---|---|---|---|
| Preoperative | ||||||
| Weight, kg (mean, SD) | 16.50 (12.3) | 10.9 (12.5) | 16.8 (12.3) | <0.001 | 0.94 (0.91-0.97) | 0.001 |
| Weight group (kg) | ||||||
| 2.5-5 | 150 (14.6) | 30 (50.8) | 120 (12.4) | <0.001 | Ref. | |
| 6-10 | 279 (27.2) | 13 (22.0) | 266 (27.5) | 5.79(2.33-14.38) | 0.001 | |
| 11-30 | 451 (44.0) | 10 (16.9) | 441 (45.7) | 1.13(0.42-3.04) | 0.810 | |
| 31 or more | 145 (14.1) | 6 (10.2) | 139 (14.4) | 0.53(0.19-1.47) | 0.220 | |
| Preoperative stay, days (mean, IQR) * | 4 (14) | 15 (40) | 4 (12.25) | <0.001 | 1.08 (1.04-1.11) | <0.001 |
| Previous stay group (days) | ||||||
| 0-1 | 302 (29.5) | 11 (18.6) | 291 (30.1) | <0.001 | Ref. | |
| 2-7 | 347 (33.9) | 13 (22.0) | 334 (34.6) | 0.29(0.14-0.59) | 0.001 | |
| 8-14 | 116 (11.3) | 5 (8.5) | 111 (11.5) | 0.30(0.15-0.58) | 0.001 | |
| 15 or more | 260 (25.4) | 30 (50.8) | 230 (23.8) | 0.35(0.13-0.91) | 0.320 | |
| Type of cardiopathy (n, %) | ||||||
| VSD | 329 (32.1) | 7 (11.9) | 322 (33.3) | <0.001 | Ref. | |
| TF | 152 (14.8) | 9 (15.3) | 143 (14.8) | 2.895 (1.06-7.93) | ||
| ASD | 123 (12.0) | 2 (3.4) | 121 (12.5) | 0.760 (0.16-3.71) | ||
| TAPVR | 77 (7.5) | 10 (16.9) | 67 (6.9) | 6.866 (2.52-18.68) | <0.001 | |
| Others | 344 (33.6) | 31 (52.5) | 313 (32.4) | 4.556 (1.98-10.49) | <0.001 | |
| Cyanotic cardiopathy (n, %) | 420 (41.0) | 41 (69.5) | 379 (39.2) | <0.001 | 3.53 (1.99-6.23) | <0.001 |
| RACHS-1 (n, %) | ||||||
| 1 | 128 (12.5) | 2 (3.4) | 126 (13.0) | <0.001 | 0.107 (0.02-0.53) | 0.001 |
| 2 | 617 (60.2) | 25 (42.4) | 592 (61.3) | 0.284 (0.12-0,69) | 0.060 | |
| 3 | 226 (22.0) | 25 (42.4) | 201 (20.8) | 0.835 (0.34-2.05) | 0.690 | |
| 4 | 54 (5.3) | 7 (11.9) | 47 (4.9) | Ref. | ||
| RACHS-1 (mean, IQR) * | 2 (2.3) | 3 (2.3) | 2 (2.3) | <0.001 | 2.20 (1.57-3.08) | <0.001 |
| Ross modified functional class (n, %) | ||||||
| I | 216 (21.1) | 5 (8.5) | 211 (21.8) | <0.001 | 0.142 (0.05-0.38) | <0.001 |
| II | 280 (27.3) | 16 (27.1) | 264 (27.3) | 0.364 (0.19-0.72) | 0.030 | |
| III | 375 (36.6) | 16 (27.1) | 359 (37.2) | 0.267 (0.14-0.53) | <0.001 | |
| IV | 154 (15.0) | 22 (37.3) | 132 (13.7) | Ref. | ||
| Ross modified functional class IV (n, %) | ||||||
| Yes | 154 (15.0) | 22 (37.29) | 132 (13.66) | <0.001 | 3.76 (2.15-6.57) | <0.001 |
| No | 871 (85.0) | 37 (62.71) | 834 (86.34) | Ref. | ||
| Genetic malformation (n, %) | 83 (8.1) | 7 (11.9) | 76 (7.9) | 0.190 | 1.576 (0.69-3.59) | 0.280 |
| Genetic disorder (n, %) | 171 (16.7) | 7 (11.9) | 164 (17.0) | 0.200 | 1.519 (0.68-3.4) | 0.310 |
| Previous cardiac surgery (n, %) | 225 (22.0) | 13 (22.0) | 212 (21.9) | 0.550 | 1.005 (0.53-1.89) | 0.980 |
| Acute malnutrition (n, %) | 492 (48.0) | 30 (50.8) | 462 (47.8) | 0.370 | 1.129 (0.67-1.91) | 0.650 |
| Mechanical ventilation (n, %) | 16 (1.6) | 7 (11.9) | 9 (0.9) | <0.001 | 0.07 (0.03-0.19) | <0.001 |
| Previous infection (n, %) | 91 (8.9) | 19 (32) | 72 (7.5) | <0.001 | 5.90 (3.25-10.71) | <0.001 |
| Tracheotomy (n, %) | 1 (0.1) | 0 (0) | 1 (0.1) | 0.940 | - | - |
| Kidney failure (n, %) | 6 (0.6) | 0 (0) | 6 (0.6) | 0.700 | - | - |
| Intraoperative | ||||||
| Intraoperative Diagnosis (n, %) | ||||||
| VSD | 321 (31.3) | 7 (11.9) | 314 (32.5) | <0.001 | Ref. | |
| TF | 146 (14.2) | 6 (10.2) | 140 (14.5) | 1.92 (0.64-5.82) | 0.250 | |
| ASD | 115 (11.2) | 2 (3.4) | 113 (11.7) | 0.79 (0.16-3.88) | 0.780 | |
| TAPVR | 76 (7.4) | 10 (16.9) | 66 (6.8) | 6.79 (2.49-18.51) | <0.001 | |
| Others | 367 (35.8) | 34 (57.6) | 333 (34.5) | 4.58 (2-10.48) | <0.001 | |
| Non-elective surgery ⱡ (n, %) | 480 (46.8) | 47 (79.7) | 433 (44.8) | <0.001 | 4.821 (2.53-9.2) | <0.001 |
| Palliative heart surgery (n, %) | 204 (19.9) | 12 (20.3) | 192 (19.9) | 0.520 | 1.029 (0.54-1.98) | 0.930 |
| Time ECC, minutes (mean, DS) | 93.23 (62.6) | 97.1 (50) | 92,9 (63.3) | 0.570 | 1.01 (0.99-1.01) | 0.630 |
| Circulatory arrest (n, %) | 23 (2.2) | 3 (5.1) | 20 (2.1) | 0.140 | 0.39 (0.11-1.37) | 0.140 |
| Ultrafiltration during ECC (n, %) | 467 (45.6) | 30 (50.8) | 437 (45.2) | 0.240 | 1.25 (0.74-2.12) | 0.400 |
| Corticoid use (n, %) | 845 (82.4) | 48 (81.4) | 797 (82.5) | 0.900 | 0.9 (0.46-1.76) | 0.760 |
* By increase in 1 unit. ⱡ vs. elective.
VSD: ventricular septal defect; TF: tetralogy of Fallot; ASD: atrial septal defect; TAPVR: total anomalous pulmonary venous return; RACHS-1: classification adjusted to the risk for congenital heart surgery; SD: standard deviation; IQR: interquartile range; OR: odds ratio; Ref: reference.
We found that patients undergoing non-elective surgery had more MPIs (79.7% vs 44.8%; p < 0.001) (Table 2). There was no difference between the cases of MPI for the year of surgery, the longest time of extracorporeal circulation, deep hypothermia, open chest, and valve prosthesis.
The total mortality rate at 30 days was 13.7% and significantly higher in the group of patients with MPI (mortality 37/59 [63%] vs. 125/966 [13%]; p < 0.001). The duration of postoperative mechanical ventilation (301.6 hours vs. 34.3 hours; p < 0.001), postoperative intensive care unit stay (20.9 days vs. 5.1 days; p < 0.001), and postoperative stay (42.2 days vs. 15.1 days; p < 0.001) were greater in patients with PMI. Nine deaths with MPI had a history of sepsis and two of endocarditis. Excluding these patients, mortality in patients with MPI remained high (58%) and significantly different from patients without MPI (p < 0.001). Of the 32 patients, two died of sepsis.
Bivariate and multivariate regression model
In the bivariate analysis of preoperative variables, a strong association was found between MPI and younger age, lower weight, longer preoperative stay, type of heart disease, cyanotic heart disease, higher RACHS-1 scale and Ross-modified functional class, and previous mechanical ventilation (Table 1 and 2). The non-elective (vs. elective) surgeries also had a statistical association with MPI (Table 2).
We created a preoperative model and a combined model (preoperative and intraoperative). In the first model, shown in Table 3, the variables associated with PMI were prolonged preoperative hospital stay (p = 0.053), diagnosis of cyanotic heart disease (p = 0.020), RACHS-1 (p = 0.013), and Ross-modified functional class IV (p = 0.009). In the second model, the associated variables were cyanotic heart disease (p = 0.020), RACHS-1 (p = 0.020), Ross-modified functional class IV (p = 0.010), and non-elective surgical condition (p = 0.020).
Table 3 Preoperative multivariate analysis
| Variables | β | OR (95% CI) | p value |
|---|---|---|---|
| Age, per increase in 1 year | -0.184 | 0.83 (0.68-1.02) | 0.070 |
| Male sex | 0.317 | 1.38 (0.77-2.50) | 0.280 |
| Weight in kg | 0.040 | 1.04 (0.96-1.12) | 0.310 |
| Preoperative stay, by increase in 1 day | 0.002 | 1.06 (1.02-1.11) | 0.053 |
| Cyanotic cardiopathy | 0.800 | 2.23 (1.17-4.26) | 0.020 |
| RACHS-1, by increase in 1 unit | 0.480 | 1.62 (1.09-2.40) | 0.013 |
| Ross Functional Class IV | 0.905 | 2.56 (1.30-5.00) | 0.009 |
| Preoperative mechanical ventilation | 0.556 | 1.91 (0.60-6.06) | 0.270 |
95% CI: 95% confidence interval; RACHS-1: risk-adjusted classification for congenital heart surgery; OR: odds ratio.
Performance of the preoperative model
Internal validation of the model produced a shrinkage factor of 0.84. The discriminatory performance of the model was good, with an adjusted c-statistic of 0.80 (95% CI 0.74-0.86). The combined preoperative and intraoperative model showed no significant improvement in discrimination compared to the preoperative model (P = 0.600), which is shown in the supplemental material (Figure S1). The net benefit analysis showed that both models had a similar performance for MPI ranging from 0% to 40%, and their use is better than not using a predictive model (supplemental material, Figure S2). The calibration of both models was good as expected in an internally validated model.
The preoperative model was presented as scores and a nomogram. The scores per variable included in the final model are shown in Table 4, and the equivalence of MPI scores and risks are shown in Table 5. Scores between 12 and 29 correspond to MPI probabilities of 5% to 95%, respectively. A nomogram is shown in Figure 1. For example, a 5-year-old girl with 20 kg, 30 days of preoperative stay, with cyanotic heart disease and a RACHS-1 scale of 3 will have a 19-point and 30% risk of MPI.
Table 4 Scores for risk of major postoperative infections after pediatric heart surgery by variable included in the preoperative multivariate model.
| Variable | Value | Score |
|---|---|---|
| Age | By year of age | -2 |
| Male sex | Yes | 3 |
| Weight | By 2 kg | 1 |
| Length of stay | 5 | 1 |
| 10 | 2 | |
| 15 | 4 | |
| 20 | 5 | |
| 25 | 6 | |
| 30 | 8 | |
| 35 | 9 | |
| 40 | 10 | |
| Cyanotic cardiopathy | Yes | 8 |
| RACHS-1 | By unit | 5 |
| Functional class IV | Yes | 9 |
| Preoperative mechanical ventilation | Yes | 6 |
*Note: The preoperative stay is divided by 5 to express the risk for increased 5-day hospital stay. For example: a patient with 25 days of hospitalization before surgery has a value of 5 (i.e., obtained dividing 25 by 5) and has 1 point according to the model.
RACHS-1: Risk-adjusted classification for congenital heart surgery.
Table 5 Risk of major postoperative infections after pediatric cardiac surgery according to the preoperative model score.
| Score | MPI risk (%) |
|---|---|
| 14 | 5 |
| 16 | 10 |
| 18 | 20 |
| 19 | 30 |
| 21 | 40 |
| 22 | 50 |
| 23 | 60 |
| 24 | 70 |
| 25 | 80 |
| 27 | 90 |
| 29 | 95 |
MPI: Major Postoperative Infections after pediatric cardiac surgery
Validation of the Society of Thoracic Surgeons model
The STS model for MPI was validated with our own database. The discriminatory performance was moderate (c-statistical 0.78; 95% CI 0.71 to 0.84) and the calibration was not good (Hosmer-Lemeshow test was 18.2; P = 0.020). The STS model underestimated the risk of MPI, particularly for the high risks observed in our population. Fisher’s exact test was used to compare both models regarding the MPI rate (supplemental material, Table S1). Significant differences were found in two surgeries: atrial septal defect and double outlet right ventricle (p = 0.010 and p = 0.030, respectively).
DISCUSSION
We developed a risk model for MPIs which showed good performance. Clinical data from all pediatric heart surgeries performed using extracorporeal circulation in a decade at a national reference institute for pediatric healthcare in Peru. In addition to the predictive model that establishes a score to estimate the clinical prognosis, a nomogram was designed for simplified use. We found a MPI rate of 5.8% (sepsis 4.8%), and some preoperative risk factors were identified, all of which were easy to measure.
The incidence of postoperative infections and the associated risk factors in the studied population are similar to those found in high-income countries as well as in low-income countries 7 , 11 - 22. The study confirms that MPI are associated with significantly high mortality, long duration of mechanical ventilation, and prolonged postoperative and ICU stay. Despite similar MPI rates, the mortality rate found was higher compared to rates described in previous studies 11 - 12 , 17 , 20 , 22. It should be noted that the previous studies excluded patients with a history of sepsis or endocarditis for calculating mortality rates in MPI. Even by excluding the seven patients with a history of sepsis and two with endocarditis, the rate remains high (58%) and significantly different from those without MPI. More studies are needed to better understand factors related to the short- and long-term consequences of postoperative cardiac infections in pediatric patients and whether interventions over a follow-up period might change outcomes.
There was no significant change in the rate of major postoperative infection over the 10-year period. There were also no major changes in medical practice during the study period regarding surgeons, operating room (OR) personnel, the UPOCV, hand washing techniques, use of antibiotics, wound care, or complexity of procedures, all of which were regulated by protocols approved by various international committees and used at the INSN 28 , 29.
We can confirm that younger age, lower weight, longer preoperative stay, higher procedure complexity (RACHS-1), diagnosis of cyanotic heart disease, preoperative mechanical ventilation, and non-elective surgical condition are variables associated with an increased risk of MPI 2 , 7 , 9 , 10 - 12 , 17 , 18. In addition, other risk factors for MPI were identified as Ross-modified functional class IV with a highly associated. There were no patients diagnosed with Di George’s syndrome; and Down syndrome cases showed no difference for MPI.
Previous research also described intraoperative factors associated with an increased risk of MPI, such as longer ECC time, deep hypothermia, open chest upon leaving the OR, and the use of prosthetic valves during surgery, these factors were not significant in our analysis 2 , 7 , 10 , 17 , 18.
With well-established predictors, this model accurately identified patients at high risk for MPI. It showed satisfactory calibration as well as good a discriminatory performance. The STS model presented by Barker et al. 11 was validated with the 10-year data and found a similar c-statistic of 0.78 and a calibration of 0.66 which differed from our study; the model presented a set of preoperative variables like ours. However, authors of the STS model found that prior heart surgery and genetic abnormalities were risk factors for MPI.
We also compared discrimination performance with the study carried out by Kansy et al. 12, the latter being slightly better regarding discrimination with a c-statistic of 0.81 and a calibration of 0.2. Both studies include variables such as age, RACH-1, previous stay, and preoperative ventilation; but our model also includes men, weight, Ross-modified functional class, and cyanotic heart disease. However, the characteristics of the population in the study by Kansy et al. differed from ours regarding younger age at the time of surgery (2.4 vs. 5.7 years) and fewer children with the weight percentile for age-sex <5% (35.2% vs. 63.8%). All the population of our study (100%) were children with ECC, while in the mentioned study, 80% of the population had ECC. The incidence of sepsis and mediastinitis was lower than what we found in this study, and there were no cases of endocarditis. On the other hand, the mentioned study did not include children with previous infections.
Sen et al. 30 evaluated bacterial sepsis and surgical site infection, such as mediastinitis, and found that risk factors for infection were younger age at surgery, greater surgical complexity, lower oxygen saturation, and other comorbidities; the incidence of sepsis was 5.5% and more than half of the cases were malnourished, data similar to those presented our study. They did not include children with previous infections, previous cardiac surgery, and mechanical ventilation; and they did not analyze preoperative length of stay. Our model includes variables similar to theirs, such as age, RACH-1, and cyanotic condition.
The scoring and nomogram tools created in this study can be easily applied in the clinical environment, which is a strength of this study. Also, both tools were developed considering the specific demographic characteristics of the studied population, which allows extrapolating the findings to similar circumstances that take place in South America and other developing countries. However, the study is not without its limitations. First, the number of patients with MPI was relatively small and belonged to a single health care facility in a specific setting. Second, the analysis and model did not include data such as the specific causative pathogen and use of preoperative prophylactic antibiotics, socioeconomic status like access to care, educational level, and family income. Third, when comparing the calibration of this model with that of the STS model (0.02 vs. 0.66; p > 0.050), it is demonstrated that our model does not fit properly, since what was observed differs from what was expected, and therefore more multicenter studies are required to obtain a more precise tool.
In conclusion, we designed a model to predict post-operative infection with adequate discrimination and good performance, which can be used as a quality control strategy and a tool for comparison with other pediatric heart surgery centers for better management decision making. The model proposed by STS was also validated and showed a good discriminative performance.
REFERENCES
1. Algra SO, Driessen MM, Schadenberg AW, Schouten AN, Haas F, Bollen CW, et al. Bedside prediction rule for infections after pediatric cardiac surgery. Intensive Care Med. 2012;38(3):474-481. doi: 10.1007/s00134-011-.2454-3. [ Links ]
2. Agarwal HS, Wolfram KB, Saville BR, Donahue BS, Bichell DP. Postoperative complications and association with outcomes in pediatric cardiac surgery. J Thorac Cardiovasc Surg. 2014;148(2):609-16. doi: 10.1016/j.jtcvs.2013.10.031 148(2):609-16. [ Links ]
3. Benavidez OJ, Gauvreau K, Del Nido P, Bacha E, Jenkins KJ. Complications and risk factors for mortality during congenital heart surgery admissions. Ann Thorac Surg. 2007;84(1):147-155. doi: 10.1016/j.athoracsur.2007.02.048. [ Links ]
4. Sarvikivi E, Lyytikäinen O, Nieminen H, Sairanen H, Saxén H. Nosocomial infections after pediatric cardiac surgery. Am J Infect Control. 2008;36(8):564-569. doi: 10.1016/j.ajic.2007.11.006. [ Links ]
5. Grisaru-Soen G, Paret G, Yahav D, Boyko V, Lerner-Geva L. Nosocomial infections in pediatric cardiovascular surgery patients: a 4-year survey. Pediatr Crit Care Med. 2009;10(2):202-206. doi: 10.1097/PCC.0b013e31819a37c5. [ Links ]
6. Guardia Camí MT, Jordan García I, Urrea Ayala M. Nosocomial infections in pediatric patients following cardiac surgery. An Pediatr (Barc). 2008;69(1):34-38. [ Links ]
7. Hasija S, Makhija N, Kiran U, Choudhary SK, Talwar S, Kapil A. Nosocomial infections in infants and children after cardiac surgery. Indian Journal of Thoracic and Cardiovascular Surgery. 2008;24:233. doi: 10.1007/s12055-008-0052-y. [ Links ]
8. Huang YF, Liu PY, Tang CW, Hsieh KS, Liu YC, Pan JY. Risk Factors and Outcome for Nosocomial Infections in Pediatric Cardiac Surgery. Int J Infect Dis. 2008;12:e352. doi: 10.1016/j.ijid.2008.05.939. [ Links ]
9. Levy I, Ovadia B, Erez E, Rinat S, Ashkenazi S, Birk E, et al. Nosocomial infections after cardiac surgery in infants and children: incidence and risk factors. J Hosp Infect. 2003;53(2): 111-116. [ Links ]
10. Valera M, Scolfaro C, Cappello N, Gramaglia E, Grassitelli S, Abbate MT, et al. Nosocomial infections in pediatric cardiac surgery, Italy. Infect Control Hosp Epidemiol. 2001;22(12):771-775. doi: 10.1086/501861. [ Links ]
11. Barker GM, O'Brien SM, Welke KF, Jacobs ML, Jacobs JP, Benjamin DK Jr, et al. Major infection after pediatric cardiac surgery: a risk estimation model. Ann Thorac Surg. 2010;89(3):843-850. doi: 10.1016/j.athoracsur.2009.11.048. [ Links ]
12. Kansy A, Jacobs JP, Pastuszko A, Mirkowicz-Malek M, Manowska M, Jezierska E, et al. Major infection after pediatric cardiac surgery: external validation of risk estimation model. Ann Thorac Surg. 2012;94(6):2091-2095. doi: 10.1016/j.athoracsur.2012.07.079. [ Links ]
13. Shah SS, Kagen J, Lautenbach E, Bilker WB, Matro J, Dominguez TE, et al. Bloodstream infections after median sternotomy at a children's hospital. J Thorac Cardiovasc Surg. 2007;133(2):435-440. doi: 10.1016/j.jtcvs.2006.09.026. [ Links ]
14. Li X, Wang X, Li S, Yan J, Li D. Diagnostic Value of Procalcitonin on Early Postoperative Infection After Pediatric Cardiac Surgery. Pediatr Crit Care Med. 2017;18(5):420-428. doi: 10.1097/PCC.0000000000001118. [ Links ]
15. Long CB, Shah SS, Lautenbach E, Coffin SE, Tabbutt S, Gaynor JW, et al. Postoperative mediastinitis in children: epidemiology, microbiology and risk factors for Gram-negative pathogens. Pediatr Infect Dis J. 2005;24(4):315-319. [ Links ]
16. Pasquali SK, He X, Jacobs ML, Hall M, Gaynor JW, Shah SS, et al. Hospital variation in postoperative infection and outcome after congenital heart surgery. Ann Thorac Surg. 2013;96(2):657-63. doi: 10.1016/j.athoracsur.2013.04.024. [ Links ]
17. Tortoriello TA, Friedman JD, McKenzie ED, Fraser C D, Feltes T F, Randall J, et al. Mediastinitis after pediatric cardiac surgery: a 15-year experience at a single institution. Ann Thorac Surg. 2003;76(5):1655-1660. [ Links ]
18. Mehta PA, Cunningham CK, Colella CB, Alferis G, Weiner LB. Risk factors for sternal wound and other infections in pediatric cardiac surgery patients. Pediatr Infect Dis J. 2000;19(10):1000-1004. [ Links ]
19. Vida VL, Leon-Wyss J, Larrazabal A, Cruz S, Castaneda AR. Mediastinitis in pediatric cardiac surgery: treatment and cost-effectiveness in a low-income country. Pediatr Cardiol. 2007;28(3):163-166. doi: 10.1007/s00246-006-0008-1. [ Links ]
20. Sidhu N, Joffe AR, Doughty P, Vatanpour S, Dinu I, Alton G, et al. Sepsis After Cardiac Surgery Early in Infancy and Adverse 4.5-Year Neurocognitive Outcomes. J Am Heart Assoc. 2015;4(8):e001954. doi: 10.1161/JAHA.115.001954. [ Links ]
21. The Society of Thoracic Surgeons. STS Congenital Heart Surgery Database Data Specifications Version 3.22 [Internet] STS; 2013 [citado el 30 de noviembre de 2019]. Disponible en: http://www.sts.org/sites/default/files/documents/CongenitalDataSpecsV3_22.pdf. [ Links ]
22. Turcotte RF, Brozovich A, Corda R, Demmer RT, Biagas KV, Mangino D, et al. Health care-associated infections in children after cardiac surgery. Pediatr Cardiol. 2014;35(8):1448-1455. doi: 10.1007/s00246-014-0953-z. [ Links ]
23. Hurtado-Sierra D, Calderón-Colmenero J, Curi-Curi P, Cervantes-Salazar J, Sandoval JP, García-Montes JA, et al. Outcomes of Delayed Sternal ons Practice Guideline Series: Antibiotic Prophylaxis in Cardiac Surgery, Part I: Duration. Ann Thor Surg. 2006;81(1):397-404. doi: 10.1016/j.athoracsur.2005.06.034. [ Links ]
24. Vickers AJ, Van Calster B, Steyerberg EW. Net benefit approaches to the evaluation of prediction models, molecular markers, and diagnostic tests. BMJ. 2016;25(352):i6. doi: https://doi.org/10.1136/bmj.i6. [ Links ]
25. Steyerberg EW, Vergouwe Y. Towards better clinical prediction models: seven steps for development and an ABCD for validation. Eur Heart J. 2014;35(29):1925-1931. doi: 10.1093/eurheartj/ehu207. [ Links ]
26. Royston P, Moons KG, Altman DG, Vergouwe Y. Prognosis and prognostic research: Developing a prognostic model. BMJ. 2009;338:b604. doi: 10.1136/bmj.b604. [ Links ]
27. Cai T, Mazzoli S, Migno S, Malossini G, Lanzafame P, Mereu L, et al. Development and validation of a nomogram predicting recurrence risk in women with symptomatic urinary tract infection. Int J Urol. 2014;21(9):929-934. doi: 10.1111/iju.12453. [ Links ]
28. Edwards FH, Engelman RM, Houck P, Shahian DM, Bridges CR. The Society of Thoracic Surgeons Practice Guideline Series: Antibiotic Prophylaxis in Cardiac Surgery, Part I: Duration. Ann Thor Surg. 2006;81(1):397-404. doi: 10.1016/j.athoracsur.2005.06.034. [ Links ]
29. Engelman R, Shahian D, Shemin R, Guy TS, Bratzler D, Edwards F, et al. The Society of Thoracic Surgeons Practice Guideline Series: Antibiotic Prophylaxis in Cardiac Surgery, Part II: Antibiotic Choice. Ann Thor Surg. 2007;83(4):1569-1576. doi: 10.1016/j.athoracsur.2006.09.046. [ Links ]
30. Sen AC, Morrow DF, Balachandran R, Du X, Gauvreau K, Jagannath BR, et al. Postoperative infection in developing world congenital heart surgery programs. Data from the International Quality Improvement Collaborative. Circ Cardiovasc Qual Outcomes. 2017;10(4):e002935. doi: 10.1161/CIRCOUTCOMES.116.002935. [ Links ]
Received: December 29, 2019; Accepted: September 09, 2020











 text in
text in